Key Insights
The global Liposomal Formulation Development Service market is poised for substantial expansion, projected to reach an estimated $28.4 million in 2025, driven by a compelling compound annual growth rate (CAGR) of 9.3% through 2033. This robust growth is fueled by the increasing demand for advanced drug delivery systems that enhance therapeutic efficacy and patient compliance. Liposomes, due to their biocompatibility, biodegradability, and ability to encapsulate both hydrophilic and hydrophobic drugs, are at the forefront of this evolution. The market is segmented by application into Research, Clinical, and Commercial, with significant activity expected across all, particularly in clinical development and commercialization phases as more liposomal drugs gain regulatory approval. The diverse range of drug types, including Small Molecules, Proteins, Peptides, and Nucleic Acids, further broadens the application scope and market potential.
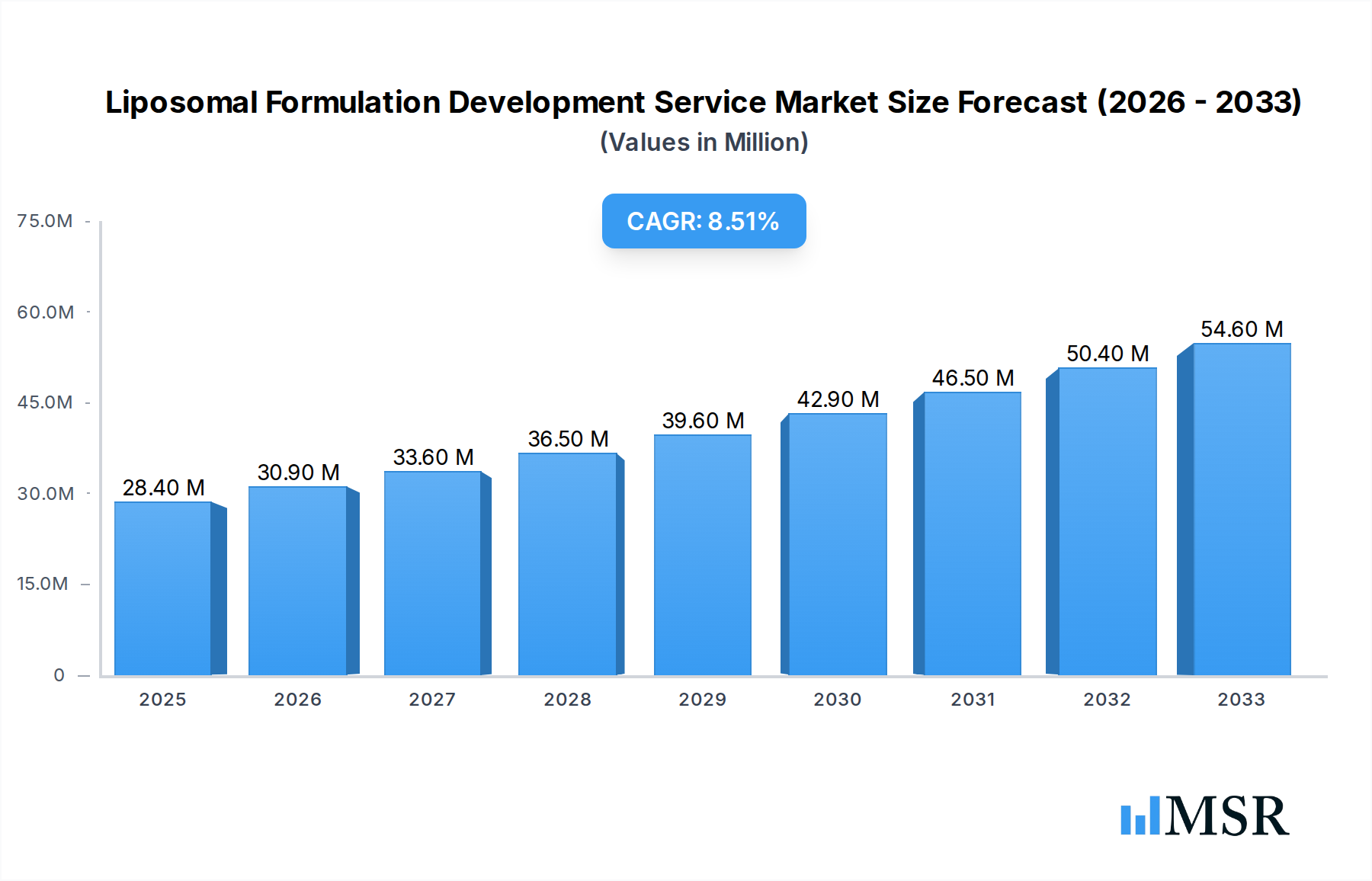
Liposomal Formulation Development Service Market Size (In Million)

Key growth drivers include the escalating prevalence of chronic diseases requiring novel treatment modalities, advancements in nanotechnology, and the growing investment in pharmaceutical R&D for targeted drug delivery. The increasing focus on personalized medicine and the development of novel therapeutics like mRNA vaccines and gene therapies are also significant contributors to market expansion. However, challenges such as the high cost of development and manufacturing, as well as stringent regulatory hurdles, could temper growth. Emerging trends like the development of stimuli-responsive liposomes and advancements in manufacturing technologies are expected to overcome some of these restraints. Prominent companies are actively investing in research and development, strategic collaborations, and expanding their service portfolios to capture a larger market share. North America and Europe currently lead the market, with the Asia Pacific region demonstrating rapid growth potential.
Liposomal Formulation Development Service Company Market Share

This in-depth report delivers a panoramic view of the global Liposomal Formulation Development Service market, offering crucial insights for industry stakeholders, researchers, and investors. Analyzing a historical period from 2019 to 2024 and forecasting through 2033, with a base and estimated year of 2025, this study uncovers market dynamics, key trends, and future growth trajectories. We meticulously examine market concentration, innovation, regulatory landscapes, and the evolving needs of end-users across research, clinical, and commercial applications, covering small molecules, proteins, peptides, nucleic acids, and other therapeutic modalities. With a projected market size of over one million dollars by the end of the forecast period, this report is an indispensable resource for navigating this rapidly expanding sector.
Liposomal Formulation Development Service Market Concentration & Dynamics
The Liposomal Formulation Development Service market exhibits a dynamic and evolving concentration, characterized by the presence of both established giants and agile innovators. While the market is not dominated by a single entity, a notable cluster of companies are driving innovation and market share. We estimate the market share of the top five players to be in the region of 60-70%, indicating a degree of consolidation. The innovation ecosystem is robust, with companies investing heavily in novel drug delivery systems, advanced encapsulation techniques, and targeted liposome designs. Regulatory frameworks, particularly those from agencies like the FDA and EMA, play a pivotal role in shaping development pathways and market access, often requiring extensive preclinical and clinical data. Substitute products, such as other nanoparticle-based drug delivery systems, represent a competitive force, yet the proven efficacy and biocompatibility of liposomes maintain their strong market position. End-user trends are strongly influenced by the increasing demand for targeted therapies, improved drug bioavailability, and reduced side effects, particularly in oncology and gene therapy. Mergers and acquisition (M&A) activities are a key indicator of market consolidation and strategic expansion; we anticipate approximately five to ten significant M&A deals annually within the forecast period, driven by the desire to acquire specialized expertise or expand service portfolios.
Liposomal Formulation Development Service Industry Insights & Trends
The global Liposomal Formulation Development Service market is poised for substantial growth, driven by an escalating demand for advanced drug delivery systems. The market size is projected to reach over one million dollars by 2033, with a Compound Annual Growth Rate (CAGR) of approximately 15-20% during the forecast period (2025-2033). This impressive growth is fueled by several key factors. Firstly, the increasing prevalence of chronic diseases, particularly cancer, necessitates the development of more effective and targeted therapeutic interventions, a domain where liposomal formulations excel. Their ability to encapsulate both hydrophobic and hydrophilic drugs, enhance drug solubility, protect sensitive biomolecules, and facilitate controlled release makes them ideal for a wide range of pharmaceutical applications.
Technological disruptions are continuously reshaping the industry. Advances in lipid nanoparticle engineering, including the development of novel lipid compositions and manufacturing processes like microfluidics, are leading to improved liposome stability, payload capacity, and targeted delivery efficiency. The rising prominence of mRNA vaccines and gene therapies has further amplified the demand for sophisticated delivery vehicles, with liposomes emerging as a leading platform for these next-generation therapeutics. For instance, the success of mRNA COVID-19 vaccines has underscored the potential of lipid-based nanoparticles, including liposomes, in delivering genetic material to target cells with high efficacy.
Evolving consumer behaviors, particularly in healthcare, are also contributing to market expansion. Patients and healthcare providers are increasingly seeking treatments that offer improved therapeutic outcomes with minimal adverse effects. Liposomal formulations, by enabling targeted drug delivery and reducing systemic exposure, align perfectly with this demand. Furthermore, the growing investment in research and development by pharmaceutical and biotechnology companies, coupled with increasing government funding for life sciences, provides a fertile ground for the growth of liposomal formulation development services. The ability to overcome biological barriers, such as the blood-brain barrier, and to deliver potent drugs to specific disease sites without causing widespread toxicity is a significant driver of innovation and market adoption. The flexibility of liposomal technology to accommodate various types of active pharmaceutical ingredients (APIs), from small molecules to complex biologics, further solidifies its market position.
Key Markets & Segments Leading Liposomal Formulation Development Service
The global Liposomal Formulation Development Service market is segmented across various applications and therapeutic types, with certain regions and segments demonstrating exceptional leadership.
Dominant Region: North America
North America, particularly the United States, stands as the leading market for liposomal formulation development services. This dominance is driven by several factors:
- Robust Pharmaceutical and Biotechnology Ecosystem: The presence of a high concentration of leading pharmaceutical and biotechnology companies, including Creative Biolabs, LipExoGen, Project Pharmaceutics, Polymun, Exelead, CD Formulation, Anglo Bio-Pharma Limited, Liposoma BV, Dalton Pharma Services, Creative Biostructure, BOC Sciences, Encapsula NanoSciences LLC, Fujifilm, CD Bioparticles, Precision NanoSystems, Evonik, Genevant Sciences, Avanti Polar Lipids, Nippon Fine Chemical, Lipoid, Corden Pharma, Acuitas Therapeutics, T&T Scientific, FormuMax Scientific, PlantaCorp, fosters significant demand for specialized formulation services.
- High R&D Investment: Substantial investments in research and development for novel drug discovery and delivery systems, exceeding one million dollars annually by many leading entities, propel the need for advanced liposomal technologies.
- Favorable Regulatory Environment: A well-established and supportive regulatory framework, coupled with a streamlined approval process for innovative drug delivery systems by agencies like the FDA, accelerates the translation of liposomal formulations from research to clinical application.
- Prevalence of Target Diseases: The high incidence of diseases like cancer, autoimmune disorders, and genetic diseases, which are prime candidates for liposomal drug delivery, further fuels market growth.
Leading Segments by Application:
- Clinical Application: This segment is experiencing the most significant growth. The increasing number of liposomal drug candidates entering clinical trials, driven by advancements in targeted therapies and personalized medicine, necessitates extensive formulation development and optimization services. The transition from preclinical to clinical stages represents a critical expenditure, with formulation services playing a pivotal role in ensuring drug safety, efficacy, and scalability.
- Commercial Application: As more liposomal drugs gain regulatory approval and reach the market, the demand for large-scale manufacturing and formulation services for commercial production is escalating. This segment is crucial for the long-term revenue generation of liposomal products.
- Research Application: While foundational, the research segment continues to be vital. Academic institutions and early-stage biotech companies rely on specialized services for proof-of-concept studies, preclinical investigations, and the development of novel liposomal platforms, laying the groundwork for future clinical and commercial successes.
Leading Segments by Type:
- Small Molecules: Liposomes have a long-standing history and established efficacy in delivering small molecule drugs, particularly those with poor solubility or low bioavailability. The ongoing development of new small molecule therapeutics ensures a consistent demand for liposomal formulation services in this category.
- Nucleic Acids: This segment is experiencing rapid growth, largely due to the burgeoning field of gene therapy and the success of mRNA vaccines. Liposomes are a key delivery vehicle for genetic material, offering protection from degradation and facilitating intracellular delivery. The demand for specialized expertise in formulating and manufacturing lipid nanoparticles for nucleic acid delivery is exceptionally high.
- Proteins and Peptides: While more complex to formulate, liposomal encapsulation of proteins and peptides is gaining traction for targeted delivery and enhanced stability. Advancements in formulation techniques are making this segment increasingly viable for therapeutic applications.
Liposomal Formulation Development Service Product Developments
Product developments in the liposomal formulation service sector are characterized by a strong focus on enhancing drug delivery efficacy, safety, and therapeutic outcomes. Innovations in lipid composition, such as the incorporation of novel functionalized lipids, are enabling improved targeting capabilities and triggered release mechanisms. Advanced manufacturing techniques, including microfluidics, are leading to greater control over liposome size, homogeneity, and scalability, which are critical for reproducible clinical and commercial production. Furthermore, there is a growing emphasis on developing liposomal formulations for challenging therapeutic modalities like nucleic acids (mRNA, siRNA) and peptides, opening up new avenues for treating genetic disorders, infectious diseases, and autoimmune conditions. The competitive edge lies in the ability to tailor liposomal characteristics to specific drug payloads and target tissues, thereby maximizing therapeutic impact and minimizing off-target effects.
Challenges in the Liposomal Formulation Development Service Market
Despite the promising growth, the Liposomal Formulation Development Service market faces several significant challenges.
- Regulatory Hurdles: Navigating the complex and evolving regulatory pathways for liposomal drug products can be time-consuming and costly. Demonstrating safety, efficacy, and manufacturing consistency to regulatory bodies like the FDA and EMA requires extensive data and rigorous adherence to Good Manufacturing Practices (GMP).
- Manufacturing Scalability and Cost: Scaling up liposomal production from laboratory to commercial quantities while maintaining quality and cost-effectiveness remains a persistent challenge. Specialized equipment and expertise are required, contributing to higher manufacturing costs compared to conventional drug formulations.
- Stability and Shelf-Life: Ensuring the long-term stability and shelf-life of liposomal formulations can be complex due to the inherent susceptibility of lipids to degradation. Developing formulations with extended stability without compromising efficacy or safety is crucial for market acceptance.
- Competitive Pressures: The increasing number of service providers, both established and emerging, intensifies competition, potentially impacting pricing and profit margins. Differentiation through specialized expertise, proprietary technologies, and a strong regulatory track record is essential.
Forces Driving Liposomal Formulation Development Service Growth
The Liposomal Formulation Development Service market is propelled by a confluence of technological advancements, favorable economic conditions, and evolving regulatory landscapes.
- Advancements in Nanotechnology: Continuous innovation in nanotechnology and material science, particularly in lipid engineering and controlled release technologies, enables the development of more sophisticated and effective liposomal drug delivery systems.
- Growing Demand for Targeted Therapies: The shift towards personalized medicine and targeted therapies, especially in oncology, immunology, and genetic disorders, directly fuels the need for advanced delivery platforms like liposomes that can selectively deliver drugs to diseased sites.
- Increasing Investment in Biopharmaceuticals: Significant investment from venture capital and pharmaceutical companies in the biopharmaceutical sector, particularly in areas like gene therapy and mRNA technologies, translates into higher demand for specialized liposomal formulation services.
- Emerging Markets and Applications: Expansion into new therapeutic areas and geographical markets, coupled with the potential of liposomes for non-pharmaceutical applications like cosmetics and diagnostics, creates new avenues for growth.
Challenges in the Liposomal Formulation Development Service Market
Long-term growth catalysts for the Liposomal Formulation Development Service market are rooted in persistent innovation, strategic collaborations, and a proactive approach to market expansion. The development of novel lipid chemistries that offer enhanced stability, improved cellular uptake, and intrinsic therapeutic properties will be a key differentiator. Strategic partnerships between liposome developers, pharmaceutical companies, and academic research institutions will accelerate the translation of cutting-edge discoveries into viable clinical products. Furthermore, the expansion of liposomal technology into under-served therapeutic areas and emerging geographical markets, where the need for advanced drug delivery is high, represents a significant growth opportunity. Continuous refinement of manufacturing processes to improve efficiency, reduce costs, and ensure consistent quality at scale will be critical for sustained market penetration and long-term success.
Emerging Opportunities in Liposomal Formulation Development Service
Emerging opportunities in the Liposomal Formulation Development Service market are diverse and rapidly evolving, driven by technological breakthroughs and unmet medical needs. The burgeoning field of CRISPR-based gene editing presents a significant opportunity for liposome developers, as these nanoparticles can serve as effective vehicles for delivering gene editing components. Furthermore, the expanding applications of liposomes in vaccine development, beyond mRNA platforms, for a wider range of infectious diseases and cancer immunotherapies, are creating new market frontiers. The growing interest in combination therapies, where liposomes can encapsulate multiple therapeutic agents for synergistic effects, offers another avenue for innovation. Additionally, the exploration of novel routes of administration, such as intranasal or ophthalmic delivery using liposomes, can address specific disease challenges and expand the therapeutic reach of liposomal formulations. The integration of artificial intelligence (AI) and machine learning (ML) in formulation design and optimization also presents an emerging opportunity to accelerate development timelines and enhance product performance.
Leading Players in the Liposomal Formulation Development Service Sector
- Creative Biolabs
- LipExoGen
- Project Pharmaceutics
- Polymun
- Exelead
- CD Formulation
- Anglo Bio-Pharma Limited
- Liposoma BV
- Dalton Pharma Services
- Creative Biostructure
- BOC Sciences
- Encapsula NanoSciences LLC
- Fujifilm
- CD Bioparticles
- Precision NanoSystems
- Evonik
- Genevant Sciences
- Avanti Polar Lipids
- Nippon Fine Chemical
- Lipoid
- Corden Pharma
- Acuitas Therapeutics
- T&T Scientific
- FormuMax Scientific
- PlantaCorp
Key Milestones in Liposomal Formulation Development Service Industry
- 2019: Increased investment in lipid nanoparticle (LNP) technology for RNA-based therapeutics.
- 2020-2021: Rapid development and global deployment of mRNA COVID-19 vaccines, showcasing the potential of liposomal delivery systems.
- 2020: Significant M&A activity as larger pharmaceutical companies acquire or partner with specialized LNP formulation companies.
- 2021: First approval of a liposomal formulation for a gene therapy, marking a new era for the technology.
- 2022: Advancements in continuous manufacturing technologies for liposomes, promising improved scalability and cost-efficiency.
- 2023: Growing focus on targeted liposomal delivery for oncology, aiming to reduce systemic toxicity and improve treatment efficacy.
- 2024: Emergence of new lipid compositions designed for enhanced cellular uptake and reduced immunogenicity.
Strategic Outlook for Liposomal Formulation Development Service Market
The strategic outlook for the Liposomal Formulation Development Service market is exceptionally positive, driven by robust growth accelerators including the relentless pursuit of novel drug delivery solutions, the increasing complexity of therapeutic payloads like gene therapies and biologics, and the expanding global demand for advanced pharmaceutical products. Companies that invest in proprietary technologies, foster strategic collaborations with drug developers, and maintain a strong regulatory track record will be best positioned for success. The market will continue to witness expansion driven by the therapeutic potential of liposomes in treating a wider array of diseases, coupled with advancements in manufacturing processes that enhance cost-effectiveness and scalability. Embracing innovation in lipid chemistry, particle engineering, and targeted delivery mechanisms will be paramount for capturing future market share and solidifying leadership in this dynamic sector.
Liposomal Formulation Development Service Segmentation
-
1. Application
- 1.1. Research
- 1.2. Clinical
- 1.3. Commercial
-
2. Types
- 2.1. Small Molecules
- 2.2. Proteins
- 2.3. Peptides
- 2.4. Nucleic Acids
- 2.5. Others
Liposomal Formulation Development Service Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
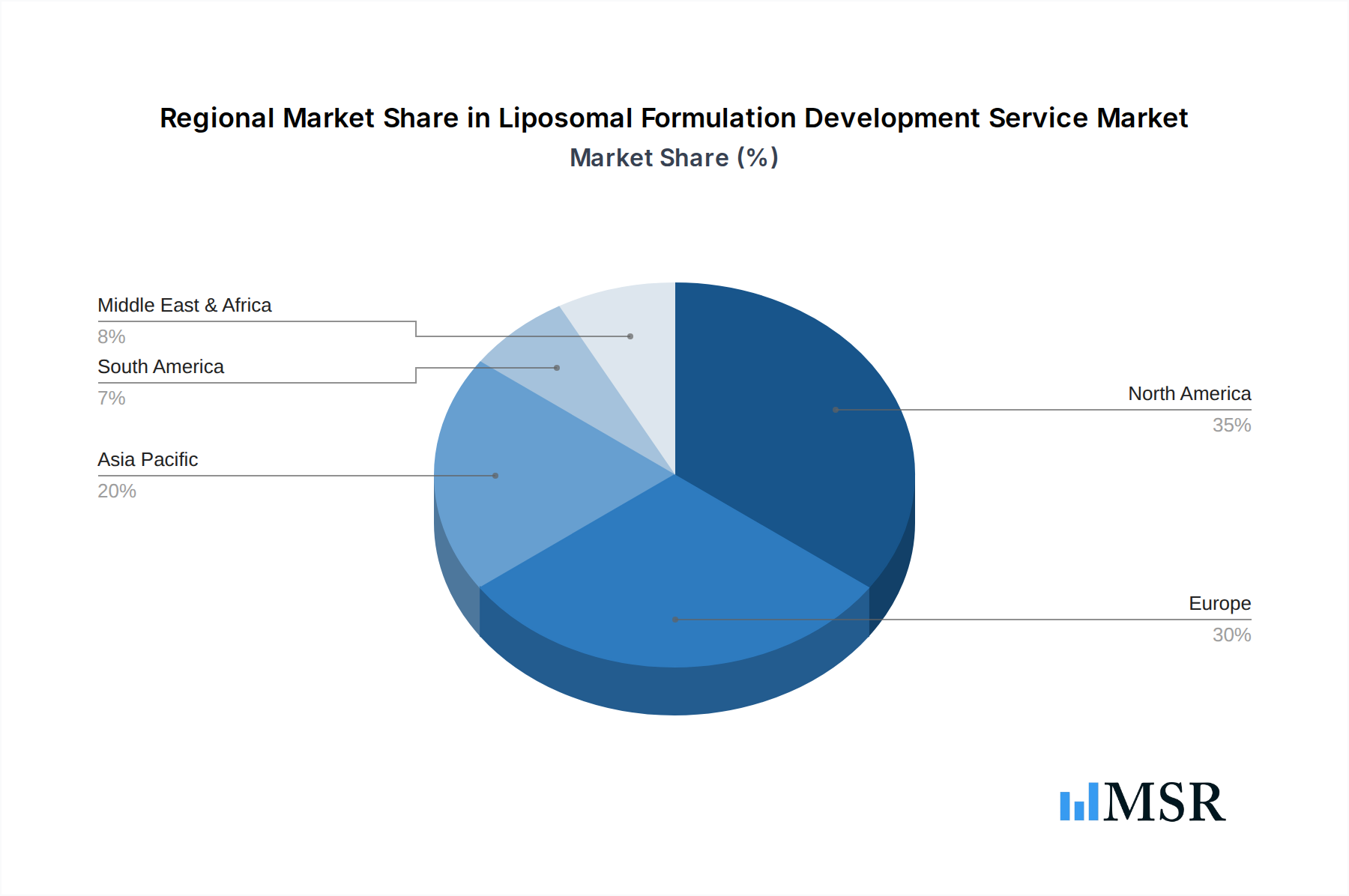
Liposomal Formulation Development Service Regional Market Share

Geographic Coverage of Liposomal Formulation Development Service
Liposomal Formulation Development Service REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.3% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. MSR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Research
- 5.1.2. Clinical
- 5.1.3. Commercial
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Small Molecules
- 5.2.2. Proteins
- 5.2.3. Peptides
- 5.2.4. Nucleic Acids
- 5.2.5. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Liposomal Formulation Development Service Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Research
- 6.1.2. Clinical
- 6.1.3. Commercial
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Small Molecules
- 6.2.2. Proteins
- 6.2.3. Peptides
- 6.2.4. Nucleic Acids
- 6.2.5. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Liposomal Formulation Development Service Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Research
- 7.1.2. Clinical
- 7.1.3. Commercial
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Small Molecules
- 7.2.2. Proteins
- 7.2.3. Peptides
- 7.2.4. Nucleic Acids
- 7.2.5. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Liposomal Formulation Development Service Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Research
- 8.1.2. Clinical
- 8.1.3. Commercial
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Small Molecules
- 8.2.2. Proteins
- 8.2.3. Peptides
- 8.2.4. Nucleic Acids
- 8.2.5. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Liposomal Formulation Development Service Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Research
- 9.1.2. Clinical
- 9.1.3. Commercial
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Small Molecules
- 9.2.2. Proteins
- 9.2.3. Peptides
- 9.2.4. Nucleic Acids
- 9.2.5. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Liposomal Formulation Development Service Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Research
- 10.1.2. Clinical
- 10.1.3. Commercial
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Small Molecules
- 10.2.2. Proteins
- 10.2.3. Peptides
- 10.2.4. Nucleic Acids
- 10.2.5. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Liposomal Formulation Development Service Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Research
- 11.1.2. Clinical
- 11.1.3. Commercial
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Small Molecules
- 11.2.2. Proteins
- 11.2.3. Peptides
- 11.2.4. Nucleic Acids
- 11.2.5. Others
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Creative Biolabs
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 LipExoGen
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Project Pharmaceutics
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Polymun
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Exelead
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 CD Formulation
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Anglo Bio-Pharma Limited
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Liposoma BV
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Dalton Pharma Services
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Creative Biostructure
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 BOC Sciences
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Encapsula NanoSciences LLC
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 Fujifilm
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.14 CD Bioparticles
- 12.1.14.1. Company Overview
- 12.1.14.2. Products
- 12.1.14.3. Company Financials
- 12.1.14.4. SWOT Analysis
- 12.1.15 Precision NanoSystems
- 12.1.15.1. Company Overview
- 12.1.15.2. Products
- 12.1.15.3. Company Financials
- 12.1.15.4. SWOT Analysis
- 12.1.16 Evonik
- 12.1.16.1. Company Overview
- 12.1.16.2. Products
- 12.1.16.3. Company Financials
- 12.1.16.4. SWOT Analysis
- 12.1.17 Genevant Sciences
- 12.1.17.1. Company Overview
- 12.1.17.2. Products
- 12.1.17.3. Company Financials
- 12.1.17.4. SWOT Analysis
- 12.1.18 Avanti Polar Lipids
- 12.1.18.1. Company Overview
- 12.1.18.2. Products
- 12.1.18.3. Company Financials
- 12.1.18.4. SWOT Analysis
- 12.1.19 Nippon Fine Chemical
- 12.1.19.1. Company Overview
- 12.1.19.2. Products
- 12.1.19.3. Company Financials
- 12.1.19.4. SWOT Analysis
- 12.1.20 Lipoid
- 12.1.20.1. Company Overview
- 12.1.20.2. Products
- 12.1.20.3. Company Financials
- 12.1.20.4. SWOT Analysis
- 12.1.21 Corden Pharma
- 12.1.21.1. Company Overview
- 12.1.21.2. Products
- 12.1.21.3. Company Financials
- 12.1.21.4. SWOT Analysis
- 12.1.22 Acuitas Therapeutics
- 12.1.22.1. Company Overview
- 12.1.22.2. Products
- 12.1.22.3. Company Financials
- 12.1.22.4. SWOT Analysis
- 12.1.23 T&T Scientific
- 12.1.23.1. Company Overview
- 12.1.23.2. Products
- 12.1.23.3. Company Financials
- 12.1.23.4. SWOT Analysis
- 12.1.24 FormuMax Scientific
- 12.1.24.1. Company Overview
- 12.1.24.2. Products
- 12.1.24.3. Company Financials
- 12.1.24.4. SWOT Analysis
- 12.1.25 PlantaCorp
- 12.1.25.1. Company Overview
- 12.1.25.2. Products
- 12.1.25.3. Company Financials
- 12.1.25.4. SWOT Analysis
- 12.1.1 Creative Biolabs
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Liposomal Formulation Development Service Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Liposomal Formulation Development Service Revenue (million), by Application 2025 & 2033
- Figure 3: North America Liposomal Formulation Development Service Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Liposomal Formulation Development Service Revenue (million), by Types 2025 & 2033
- Figure 5: North America Liposomal Formulation Development Service Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Liposomal Formulation Development Service Revenue (million), by Country 2025 & 2033
- Figure 7: North America Liposomal Formulation Development Service Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Liposomal Formulation Development Service Revenue (million), by Application 2025 & 2033
- Figure 9: South America Liposomal Formulation Development Service Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Liposomal Formulation Development Service Revenue (million), by Types 2025 & 2033
- Figure 11: South America Liposomal Formulation Development Service Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Liposomal Formulation Development Service Revenue (million), by Country 2025 & 2033
- Figure 13: South America Liposomal Formulation Development Service Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Liposomal Formulation Development Service Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Liposomal Formulation Development Service Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Liposomal Formulation Development Service Revenue (million), by Types 2025 & 2033
- Figure 17: Europe Liposomal Formulation Development Service Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Liposomal Formulation Development Service Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Liposomal Formulation Development Service Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Liposomal Formulation Development Service Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Liposomal Formulation Development Service Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Liposomal Formulation Development Service Revenue (million), by Types 2025 & 2033
- Figure 23: Middle East & Africa Liposomal Formulation Development Service Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Liposomal Formulation Development Service Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Liposomal Formulation Development Service Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Liposomal Formulation Development Service Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Liposomal Formulation Development Service Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Liposomal Formulation Development Service Revenue (million), by Types 2025 & 2033
- Figure 29: Asia Pacific Liposomal Formulation Development Service Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Liposomal Formulation Development Service Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Liposomal Formulation Development Service Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Liposomal Formulation Development Service Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Liposomal Formulation Development Service Revenue million Forecast, by Types 2020 & 2033
- Table 3: Global Liposomal Formulation Development Service Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Liposomal Formulation Development Service Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Liposomal Formulation Development Service Revenue million Forecast, by Types 2020 & 2033
- Table 6: Global Liposomal Formulation Development Service Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Liposomal Formulation Development Service Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Liposomal Formulation Development Service Revenue million Forecast, by Types 2020 & 2033
- Table 12: Global Liposomal Formulation Development Service Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Liposomal Formulation Development Service Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Liposomal Formulation Development Service Revenue million Forecast, by Types 2020 & 2033
- Table 18: Global Liposomal Formulation Development Service Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Liposomal Formulation Development Service Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Liposomal Formulation Development Service Revenue million Forecast, by Types 2020 & 2033
- Table 30: Global Liposomal Formulation Development Service Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Liposomal Formulation Development Service Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Liposomal Formulation Development Service Revenue million Forecast, by Types 2020 & 2033
- Table 39: Global Liposomal Formulation Development Service Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Liposomal Formulation Development Service Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Liposomal Formulation Development Service?
The projected CAGR is approximately 9.3%.
2. Which companies are prominent players in the Liposomal Formulation Development Service?
Key companies in the market include Creative Biolabs, LipExoGen, Project Pharmaceutics, Polymun, Exelead, CD Formulation, Anglo Bio-Pharma Limited, Liposoma BV, Dalton Pharma Services, Creative Biostructure, BOC Sciences, Encapsula NanoSciences LLC, Fujifilm, CD Bioparticles, Precision NanoSystems, Evonik, Genevant Sciences, Avanti Polar Lipids, Nippon Fine Chemical, Lipoid, Corden Pharma, Acuitas Therapeutics, T&T Scientific, FormuMax Scientific, PlantaCorp.
3. What are the main segments of the Liposomal Formulation Development Service?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 28.4 million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4900.00, USD 7350.00, and USD 9800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Liposomal Formulation Development Service," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Liposomal Formulation Development Service report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Liposomal Formulation Development Service?
To stay informed about further developments, trends, and reports in the Liposomal Formulation Development Service, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
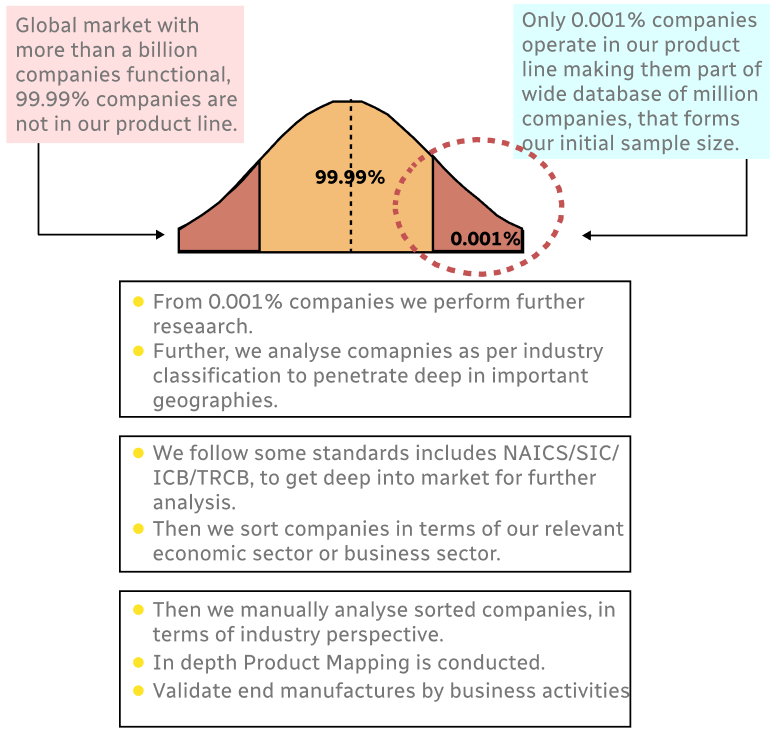
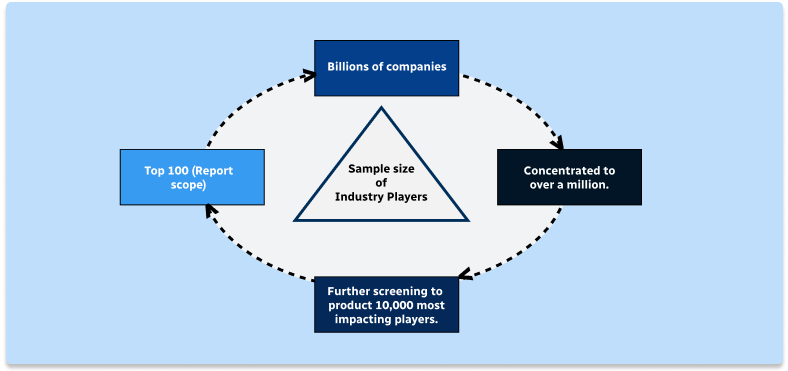
Step 1 - Identification of Relevant Samples Size from Population Database
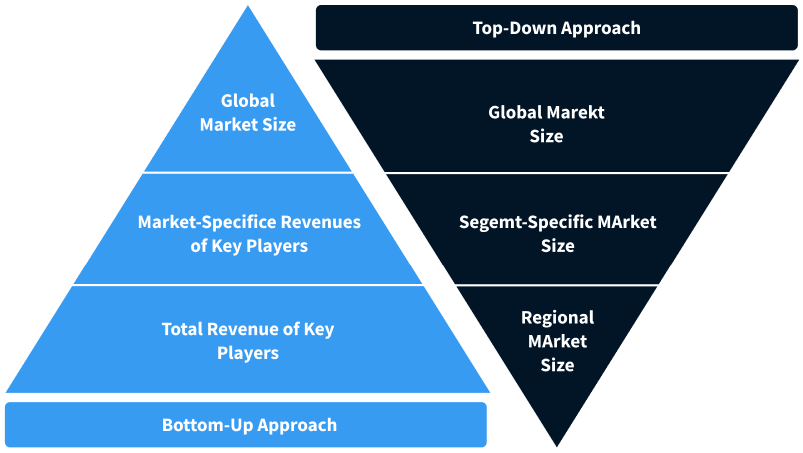
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
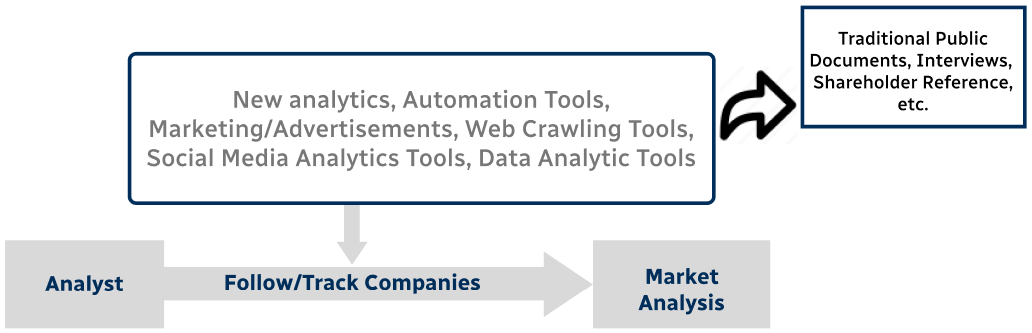
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


