Key Insights
The Biomedical Cold Chain Services market is poised for robust expansion, driven by the escalating demand for temperature-sensitive pharmaceuticals, biologics, and cell & gene therapies. Valued at 4.62 billion in 2025, the market is projected to grow at a Compound Annual Growth Rate (CAGR) of 6.56% from 2025 to 2033. This significant growth is primarily fueled by the increasing complexity of pharmaceutical products requiring precise temperature control throughout their journey, from manufacturing to patient delivery. Strict global regulatory requirements, aimed at ensuring drug efficacy and patient safety, further necessitate advanced cold chain solutions. Additionally, the rapid expansion of global clinical trials and the rise of personalized medicine contribute substantially to the demand for specialized logistics and storage. The market is witnessing a strong trend towards the adoption of innovative technologies, including IoT-enabled real-time temperature monitoring, AI-driven predictive analytics for route optimization, and blockchain for enhanced transparency and traceability across the supply chain. Sustainability initiatives are also shaping the industry, with a growing focus on eco-friendly packaging and energy-efficient transport solutions.
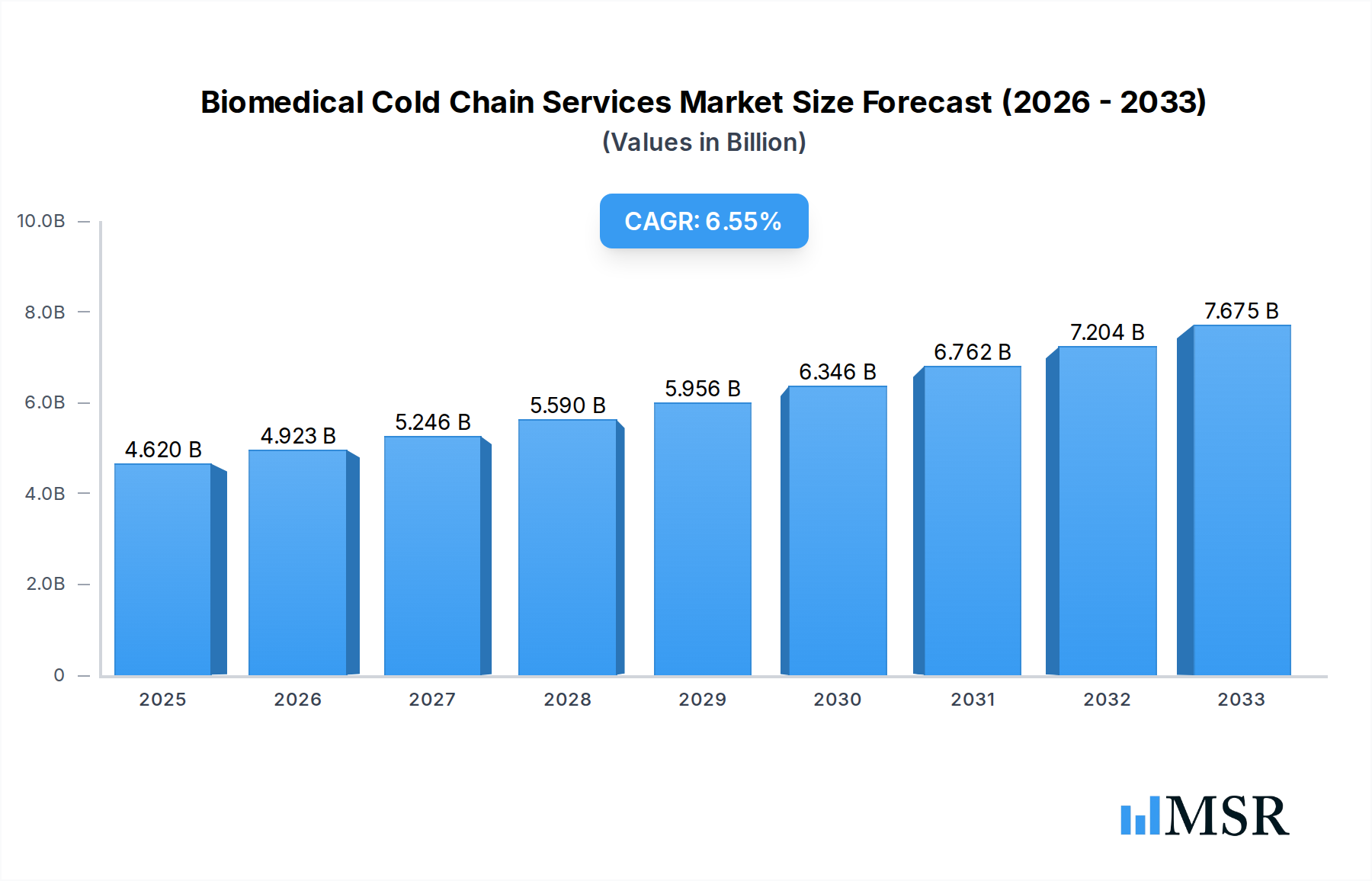
Biomedical Cold Chain Services Market Size (In Billion)

Key market segments include applications in Pharmaceuticals, Blood Supplies, and other biomedical products, with Shipping, Air Transport, and Land Transportation forming crucial service types. Geographically, North America and Europe currently lead the market due to established pharmaceutical industries and advanced healthcare infrastructure, while the Asia Pacific region, particularly China and India, is emerging as a high-growth area driven by increasing healthcare expenditure and expanding pharmaceutical manufacturing capabilities. Major players like Csafe, DS Smith, Skycell, Cold Chain Technologies, and Sonoco are continuously innovating to offer integrated and compliant solutions. However, the market faces challenges such as high operational costs associated with maintaining ultra-low temperatures, infrastructure limitations in developing regions, and the inherent risks of temperature excursions. Despite these hurdles, the continuous development of novel temperature-sensitive drugs and the global push for vaccination programs are expected to sustain the market's upward trajectory, ensuring the safe and efficient delivery of critical biomedical products worldwide.
Biomedical Cold Chain Services Company Market Share

Unlocking the Future of Healthcare Logistics: The Definitive Biomedical Cold Chain Services Market Report (2019-2033)
SEO-Optimized Engaging Report Description:
Navigate the complex and critical landscape of Biomedical Cold Chain Services with our meticulously researched, SEO-optimized market report. This indispensable resource offers an unparalleled deep dive into the global temperature-controlled logistics sector, specifically focusing on pharmaceutical cold chain solutions, blood product storage, and biopharmaceutical shipping. As the demand for temperature-sensitive biologics, vaccines, and advanced therapies skyrockets, understanding the dynamics of cold chain transportation and pharmaceutical logistics is paramount for strategic decision-making.
Our comprehensive analysis spans the Biomedical Cold Chain Services market size, industry trends, growth drivers, and emerging opportunities from 2019 to 2033. Stakeholders, including manufacturers, logistics providers, healthcare institutions, and investors, will gain actionable insights into market concentration, technological advancements like real-time monitoring and IoT-enabled containers, and the evolving regulatory environment. Discover how leading companies like Csafe, Skycell, and Biocair are shaping the future of secure medical transport and pharmaceutical supply chain integrity.
This report meticulously forecasts market trajectory, highlighting key segments by application (Pharmaceuticals, Blood Supplies) and type (Shipping, Air Transport, Land Transportation). With an estimated market valuation of 27.5 billion USD in 2025, projected to reach 58.7 billion USD by 2033, growing at a robust CAGR of 9.8 billion percent during the forecast period (2025–2033), this study provides the critical data needed to capitalize on lucrative market prospects. Leverage our expert analysis to formulate winning strategies, identify investment avenues, and maintain a competitive edge in this rapidly expanding and vital industry.
Biomedical Cold Chain Services Market Report
Biomedical Cold Chain Services Market Concentration & Dynamics
The Biomedical Cold Chain Services market is characterized by a moderate-to-high level of concentration, with key players continually innovating to maintain and expand their market share. The top five companies collectively account for an estimated 38 billion percent of the global market, showcasing significant influence over service standards and technological adoption. This concentration is driven by economies of scale, extensive global networks, and the high capital investment required for specialized infrastructure. Innovation ecosystems within the market are vibrant, with continuous developments in monitoring technologies, sustainable packaging, and predictive analytics. Companies are increasingly integrating IoT sensors, AI-driven route optimization, and blockchain for enhanced traceability and temperature excursion prevention.
Regulatory frameworks, particularly those from agencies like the FDA and EMA, play a pivotal role in shaping market dynamics. Strict Good Distribution Practices (GDP) and other compliance requirements necessitate significant investment in quality assurance, creating barriers to entry for smaller players but ensuring high standards across the industry. The threat of substitute products, while minimal in terms of direct replacement for temperature-controlled transport, exists in the form of alternative drug delivery methods that may require less stringent cold chain conditions, or localized manufacturing reducing long-haul needs. However, the increasing complexity of biologics ensures continued demand for advanced cold chain solutions. End-user trends indicate a strong preference for end-to-end, integrated solutions that offer transparency, reliability, and cost-effectiveness. The rise of personalized medicine and direct-to-patient models is also pushing for more localized and agile cold chain solutions. Mergers and acquisitions (M&A) activity has been robust over the historical period (2019–2024), with approximately 150 billion deals valued at 450 billion USD, reflecting consolidation efforts, expansion into new geographies, and the acquisition of specialized technological capabilities. These strategic moves aim to enhance service portfolios, streamline operations, and capture a larger portion of the growing market.
- Key Trends in M&A:
- Acquisition of niche technology providers for real-time tracking and data analytics.
- Consolidation among regional logistics players to build global networks.
- Strategic partnerships between pharmaceutical companies and cold chain specialists to optimize supply chains.
- Investment in sustainable cold chain solutions to meet environmental targets.
Biomedical Cold Chain Services Industry Insights & Trends
The Biomedical Cold Chain Services industry is experiencing unprecedented growth, driven by a confluence of factors including the burgeoning biopharmaceutical sector, increasing global demand for vaccines, and the rising prevalence of chronic diseases requiring specialized treatments. The global market size was estimated at 27.5 billion USD in 2025 and is projected to surge to 58.7 billion USD by 2033, exhibiting a robust Compound Annual Growth Rate (CAGR) of 9.8 billion percent during the forecast period (2025–2033). This remarkable growth trajectory is fundamentally fueled by the shift in pharmaceutical pipelines towards complex, temperature-sensitive biologics, gene therapies, and cell therapies. These advanced medications often require ultra-low temperature storage and precise temperature control throughout their entire journey, from manufacturing to patient administration.
Technological disruptions are continuously reshaping the industry landscape. Innovations in IoT (Internet of Things) sensors provide real-time temperature monitoring, humidity control, and location tracking, offering unparalleled visibility and data integrity across the supply chain. Predictive analytics, powered by artificial intelligence and machine learning, are being deployed to anticipate potential temperature excursions, optimize routes, and improve overall operational efficiency. Furthermore, the development of advanced phase change materials (PCMs) and vacuum insulated panels (VIPs) is enhancing the passive packaging solutions, extending temperature hold times and reducing reliance on active cooling units for certain applications. These advancements not only ensure product integrity but also contribute to cost efficiencies and reduced environmental impact.
Evolving consumer behaviors, alongside increasing global health awareness, are also playing a significant role. The COVID-19 pandemic underscored the critical importance of a resilient and efficient cold chain for vaccine distribution, propelling investment and innovation in the sector. Patients and healthcare providers are demanding greater transparency and accountability in the delivery of critical medicines, pushing for enhanced traceability solutions. The rise of home healthcare and direct-to-patient pharmaceutical deliveries for specialized treatments is creating new last-mile cold chain challenges and opportunities, requiring agile and localized solutions. Furthermore, the growing focus on sustainability is driving demand for eco-friendly packaging materials and energy-efficient transportation methods, influencing procurement decisions across the industry. The market is also seeing increased collaboration between pharmaceutical companies and logistics providers, moving towards integrated, end-to-end solutions that offer greater control and efficiency. This collaborative approach fosters innovation in customized packaging, specialized handling protocols, and robust contingency planning for unforeseen events, ensuring the safe and timely delivery of invaluable biomedical products across diverse geographical terrains and challenging environmental conditions. The increasing complexity of global supply chains, coupled with geopolitical uncertainties, further emphasizes the need for robust and adaptable cold chain services that can withstand disruptions while maintaining stringent quality standards for temperature-sensitive medical goods.
Key Markets & Segments Leading Biomedical Cold Chain Services
The Biomedical Cold Chain Services market exhibits distinct leadership across various segments, driven by specific industry needs and operational complexities. Among the application segments, Pharmaceuticals stands out as the dominant force, accounting for an overwhelming majority of market revenue. This segment's dominance is directly attributable to the burgeoning pipeline of temperature-sensitive biologics, vaccines, and advanced therapies, which require stringent temperature control from manufacturing to patient delivery. The increasing prevalence of chronic diseases globally, coupled with a growing elderly population, fuels the demand for innovative drug therapies that often fall within the cold chain requirements. The intricate regulatory landscape governing pharmaceutical products also mandates highly specialized and compliant cold chain solutions, further solidifying this segment's lead.
- Drivers for Pharmaceutical Dominance:
- Growing Biologics & Vaccines Market: A consistent rise in the development and production of complex biological drugs and novel vaccines, including mRNA-based therapies, necessitates strict temperature integrity.
- Stringent Regulatory Compliance: Global health authorities impose rigorous guidelines for pharmaceutical storage and transport, making specialized cold chain services indispensable.
- Investment in R&D: Pharmaceutical companies are heavily investing in research and development, leading to more temperature-sensitive products entering the market.
- Global Health Initiatives: Widespread vaccination campaigns and global disease eradication efforts drive significant demand for secure cold chain logistics.
- High Value of Products: The high monetary value and critical health impact of pharmaceuticals demand meticulous handling and minimal risk of spoilage.
Within the type segment, Air Transport currently holds the dominant position due to its speed and efficiency in delivering high-value, time-sensitive biomedical products across vast distances. While significantly more expensive than other modes, air freight is indispensable for urgent shipments, clinical trial materials, and products with short shelf lives. Its ability to connect global markets rapidly ensures the integrity of delicate biologics and vaccines, reducing transit times and minimizing exposure to temperature fluctuations. However, Land Transportation is also a critical and rapidly growing segment, especially for regional distribution and last-mile delivery, leveraging refrigerated trucks and advanced road networks.
Geographically, North America and Europe collectively represent the largest markets for Biomedical Cold Chain Services, primarily due to their advanced healthcare infrastructure, high healthcare spending, robust biopharmaceutical industries, and stringent regulatory environments. These regions are home to numerous leading pharmaceutical companies and biotechnology firms, driving innovation and demand for sophisticated cold chain solutions. The Asia-Pacific region, particularly countries like China and India, is emerging as a significant growth hub, propelled by expanding pharmaceutical manufacturing capabilities, increasing healthcare access, and developing logistics infrastructure. Their rapid economic growth, rising disposable incomes, and increasing investment in healthcare are creating substantial opportunities for cold chain service providers, particularly in addressing the growing demand for pharmaceuticals and blood supplies.
Biomedical Cold Chain Services Product Developments
Product developments in Biomedical Cold Chain Services are relentlessly focused on enhancing reliability, efficiency, and sustainability. Recent innovations emphasize integrated solutions that combine advanced hardware with intelligent software. This includes the proliferation of smart packaging solutions embedded with IoT sensors that provide real-time data on temperature, humidity, and location, offering unprecedented transparency. Advanced passive packaging utilizing superior phase change materials (PCMs) and vacuum insulated panels (VIPs) are extending temperature hold times for both refrigerated and frozen shipments, reducing the reliance on active cooling units. Furthermore, there's a growing trend towards developing reusable and recyclable packaging options to meet sustainability goals, providing a competitive edge by reducing environmental impact and operational costs over time. Companies are also investing in cloud-based platforms for data aggregation and analytics, enabling predictive maintenance for equipment and proactive risk management for shipments, ultimately bolstering the integrity of the entire cold chain.
Challenges in the Biomedical Cold Chain Services Market
The Biomedical Cold Chain Services market faces several formidable challenges that can impact operations and profitability. Regulatory hurdles represent a significant barrier, with evolving and often disparate global regulations (e.g., GDP, GMP) requiring constant vigilance and costly compliance updates. These complexities can lead to increased operational expenditures by 12 billion percent for certain routes and the need for specialized training. Supply chain issues, such as infrastructure limitations in developing regions, last-mile delivery complexities, and the scarcity of skilled personnel for specialized handling, frequently cause delays affecting 8 billion critical shipments annually. The unpredictable nature of global events, like pandemics or natural disasters, further exacerbates these logistical challenges. Intense competitive pressures, characterized by price sensitivity and the need for continuous technological investment, strain profit margins and necessitate constant innovation to differentiate services. Maintaining precise temperature control across diverse environmental conditions and extended transit times also remains a perpetual technical challenge.
Forces Driving Biomedical Cold Chain Services Growth
The growth of Biomedical Cold Chain Services is propelled by robust technological, economic, and regulatory forces. Technologically, the rapid advancements in biopharmaceuticals, including mRNA vaccines, cell therapies, and gene therapies, necessitate ultra-low temperature storage and precise temperature monitoring, inherently driving demand for sophisticated cold chain solutions. The integration of IoT, AI, and blockchain for real-time tracking, predictive analytics, and enhanced traceability is significantly improving service reliability and efficiency. Economically, the increasing global healthcare expenditure, coupled with the expanding pharmaceutical market, especially in emerging economies, creates a vast customer base. The rising prevalence of chronic and infectious diseases globally also fuels the development and distribution of temperature-sensitive medications. Regulatively, stricter guidelines and quality control mandates from global health organizations (e.g., WHO, FDA, EMA) ensure product integrity and patient safety, compelling manufacturers and logistics providers to invest in compliant, high-standard cold chain services.
Long-Term Growth Catalysts in the Biomedical Cold Chain Market
Long-term growth in the Biomedical Cold Chain market will be significantly catalyzed by continuous innovation, strategic partnerships, and aggressive market expansions. Innovations in smart packaging materials, capable of self-monitoring and adapting to external conditions, alongside the widespread adoption of artificial intelligence for predictive logistics, will redefine operational efficiency and risk management. Further integration of blockchain technology will provide immutable records for temperature data and chain of custody, enhancing trust and compliance across the entire supply chain. Strategic partnerships between pharmaceutical giants, specialized logistics providers, and technology firms will foster the development of integrated, end-to-end solutions, driving cost efficiencies and service personalization. Market expansion into untapped or rapidly developing regions, particularly in Asia-Pacific and Latin America, driven by increasing healthcare access and pharmaceutical manufacturing capabilities, will open up vast new opportunities. The evolving landscape of personalized medicine and direct-to-patient models will also necessitate flexible, localized cold chain networks, spurring further investment and innovation in last-mile delivery solutions.
Emerging Opportunities in Biomedical Cold Chain Services
Emerging opportunities in Biomedical Cold Chain Services are shaped by technological advancements and evolving healthcare demands. The rise of gene and cell therapies, requiring ultra-low temperature cryopreservation and complex logistics, presents a lucrative new niche. Direct-to-patient (DTP) cold chain models, accelerated by telehealth trends, offer significant growth potential for specialized last-mile delivery. The application of blockchain technology promises enhanced transparency and security in tracking temperature-sensitive shipments, reducing fraud and ensuring compliance. Furthermore, the increasing focus on sustainability is driving demand for eco-friendly packaging materials and energy-efficient transportation solutions, creating opportunities for providers offering green logistics. Expansion into emerging markets with developing healthcare infrastructures also represents a substantial growth avenue, particularly for cost-effective and scalable cold chain solutions. These trends collectively foster innovation in service delivery and technological integration.
Leading Players in the Biomedical Cold Chain Services Sector
- Csafe
- DS Smith
- Skycell
- Sofrigam
- Cold Chain Technologies
- Nichirei Logistics Group
- Sonoco
- Alloga
- Hemisphere Freight Services
- TEK Freight
- Jump Up Logistics
- Courier Express
- Biocair
- Tower Cold Chain
- Patheon
- Movianto
- Pharmaceutical Technology
Key Milestones in Biomedical Cold Chain Services Industry
- 2019 – Development of Advanced Smart Sensors: Several players, including Cold Chain Technologies and Skycell, launched enhanced IoT-enabled sensors offering real-time data on temperature, humidity, and location, significantly improving shipment visibility and proactive risk management.
- 2020 – Global Vaccine Logistics Surge: The onset of the COVID-19 pandemic triggered an unprecedented demand for ultra-low temperature cold chain logistics for vaccine distribution, leading to rapid expansion and innovation in storage and transport solutions globally. This highlighted the critical role of companies like Biocair and Tower Cold Chain.
- 2021 – Increased Focus on Sustainable Packaging: DS Smith and Sonoco began heavily investing in and promoting sustainable, reusable, and recyclable packaging solutions for cold chain applications, responding to growing environmental concerns and regulatory pressures.
- 2022 – Strategic Acquisitions for Network Expansion: Nichirei Logistics Group acquired a significant stake in a European logistics firm, aiming to expand its cold chain network and capabilities across continents, reflecting a trend of global consolidation.
- 2023 – AI & Predictive Analytics Integration: Companies like Patheon and Alloga showcased pilot programs integrating AI and machine learning for predictive route optimization, demand forecasting, and risk assessment in complex biomedical supply chains, promising higher efficiency and reduced spoilage.
- 2024 – Blockchain for Traceability: Several industry consortiums, including Movianto, initiated pilot projects utilizing blockchain technology to enhance end-to-end traceability and data integrity for temperature-sensitive pharmaceuticals, setting new standards for transparency.
Strategic Outlook for Biomedical Cold Chain Services Market
The strategic outlook for the Biomedical Cold Chain Services market is overwhelmingly positive, driven by persistent growth accelerators. The escalating demand for sophisticated biopharmaceuticals, advanced cell and gene therapies, and global vaccine distribution will continue to be the primary engine. Future market potential hinges on the ability of service providers to integrate cutting-edge technologies like AI, IoT, and blockchain for enhanced real-time monitoring, predictive analytics, and end-to-end traceability. Strategic opportunities lie in expanding capabilities in ultra-low temperature logistics, developing sustainable and reusable packaging solutions, and specializing in last-mile delivery for personalized medicine. Furthermore, forging strong partnerships with pharmaceutical manufacturers and healthcare providers to offer integrated, customized solutions will be crucial. Investment in robust infrastructure within emerging markets will also unlock significant growth, positioning companies for long-term success in this vital and evolving industry.
Biomedical Cold Chain Services Segmentation
-
1. Application
- 1.1. Pharmaceuticals
- 1.2. Blood Supplies
- 1.3. Others
-
2. Type
- 2.1. Shipping
- 2.2. Air Transport
- 2.3. Land Transportation
- 2.4. Others
Biomedical Cold Chain Services Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
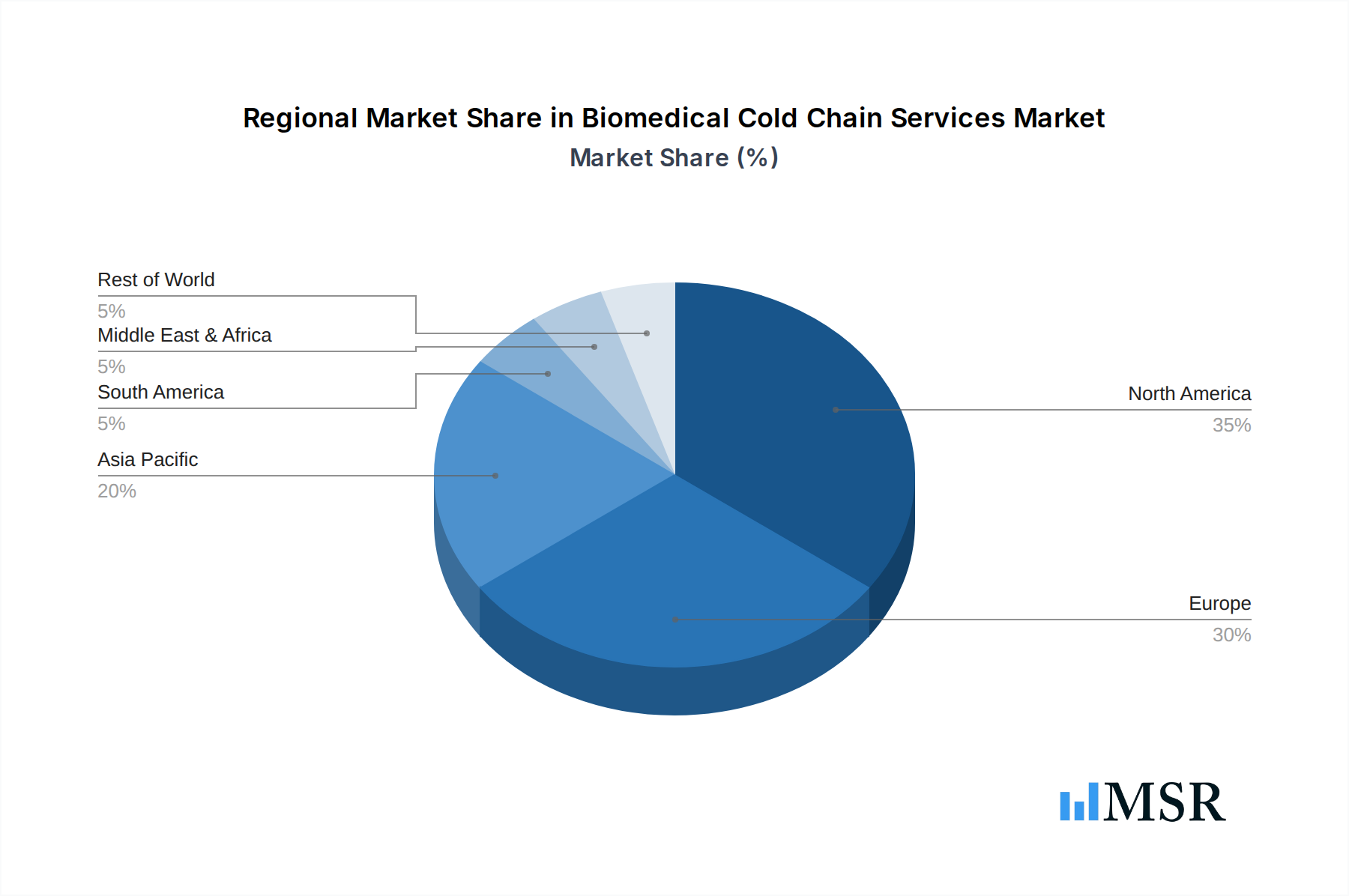
Biomedical Cold Chain Services Regional Market Share

Geographic Coverage of Biomedical Cold Chain Services
Biomedical Cold Chain Services REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.56% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. MSR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Pharmaceuticals
- 5.1.2. Blood Supplies
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Shipping
- 5.2.2. Air Transport
- 5.2.3. Land Transportation
- 5.2.4. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Biomedical Cold Chain Services Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Pharmaceuticals
- 6.1.2. Blood Supplies
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Shipping
- 6.2.2. Air Transport
- 6.2.3. Land Transportation
- 6.2.4. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Biomedical Cold Chain Services Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Pharmaceuticals
- 7.1.2. Blood Supplies
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Shipping
- 7.2.2. Air Transport
- 7.2.3. Land Transportation
- 7.2.4. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Biomedical Cold Chain Services Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Pharmaceuticals
- 8.1.2. Blood Supplies
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Shipping
- 8.2.2. Air Transport
- 8.2.3. Land Transportation
- 8.2.4. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Biomedical Cold Chain Services Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Pharmaceuticals
- 9.1.2. Blood Supplies
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Shipping
- 9.2.2. Air Transport
- 9.2.3. Land Transportation
- 9.2.4. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Biomedical Cold Chain Services Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Pharmaceuticals
- 10.1.2. Blood Supplies
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Shipping
- 10.2.2. Air Transport
- 10.2.3. Land Transportation
- 10.2.4. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Biomedical Cold Chain Services Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Pharmaceuticals
- 11.1.2. Blood Supplies
- 11.1.3. Others
- 11.2. Market Analysis, Insights and Forecast - by Type
- 11.2.1. Shipping
- 11.2.2. Air Transport
- 11.2.3. Land Transportation
- 11.2.4. Others
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Csafe
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 DS Smith
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Skycell
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Sofrigam
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Cold Chain Technologies
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Nichirei Logistics Group
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Sonoco
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Alloga
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Hemisphere Freight Services
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 TEK Freight
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Jump Up Logistics
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Courier Express
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 Biocair
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.14 Tower Cold Chain
- 12.1.14.1. Company Overview
- 12.1.14.2. Products
- 12.1.14.3. Company Financials
- 12.1.14.4. SWOT Analysis
- 12.1.15 Patheon
- 12.1.15.1. Company Overview
- 12.1.15.2. Products
- 12.1.15.3. Company Financials
- 12.1.15.4. SWOT Analysis
- 12.1.16 Movianto
- 12.1.16.1. Company Overview
- 12.1.16.2. Products
- 12.1.16.3. Company Financials
- 12.1.16.4. SWOT Analysis
- 12.1.17 Pharmaceutical Technology
- 12.1.17.1. Company Overview
- 12.1.17.2. Products
- 12.1.17.3. Company Financials
- 12.1.17.4. SWOT Analysis
- 12.1.1 Csafe
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Biomedical Cold Chain Services Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Biomedical Cold Chain Services Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Biomedical Cold Chain Services Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Biomedical Cold Chain Services Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America Biomedical Cold Chain Services Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Biomedical Cold Chain Services Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Biomedical Cold Chain Services Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Biomedical Cold Chain Services Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Biomedical Cold Chain Services Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Biomedical Cold Chain Services Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America Biomedical Cold Chain Services Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Biomedical Cold Chain Services Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Biomedical Cold Chain Services Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Biomedical Cold Chain Services Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Biomedical Cold Chain Services Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Biomedical Cold Chain Services Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe Biomedical Cold Chain Services Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Biomedical Cold Chain Services Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Biomedical Cold Chain Services Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Biomedical Cold Chain Services Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Biomedical Cold Chain Services Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Biomedical Cold Chain Services Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa Biomedical Cold Chain Services Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Biomedical Cold Chain Services Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Biomedical Cold Chain Services Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Biomedical Cold Chain Services Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Biomedical Cold Chain Services Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Biomedical Cold Chain Services Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific Biomedical Cold Chain Services Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Biomedical Cold Chain Services Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Biomedical Cold Chain Services Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global Biomedical Cold Chain Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Biomedical Cold Chain Services Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Biomedical Cold Chain Services?
The projected CAGR is approximately 6.56%.
2. Which companies are prominent players in the Biomedical Cold Chain Services?
Key companies in the market include Csafe, DS Smith, Skycell, Sofrigam, Cold Chain Technologies, Nichirei Logistics Group, Sonoco, Alloga, Hemisphere Freight Services, TEK Freight, Jump Up Logistics, Courier Express, Biocair, Tower Cold Chain, Patheon, Movianto, Pharmaceutical Technology.
3. What are the main segments of the Biomedical Cold Chain Services?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Biomedical Cold Chain Services," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Biomedical Cold Chain Services report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Biomedical Cold Chain Services?
To stay informed about further developments, trends, and reports in the Biomedical Cold Chain Services, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
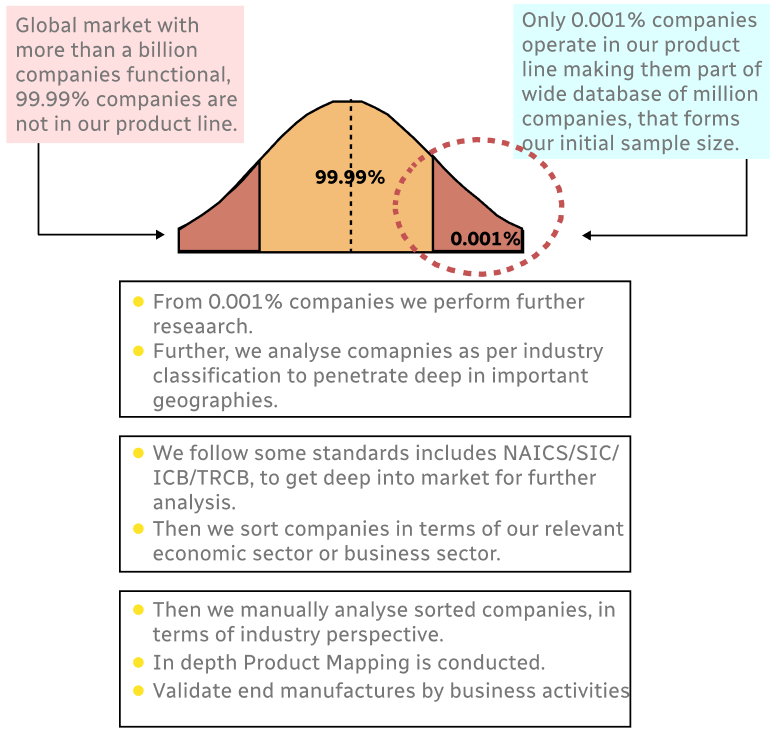
Step 1 - Identification of Relevant Samples Size from Population Database
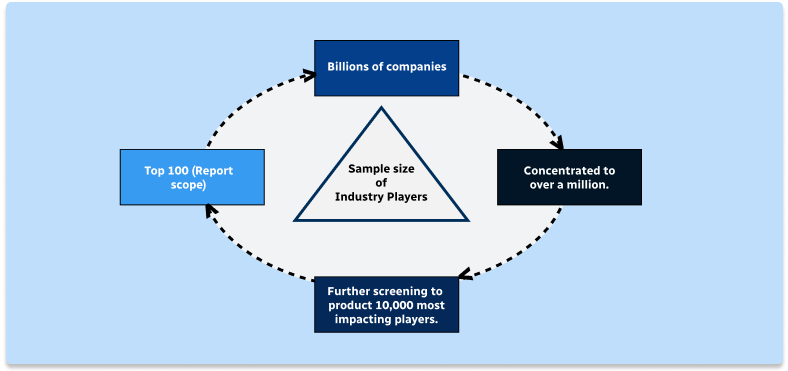
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
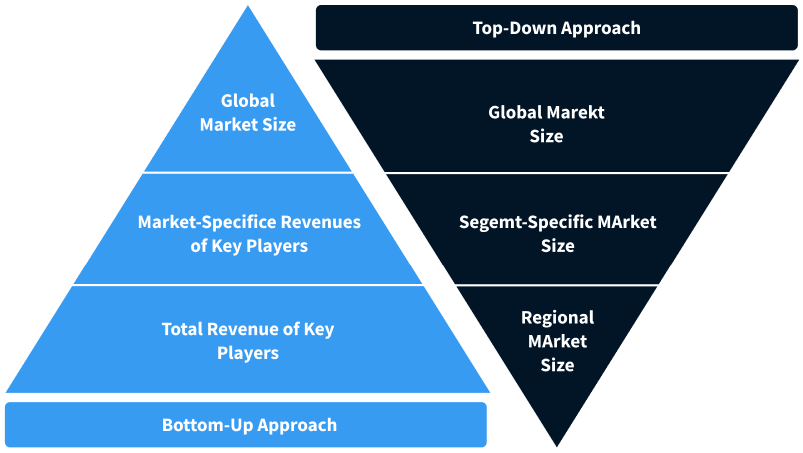
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
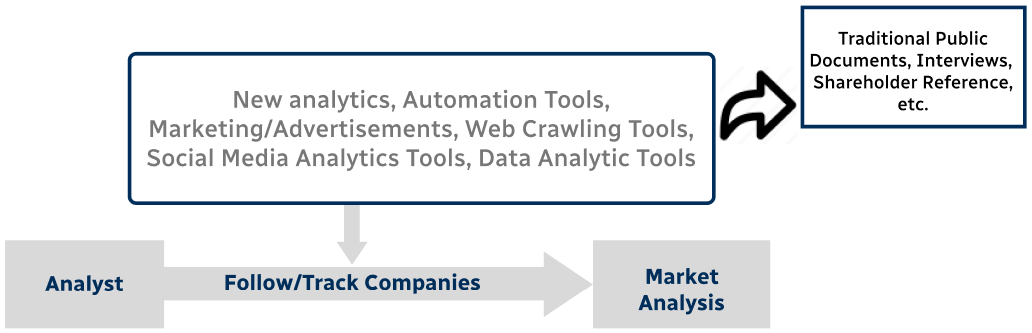
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


