Key Insights
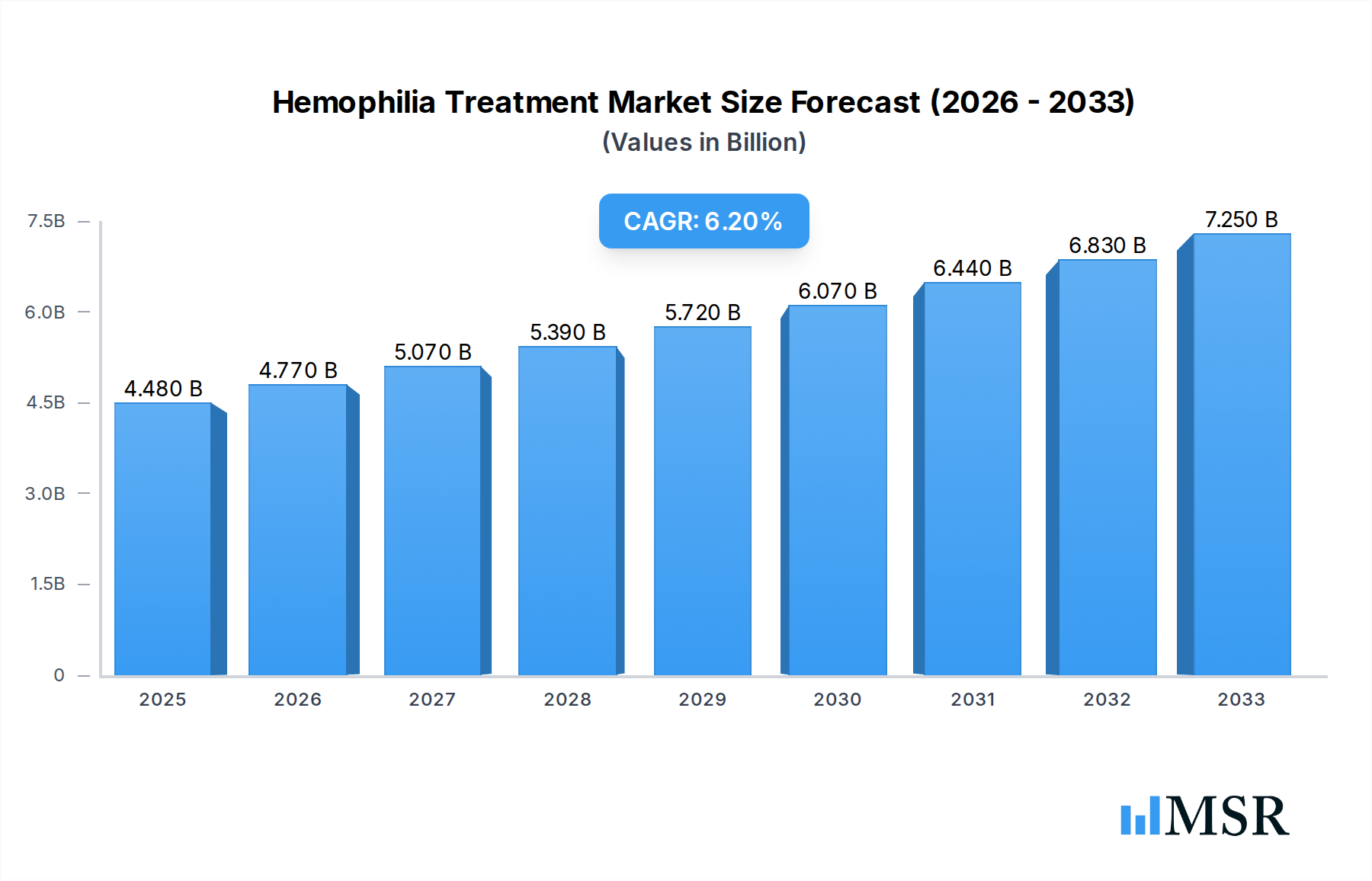
The global Hemophilia Treatment market is projected for substantial growth, anticipated to reach $4.48 billion by 2025 and expand at a robust Compound Annual Growth Rate (CAGR) of 6.4% through the forecast period of 2025-2033. This upward trajectory is primarily fueled by advancements in treatment modalities, including the increasing adoption of gene therapy and novel prophylactic treatments that offer improved patient outcomes and quality of life. The rising global prevalence of hemophilia, coupled with increased awareness and improved diagnostic capabilities, also contributes significantly to market expansion. Furthermore, the growing focus on personalized medicine and the development of targeted therapies are creating new avenues for market players, driving innovation and investment. The market is segmented into various applications such as Replacement Therapy, ITI Therapy, and Gene Therapy, with gene therapy emerging as a key growth driver due to its potential for long-term disease management.
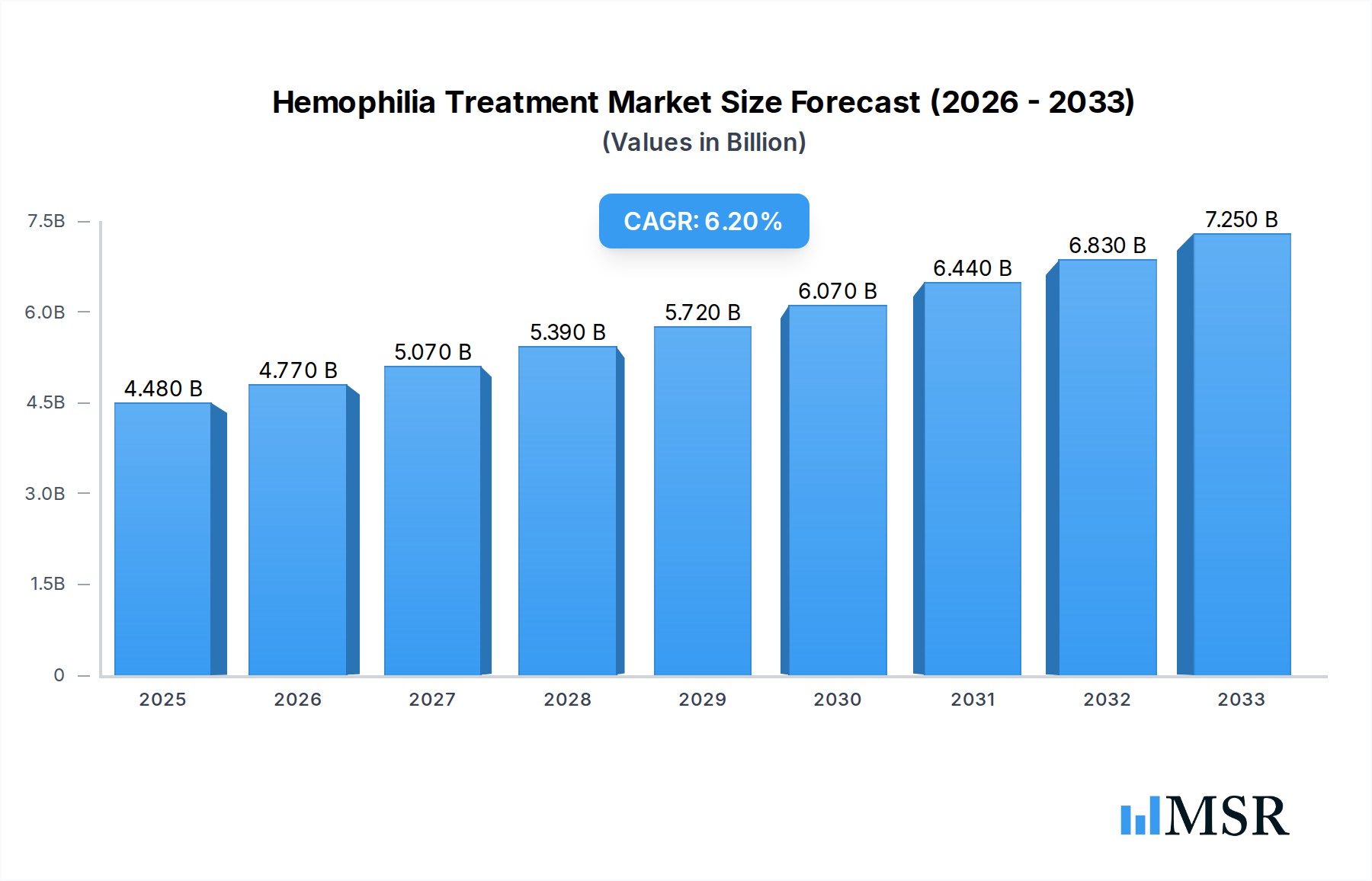
Hemophilia Treatment Market Size (In Billion)

The Hemophilia Treatment market is being significantly shaped by several key trends, including the development of extended half-life (EHL) factor concentrates, which reduce the frequency of infusions and improve patient adherence. The rise of home-based treatment and self-infusion among patients, empowered by technological advancements and improved understanding of the disease, is another crucial trend. Key companies like CSL Behring, Baxalta, Pfizer Inc., BioMarin, Bayer Healthcare, Biogen, Novo Nordisk, Roche, and Takeda are heavily investing in research and development to bring innovative treatments to market, including gene therapies and bispecific antibodies. While the market shows strong growth potential, challenges such as the high cost of novel treatments, particularly gene therapy, and reimbursement complexities in certain regions could act as restraints. North America and Europe currently lead the market due to advanced healthcare infrastructure and higher patient awareness, but the Asia Pacific region is expected to witness significant growth driven by increasing healthcare expenditure and a growing patient pool.
Hemophilia Treatment Company Market Share

Hemophilia Treatment Market: A Comprehensive Analysis and Forecast (2019–2033)
This in-depth report provides a definitive analysis of the global Hemophilia Treatment market, encompassing a detailed study of its dynamics, trends, key segments, product developments, challenges, growth drivers, and emerging opportunities. The report leverages a robust Study Period: 2019–2033, with a Base Year: 2025, Estimated Year: 2025, and a comprehensive Forecast Period: 2025–2033. It offers actionable insights for stakeholders, including pharmaceutical manufacturers, biotech companies, research institutions, investors, and healthcare providers, aiming to capitalize on the evolving landscape of hemophilia therapeutics.
Hemophilia Treatment Market Concentration & Dynamics
The global Hemophilia Treatment market exhibits a moderate to high concentration, characterized by the significant influence of established players and a burgeoning innovation ecosystem. Key companies like CSL Behring, Baxalta, Pfizer Inc, BioMarin, Bayer Healthcare, Biogen, Novo Nordisk, Roche, and Takeda hold substantial market shares, driven by their robust product pipelines and strategic investments in research and development. The innovation ecosystem is rapidly evolving, with a strong focus on Gene Therapy and novel ITI Therapy approaches, alongside the traditional Replacement Therapy. Regulatory frameworks, particularly in North America and Europe, are sophisticated, with a growing emphasis on patient access and reimbursement policies for advanced treatments. Substitute products, primarily supportive therapies and lifestyle management, play a secondary role but are crucial for comprehensive patient care. End-user trends highlight a growing demand for more convenient and less invasive treatment modalities, driving the shift towards prophylactic treatments and at-home infusion systems. Mergers and Acquisitions (M&A) activities remain a significant dynamic, with an estimated XX M&A deals recorded in the historical period, aimed at consolidating market presence, acquiring innovative technologies, and expanding geographical reach. The market share of leading players collectively accounts for over $XXX billion.
Hemophilia Treatment Industry Insights & Trends
The Hemophilia Treatment industry is poised for substantial growth, projected to reach a global market size of over $XXX billion by the end of the forecast period. The Compound Annual Growth Rate (CAGR) is estimated at XX.X%, fueled by a confluence of factors including increasing hemophilia diagnoses, advancements in treatment methodologies, and expanding access to healthcare globally. Technological disruptions are at the forefront, with Gene Therapy emerging as a transformative force, promising long-term remission or a potential cure for hemophilia patients. The development of bispecific antibodies and extended half-life clotting factor concentrates is also revolutionizing Replacement Therapy, offering improved patient convenience and reduced treatment burden. Evolving consumer behaviors underscore a rising patient advocacy and a demand for personalized medicine, pushing manufacturers to develop tailored treatment plans and focus on patient-centric care models. The increasing prevalence of hemophilia A and hemophilia B, coupled with a growing awareness of early diagnosis and treatment, are significant market growth drivers. Furthermore, the growing number of clinical trials investigating novel therapeutic targets and delivery systems indicates a vibrant R&D landscape. The integration of digital health technologies for remote patient monitoring and adherence tracking is also a key trend shaping the future of hemophilia management. The market is witnessing a paradigm shift from managing acute bleeds to preventing them, with a greater emphasis on Prophylaxis becoming the standard of care in developed economies. The rising disposable income in emerging economies and improved healthcare infrastructure are further contributing to the market's upward trajectory.
Key Markets & Segments Leading Hemophilia Treatment
North America currently dominates the global Hemophilia Treatment market, driven by robust healthcare infrastructure, high prevalence rates, and advanced reimbursement policies that facilitate access to cutting-edge therapies. The United States, in particular, represents a significant market share, propelled by substantial investments in R&D and a strong presence of key pharmaceutical companies.
The Application segments are vital to understanding market dynamics:
Replacement Therapy: This segment continues to be the cornerstone of hemophilia treatment, comprising a significant market share of over $XXX billion. Its dominance is attributed to established efficacy, widespread availability, and a comprehensive understanding of its long-term safety profile. Drivers for its continued strength include:
- High unmet need: Still the primary treatment for many hemophilia patients globally.
- Established infrastructure: Extensive experience in manufacturing and distribution of clotting factors.
- Favorable reimbursement: Generally well-covered by insurance and public health programs.
- Continuous innovation: Development of extended half-life products enhancing convenience.
ITI Therapy (Inhibitor Treatment Inhibitor Therapy): This segment is crucial for managing patients who develop inhibitors to clotting factor concentrates. While a smaller segment compared to replacement therapy, its importance is growing with the increasing sophistication of inhibitor management. Drivers include:
- Rising inhibitor incidence: A significant challenge in hemophilia management.
- Development of novel bypassing agents: Offering effective treatment options for bleeding episodes.
- Growing research into immune tolerance induction: Aiming for long-term resolution of inhibitors.
Gene Therapy: This revolutionary segment is rapidly gaining traction and is projected to witness the highest growth rate, with an estimated market potential exceeding $XXX billion by the end of the forecast period. Its dominance will stem from its potential for a one-time curative treatment. Key drivers include:
- Potential for a cure: Offering a paradigm shift from chronic management to long-term remission.
- Advancements in viral vector technology: Improving safety and efficacy of gene delivery.
- Increasing clinical trial successes: Building confidence in its therapeutic value.
- Growing investment and partnerships: Accelerating development and commercialization.
The Types of hemophilia treatment also segment the market:
Prophylaxis: This approach, involving regular administration of clotting factors to prevent bleeds, is increasingly becoming the standard of care, particularly in developed countries. Its dominance is driven by:
- Improved quality of life: Significantly reducing joint damage and chronic pain.
- Reduced healthcare costs: Preventing severe bleeds and hospitalizations.
- Patient preference: Offering greater predictability and freedom from acute bleeding episodes.
On-demand: This treatment strategy is used to manage acute bleeding episodes. While still important, its relative market share is expected to decrease as prophylaxis becomes more widespread.
Hemophilia Treatment Product Developments
Significant product innovations are reshaping the Hemophilia Treatment landscape. The development of extended half-life clotting factor concentrates has revolutionized Replacement Therapy, offering patients less frequent infusions and improved convenience, thereby enhancing adherence and quality of life. Gene Therapy is at the forefront of innovation, with groundbreaking advancements in viral vector delivery and gene editing technologies showing immense promise for potentially curative treatments for both hemophilia A and B. Bispecific antibodies are also emerging as novel therapeutic agents for hemophilia A, providing a non-factor alternative for patients with inhibitors. These advancements are critical in addressing unmet needs and expanding the therapeutic arsenal against hemophilia.
Challenges in the Hemophilia Treatment Market
Despite promising advancements, the Hemophilia Treatment market faces several challenges. The exorbitant cost of novel therapies, particularly Gene Therapy, presents a significant barrier to access, impacting patient affordability and reimbursement discussions globally. Regulatory hurdles and the lengthy approval processes for new Gene Therapy and ITI Therapy treatments can slow down market penetration. Furthermore, the risk of immune reactions and the long-term safety profiles of some emerging therapies require extensive monitoring. Supply chain complexities for specialized biologics and the need for cold chain logistics can also pose logistical challenges, especially in developing regions. Competitive pressures from multiple players vying for market share in the Replacement Therapy segment also necessitate continuous innovation and cost-effective strategies.
Forces Driving Hemophilia Treatment Growth
Several forces are propelling the Hemophilia Treatment market forward. Technological innovation, particularly in Gene Therapy and novel ITI Therapy development, is a primary driver, offering the potential for transformative treatments and cures. Increasing global prevalence of hemophilia, coupled with rising awareness and improved diagnostic capabilities, expands the patient pool. Favorable reimbursement policies in developed nations and expanding healthcare access in emerging economies are crucial economic drivers. Regulatory bodies are also increasingly streamlining approval pathways for innovative hemophilia treatments, further accelerating market growth.
Challenges in the Hemophilia Treatment Market
Long-term growth catalysts for the Hemophilia Treatment market are multifaceted. The continuous pursuit of functional cures through Gene Therapy remains a pivotal long-term catalyst, offering the potential to fundamentally alter the treatment paradigm. Strategic partnerships between pharmaceutical giants and biotech startups are accelerating the development and commercialization of groundbreaking therapies. Market expansion into underserved regions with increasing healthcare expenditure and a growing understanding of hemophilia is another critical growth accelerator. The development of personalized medicine approaches tailored to individual patient genetics and inhibitor profiles will also drive future growth.
Emerging Opportunities in Hemophilia Treatment
Emerging opportunities in the Hemophilia Treatment sector are abundant. The untapped potential of emerging markets in Asia-Pacific and Latin America, where hemophilia diagnosis and treatment are still developing, presents significant growth avenues. The advancement of non-viral gene delivery systems could mitigate safety concerns associated with current viral vectors, opening new avenues for Gene Therapy. The development of oral Replacement Therapy options, if proven safe and effective, would revolutionize patient convenience. Furthermore, focusing on patient-centric digital health solutions for remote monitoring and personalized care management offers a significant opportunity to enhance treatment adherence and outcomes.
Leading Players in the Hemophilia Treatment Sector
- CSL Behring
- Baxalta
- Pfizer Inc
- BioMarin
- Bayer Healthcare
- Biogen
- Novo Nordisk
- Roche
- Takeda
Key Milestones in Hemophilia Treatment Industry
- 2019: Approval of multiple extended half-life factor VIII and IX concentrates in major markets, improving treatment convenience.
- 2020: Significant progress in clinical trials for hemophilia B gene therapy, demonstrating promising results.
- 2021: Launch of new bispecific antibody therapies for hemophilia A with inhibitors, offering an alternative to factor replacement.
- 2022: Increased investment and strategic partnerships in the gene therapy space, signaling growing confidence in its potential.
- 2023: Further advancements in the development of gene therapies for hemophilia A, with promising clinical trial data.
- 2024: Anticipated regulatory submissions for novel gene therapy candidates for both hemophilia A and B.
Strategic Outlook for Hemophilia Treatment Market
The strategic outlook for the Hemophilia Treatment market is overwhelmingly positive, driven by a pipeline rich with innovation. The ongoing revolution in Gene Therapy promises to fundamentally alter the treatment landscape, moving towards curative solutions. Continued advancements in Replacement Therapy with extended half-life products will solidify its position, while novel ITI Therapy options will address critical unmet needs. Strategic collaborations, increased R&D investment, and expansion into emerging markets will be key growth accelerators. The focus will increasingly shift towards precision medicine, personalized treatment plans, and improved patient outcomes, creating substantial future market potential.
Hemophilia Treatment Segmentation
-
1. Application
- 1.1. Replacement Therapy
- 1.2. ITI Therapy
- 1.3. Gene Therapy
-
2. Types
- 2.1. On-demand
- 2.2. Prophylaxis
Hemophilia Treatment Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
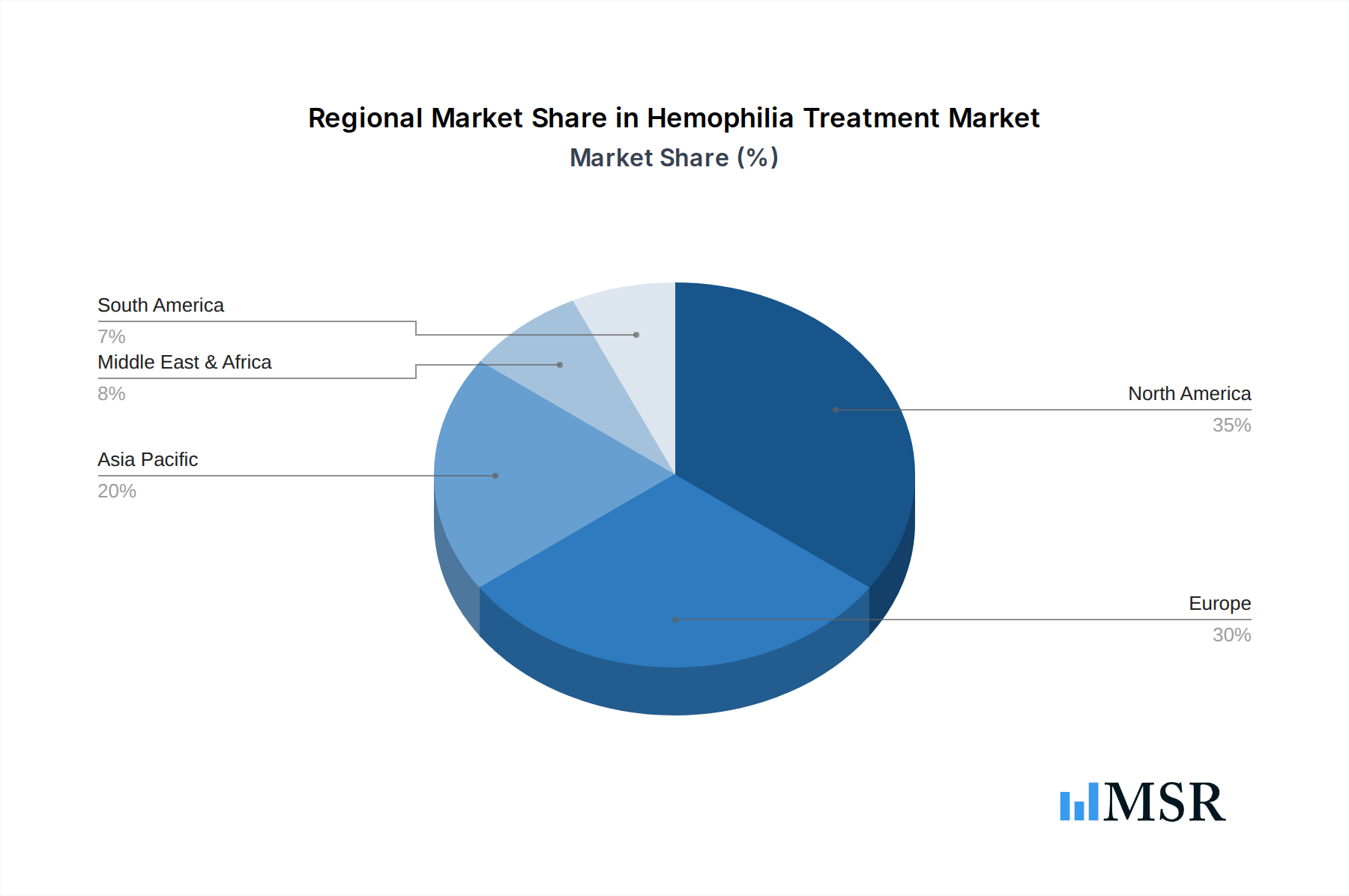
Hemophilia Treatment Regional Market Share

Geographic Coverage of Hemophilia Treatment
Hemophilia Treatment REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Hemophilia Treatment Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Replacement Therapy
- 5.1.2. ITI Therapy
- 5.1.3. Gene Therapy
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. On-demand
- 5.2.2. Prophylaxis
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Hemophilia Treatment Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Replacement Therapy
- 6.1.2. ITI Therapy
- 6.1.3. Gene Therapy
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. On-demand
- 6.2.2. Prophylaxis
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Hemophilia Treatment Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Replacement Therapy
- 7.1.2. ITI Therapy
- 7.1.3. Gene Therapy
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. On-demand
- 7.2.2. Prophylaxis
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Hemophilia Treatment Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Replacement Therapy
- 8.1.2. ITI Therapy
- 8.1.3. Gene Therapy
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. On-demand
- 8.2.2. Prophylaxis
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Hemophilia Treatment Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Replacement Therapy
- 9.1.2. ITI Therapy
- 9.1.3. Gene Therapy
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. On-demand
- 9.2.2. Prophylaxis
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Hemophilia Treatment Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Replacement Therapy
- 10.1.2. ITI Therapy
- 10.1.3. Gene Therapy
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. On-demand
- 10.2.2. Prophylaxis
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 CSL Behring
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Baxalta
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Pfizer Inc
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 BioMarin
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Bayer Healthcare
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Biogen
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Novo Nordisk
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Roche
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Takeda
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.1 CSL Behring
List of Figures
- Figure 1: Global Hemophilia Treatment Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Hemophilia Treatment Revenue (billion), by Application 2025 & 2033
- Figure 3: North America Hemophilia Treatment Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Hemophilia Treatment Revenue (billion), by Types 2025 & 2033
- Figure 5: North America Hemophilia Treatment Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Hemophilia Treatment Revenue (billion), by Country 2025 & 2033
- Figure 7: North America Hemophilia Treatment Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Hemophilia Treatment Revenue (billion), by Application 2025 & 2033
- Figure 9: South America Hemophilia Treatment Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Hemophilia Treatment Revenue (billion), by Types 2025 & 2033
- Figure 11: South America Hemophilia Treatment Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Hemophilia Treatment Revenue (billion), by Country 2025 & 2033
- Figure 13: South America Hemophilia Treatment Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Hemophilia Treatment Revenue (billion), by Application 2025 & 2033
- Figure 15: Europe Hemophilia Treatment Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Hemophilia Treatment Revenue (billion), by Types 2025 & 2033
- Figure 17: Europe Hemophilia Treatment Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Hemophilia Treatment Revenue (billion), by Country 2025 & 2033
- Figure 19: Europe Hemophilia Treatment Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Hemophilia Treatment Revenue (billion), by Application 2025 & 2033
- Figure 21: Middle East & Africa Hemophilia Treatment Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Hemophilia Treatment Revenue (billion), by Types 2025 & 2033
- Figure 23: Middle East & Africa Hemophilia Treatment Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Hemophilia Treatment Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East & Africa Hemophilia Treatment Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Hemophilia Treatment Revenue (billion), by Application 2025 & 2033
- Figure 27: Asia Pacific Hemophilia Treatment Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Hemophilia Treatment Revenue (billion), by Types 2025 & 2033
- Figure 29: Asia Pacific Hemophilia Treatment Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Hemophilia Treatment Revenue (billion), by Country 2025 & 2033
- Figure 31: Asia Pacific Hemophilia Treatment Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Hemophilia Treatment Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global Hemophilia Treatment Revenue billion Forecast, by Types 2020 & 2033
- Table 3: Global Hemophilia Treatment Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global Hemophilia Treatment Revenue billion Forecast, by Application 2020 & 2033
- Table 5: Global Hemophilia Treatment Revenue billion Forecast, by Types 2020 & 2033
- Table 6: Global Hemophilia Treatment Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global Hemophilia Treatment Revenue billion Forecast, by Application 2020 & 2033
- Table 11: Global Hemophilia Treatment Revenue billion Forecast, by Types 2020 & 2033
- Table 12: Global Hemophilia Treatment Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Brazil Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: Argentina Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Global Hemophilia Treatment Revenue billion Forecast, by Application 2020 & 2033
- Table 17: Global Hemophilia Treatment Revenue billion Forecast, by Types 2020 & 2033
- Table 18: Global Hemophilia Treatment Revenue billion Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 20: Germany Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 21: France Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 22: Italy Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Spain Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: Russia Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Benelux Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Nordics Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global Hemophilia Treatment Revenue billion Forecast, by Application 2020 & 2033
- Table 29: Global Hemophilia Treatment Revenue billion Forecast, by Types 2020 & 2033
- Table 30: Global Hemophilia Treatment Revenue billion Forecast, by Country 2020 & 2033
- Table 31: Turkey Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Israel Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: GCC Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: North Africa Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 35: South Africa Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 37: Global Hemophilia Treatment Revenue billion Forecast, by Application 2020 & 2033
- Table 38: Global Hemophilia Treatment Revenue billion Forecast, by Types 2020 & 2033
- Table 39: Global Hemophilia Treatment Revenue billion Forecast, by Country 2020 & 2033
- Table 40: China Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 41: India Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: Japan Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 43: South Korea Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 45: Oceania Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Hemophilia Treatment Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Hemophilia Treatment?
The projected CAGR is approximately 6.4%.
2. Which companies are prominent players in the Hemophilia Treatment?
Key companies in the market include CSL Behring, Baxalta, Pfizer Inc, BioMarin, Bayer Healthcare, Biogen, Novo Nordisk, Roche, Takeda.
3. What are the main segments of the Hemophilia Treatment?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 4.48 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4900.00, USD 7350.00, and USD 9800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Hemophilia Treatment," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Hemophilia Treatment report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Hemophilia Treatment?
To stay informed about further developments, trends, and reports in the Hemophilia Treatment, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
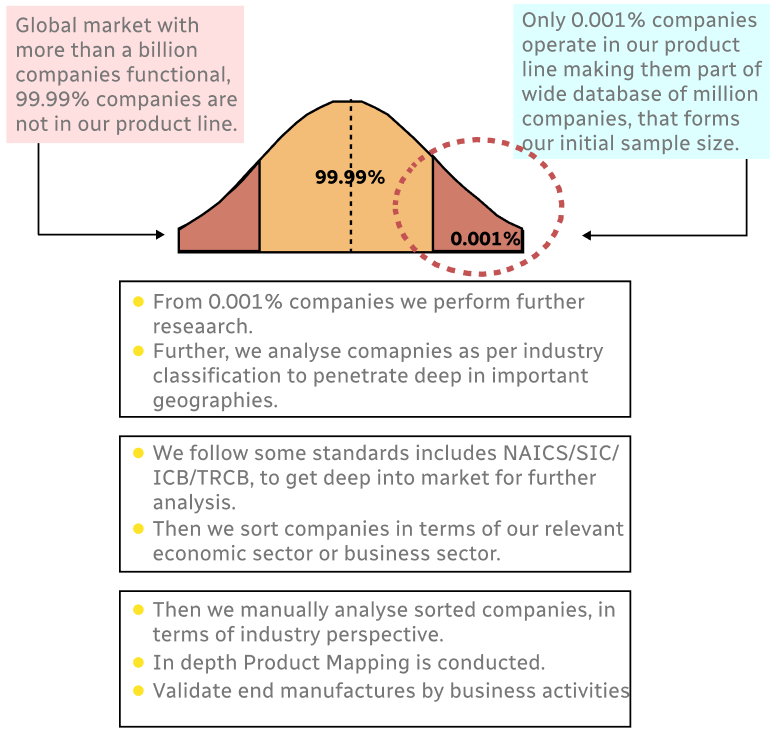
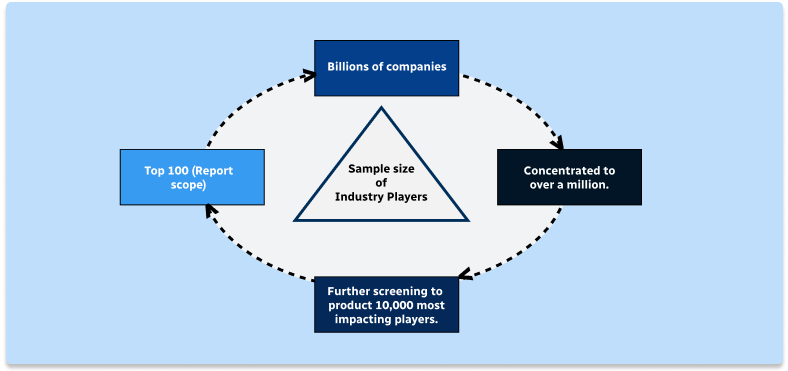
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
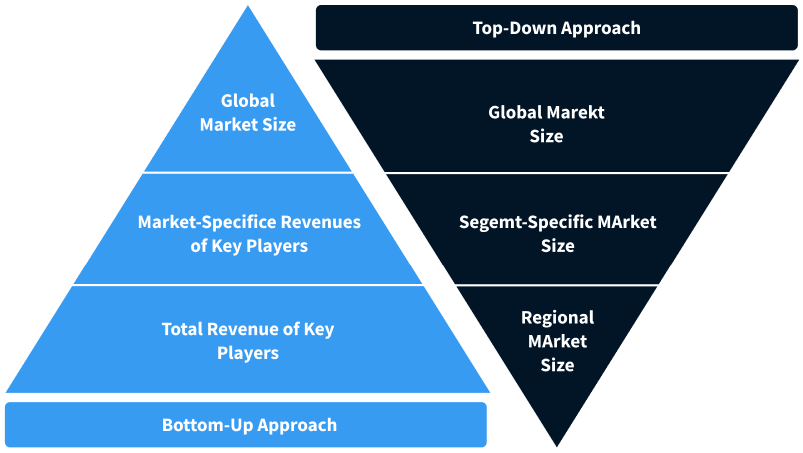
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
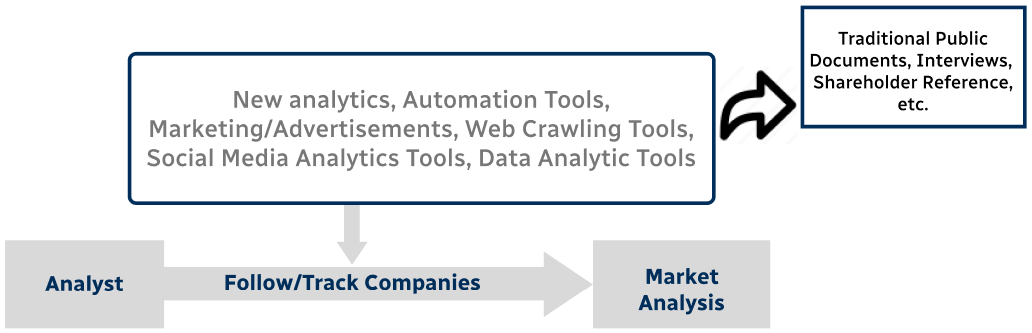
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


