Key Insights
The Japan Respiratory Devices Industry is poised for significant expansion, projected to reach $23.6 billion by 2025. This robust growth is driven by a confluence of factors, including the increasing prevalence of chronic respiratory diseases such as asthma, COPD, and sleep apnea, largely attributed to an aging population and rising air pollution levels. Furthermore, advancements in medical technology are continuously introducing more sophisticated and user-friendly diagnostic and therapeutic devices, enhancing patient care and treatment outcomes. The market is segmented into Diagnostic and Monitoring Devices, Therapeutic Devices, and Disposables, each playing a crucial role in the respiratory care continuum. Diagnostic tools like spirometers and sleep test devices are vital for early detection and accurate assessment, while therapeutic solutions like ventilators and CPAP machines are indispensable for managing severe respiratory conditions. The disposable segment, encompassing items like masks and breathing circuits, also exhibits steady growth due to increasing healthcare expenditure and a focus on infection control. Key players in this dynamic market include CHEST M I Inc, Terumo Corporation, GE Healthcare, Medtronic PLC, Koninklijke Philips NV, and ResMed Inc, who are instrumental in driving innovation and market penetration.
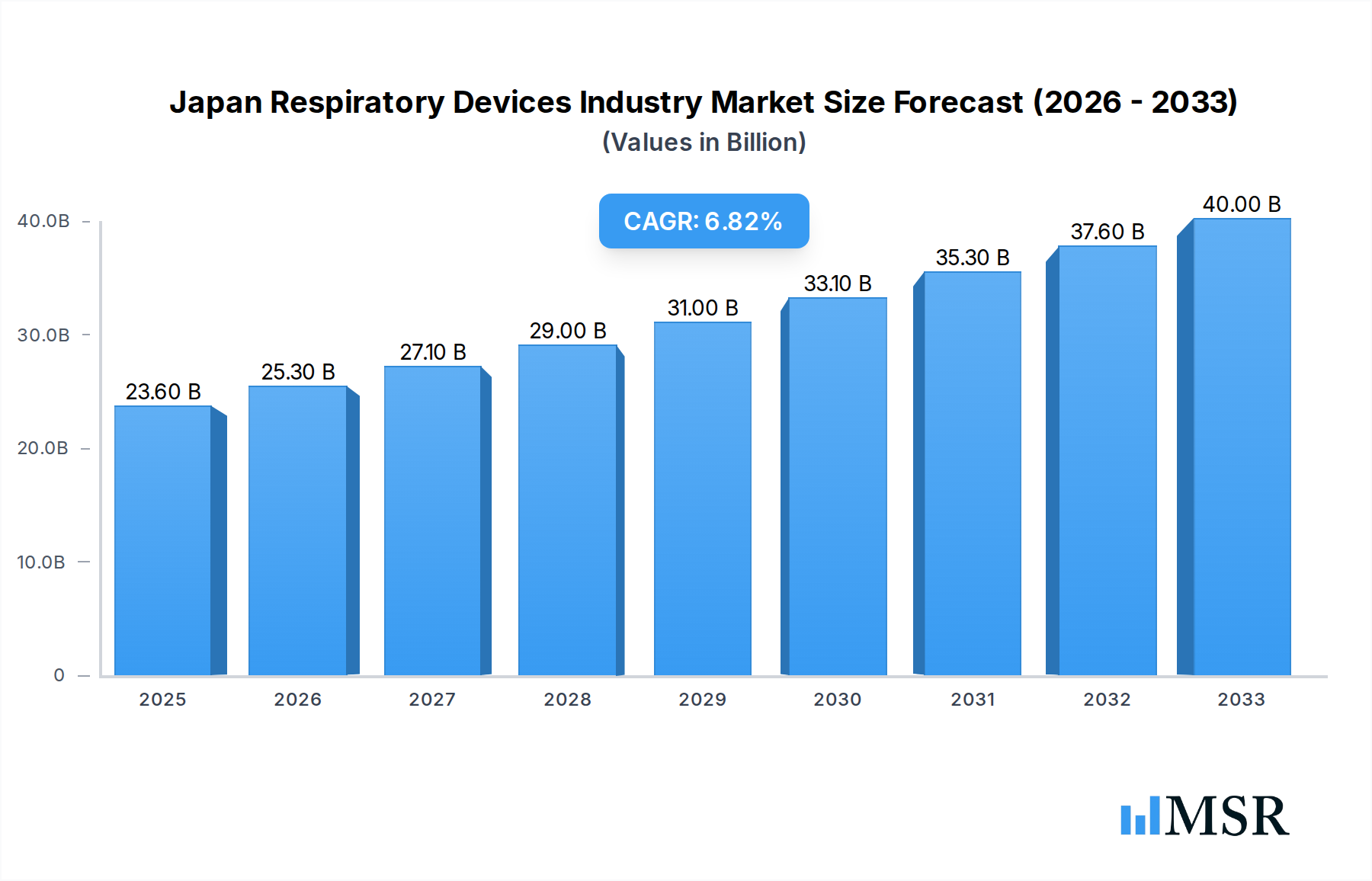
Japan Respiratory Devices Industry Market Size (In Billion)

The industry is expected to experience a Compound Annual Growth Rate (CAGR) of 7.3% from 2025 to 2033, indicating sustained momentum in market value. This growth trajectory is underpinned by several key trends, including the rising adoption of home-based respiratory care solutions, the development of smart and connected respiratory devices for remote patient monitoring, and an increased focus on personalized treatment approaches. The aging demographic in Japan, characterized by a higher susceptibility to respiratory ailments, acts as a primary driver for increased demand. Technological innovations, such as miniaturization of devices and development of AI-powered diagnostic tools, further fuel market expansion. Despite the optimistic outlook, certain restraints, such as stringent regulatory approvals for new medical devices and the high cost of advanced respiratory equipment, could present challenges. However, the overall market sentiment remains positive, with continuous efforts by manufacturers and healthcare providers to improve accessibility and affordability of respiratory care solutions, ensuring the industry's upward trajectory.
Japan Respiratory Devices Industry Company Market Share

Unlocking the Potential: Japan Respiratory Devices Industry Market Analysis 2019-2033
This comprehensive report offers an in-depth analysis of the burgeoning Japan respiratory devices market, a critical sector driven by an aging population, increasing prevalence of respiratory diseases, and advancements in medical technology. Covering the study period from 2019 to 2033, with a base year and estimated year of 2025, and a forecast period of 2025-2033, this report provides actionable insights for respiratory device manufacturers, healthcare providers, investors, and policy makers. Explore the market's dynamics, key growth drivers, leading segments, and future opportunities in this essential industry.
Japan Respiratory Devices Industry Market Concentration & Dynamics
The Japan respiratory devices market exhibits a moderate to high level of concentration, with a few dominant players holding significant market share. Key companies such as CHEST M I Inc, Terumo Corporation, GE Healthcare, Medtronic PLC, Getinge AB, Metran Co Ltd, Koninklijke Philips NV, ResMed Inc, Dragerwerk AG, and Fisher & Paykel Healthcare Ltd are at the forefront of innovation and market penetration. The innovation ecosystem is robust, fueled by continuous R&D investments in areas like digital respiratory health, AI-powered diagnostics, and minimally invasive therapeutic devices. Regulatory frameworks, primarily overseen by the Ministry of Health, Labour and Welfare (MHLW), ensure product safety and efficacy, though navigating these can present challenges. Substitute products, such as drug therapies, exist but are increasingly complemented by advanced respiratory monitoring devices and therapeutic ventilators. End-user trends are heavily influenced by an aging demographic, leading to a surge in demand for homecare respiratory solutions and long-term ventilation support. Mergers and Acquisitions (M&A) activities are moderate, often focused on acquiring innovative technologies or expanding market reach within specific respiratory device segments. The Japan respiratory devices market size is projected to reach several billion by 2025 and continue its upward trajectory. M&A deal counts have seen a steady increase, indicating strategic consolidation within the sector.
Japan Respiratory Devices Industry Industry Insights & Trends
The Japan respiratory devices industry is poised for substantial growth, driven by a confluence of demographic, technological, and healthcare-related factors. The market size is estimated to reach XX billion in 2025, with a projected Compound Annual Growth Rate (CAGR) of XX% during the forecast period of 2025-2033. A primary growth driver is the rapidly aging population in Japan, leading to an increased incidence of age-related respiratory conditions such as Chronic Obstructive Pulmonary Disease (COPD), asthma, and sleep apnea. This demographic shift directly translates to a higher demand for diagnostic and monitoring devices like spirometers and sleep test devices, as well as therapeutic devices such as ventilators and CPAP devices.
Technological disruptions are significantly reshaping the landscape. The integration of artificial intelligence (AI) and machine learning (ML) into respiratory monitoring solutions is enhancing diagnostic accuracy and enabling personalized treatment plans. For instance, the development of smart inhalers and connected oxygen concentrators allows for real-time data collection and remote patient monitoring, improving adherence and outcomes. The increasing focus on telehealth and remote patient management further amplifies the need for advanced respiratory care devices that can facilitate seamless communication between patients and healthcare providers.
Evolving consumer behaviors also play a crucial role. Patients are increasingly seeking convenient and accessible healthcare solutions, driving the demand for homecare respiratory devices. This trend is further supported by government initiatives to promote home-based care and reduce the burden on hospitals. The rise of preventative healthcare awareness is also boosting the adoption of diagnostic respiratory equipment for early detection and management of respiratory ailments. Furthermore, the COVID-19 pandemic underscored the importance of robust ventilation systems and oxygen therapy devices, leading to increased investment and innovation in these areas. The Japan respiratory devices market forecast indicates continued expansion driven by these powerful trends. The market is expected to reach XX billion by 2033.
Key Markets & Segments Leading Japan Respiratory Devices Industry
The Japan respiratory devices industry is characterized by a dynamic interplay of various segments, with Therapeutic Devices emerging as a dominant category, closely followed by Diagnostic and Monitoring Devices. Within Therapeutic Devices, Ventilators are a key growth driver, propelled by the increasing prevalence of respiratory failure, particularly among the elderly, and the ongoing need for critical care solutions in hospitals. The demand for advanced CPAP Devices is also substantial, fueled by the high rates of sleep apnea in Japan and growing awareness of its health implications. Oxygen Concentrators are experiencing steady growth due to their crucial role in managing chronic respiratory diseases and facilitating home-based oxygen therapy.
Diagnostic and Monitoring Devices are also vital, with Sleep Test Devices showing significant expansion due to increased diagnosis of sleep disorders. Spirometers remain essential for diagnosing and monitoring conditions like asthma and COPD. The "Other Diagnostic and Monitoring Devices" sub-segment, encompassing pulse oximeters and respiratory monitors, is also contributing to overall growth as technology advances offer more portable and accurate monitoring capabilities.
The Disposables segment, including Masks and Breathing Circuits, is directly correlated with the adoption of therapeutic devices. As the use of ventilators, CPAP machines, and nebulizers increases, so does the demand for associated consumables, ensuring a steady revenue stream for manufacturers.
Dominance Analysis:
- Therapeutic Devices: The increasing burden of chronic respiratory diseases, coupled with the aging population, makes therapeutic interventions paramount. The market for ventilators is significantly influenced by the need for life support in intensive care units and the growing trend of non-invasive ventilation for home use. CPAP devices are benefiting from increased diagnosis rates and wider insurance coverage for sleep apnea treatment.
- Diagnostic and Monitoring Devices: The proactive approach to healthcare in Japan, emphasizing early detection and disease management, drives the demand for sophisticated diagnostic tools. The rising awareness of sleep disorders and the development of user-friendly home sleep apnea testing devices are key factors.
- Disposables: The consistent need for replacement parts for therapeutic devices ensures this segment's stability and growth, directly mirroring the expansion of the therapeutic device market.
Economic growth and robust healthcare infrastructure in Japan provide a fertile ground for all segments. Government initiatives promoting early diagnosis and home-based care also act as significant drivers. The continuous integration of digital technologies within these segments is further enhancing their market position and adoption rates. The Japan respiratory devices market share across these segments is constantly evolving with technological advancements and changing healthcare needs.
Japan Respiratory Devices Industry Product Developments
Product innovation is a cornerstone of the Japan respiratory devices industry. Recent advancements include the introduction of smart inhalers like Aptar Pharma's HeroTracker Sense, transforming standard MDIs into connected healthcare devices for enhanced adherence and data tracking. PARI Pharma GmbH's authorization of the LAMIRA Nebulizer System for delivering specific drug products in Japan highlights the growing synergy between drug delivery systems and advanced nebulizer technology. These developments underscore a trend towards more personalized, data-driven, and patient-centric respiratory care solutions, offering improved efficacy, convenience, and remote monitoring capabilities. The focus is on miniaturization, enhanced usability, and integration with digital health platforms.
Challenges in the Japan Respiratory Devices Industry Market
Despite its strong growth trajectory, the Japan respiratory devices market faces several challenges. Regulatory hurdles can sometimes lead to extended approval timelines for new and innovative devices, impacting time-to-market. Supply chain disruptions, as witnessed globally, can affect the availability of critical components and finished products, leading to increased costs and potential shortages. Intense competitive pressures from both domestic and international players necessitate continuous innovation and cost-effective solutions. Furthermore, the high cost of advanced respiratory medical devices can be a barrier to adoption for some segments of the population and healthcare facilities, impacting market penetration for premium products.
Forces Driving Japan Respiratory Devices Industry Growth
Several key forces are propelling the Japan respiratory devices industry forward. The aging demographic is a primary driver, increasing the incidence of respiratory conditions. Technological advancements, including AI integration, miniaturization, and the development of connected devices, are enhancing product efficacy and user experience. Government support for home healthcare and initiatives promoting early disease detection further stimulate demand. Growing awareness among patients and healthcare professionals about the importance of respiratory health management and the availability of sophisticated respiratory support systems also contribute significantly.
Challenges in the Japan Respiratory Devices Industry Market
Long-term growth catalysts for the Japan respiratory devices industry lie in sustained innovation and strategic market expansion. The continued development of AI-powered diagnostic tools and predictive analytics for respiratory diseases will be crucial. Partnerships between device manufacturers and pharmaceutical companies for integrated drug delivery and monitoring solutions will unlock new value propositions. Expanding the reach of tele-respiratory care and remote patient monitoring beyond urban centers will tap into underserved markets. Moreover, focusing on cost-effective yet high-quality respiratory solutions to improve accessibility for a broader population remains a key long-term growth imperative.
Emerging Opportunities in Japan Respiratory Devices Industry
Emerging opportunities in the Japan respiratory devices industry are plentiful. The burgeoning field of digital respiratory health presents immense potential for connected devices that offer real-time data analytics and personalized interventions. There is a growing demand for wearable respiratory sensors and non-invasive diagnostic tools for continuous monitoring. Furthermore, the increasing adoption of telemedicine platforms creates a significant opportunity for remote patient management solutions. The market for pediatric respiratory devices is also gaining traction, with a need for specialized and user-friendly equipment. Exploring novel drug delivery systems and advanced aerosol therapy technologies also represents a promising avenue.
Leading Players in the Japan Respiratory Devices Industry Sector
- CHEST M I Inc
- Terumo Corporation
- GE Healthcare
- Medtronic PLC
- Getinge AB
- Metran Co Ltd
- Koninklijke Philips NV
- ResMed Inc
- Dragerwerk AG
- Fisher & Paykel Healthcare Ltd
Key Milestones in Japan Respiratory Devices Industry Industry
- Feb 2022: Aptar Pharma announced the launch of HeroTracker Sense, a novel digital respiratory health solution that transforms a standard metered dose inhaler (pMDI) into a smart, connected healthcare device. This development marks a significant step towards connected inhaler technology and improved patient adherence.
- Mar 2021: PARI Pharma GmbH, a company focused on advanced aerosol delivery systems based on eFlow Technology, announced the authorization of the LAMIRA Nebulizer System for delivery of Insmed's drug product ARIKAYCE (amikacin liposome inhalation suspension) in Japan. This milestone highlights the growing importance of specialized nebulizer systems for targeted drug delivery in the Japanese market.
Strategic Outlook for Japan Respiratory Devices Industry Market
The Japan respiratory devices industry market is set for robust future growth, fueled by ongoing innovation and increasing demand. Key growth accelerators include the continued development and adoption of digital respiratory health technologies, enabling remote patient monitoring and personalized treatment plans. Strategic partnerships between device manufacturers, pharmaceutical companies, and healthcare providers will foster integrated care solutions. The expanding homecare market presents a significant opportunity for user-friendly and cost-effective respiratory support devices. Furthermore, advancements in AI and data analytics will drive the development of more predictive and proactive respiratory care, solidifying Japan's position as a leader in this critical healthcare sector. The market is expected to reach XX billion by 2033.
Japan Respiratory Devices Industry Segmentation
-
1. Type
-
1.1. Diagnostic and Monitoring Devices
- 1.1.1. Spirometers
- 1.1.2. Sleep Test Devices
- 1.1.3. Peak Flow Meters
- 1.1.4. Other Diagnostic and Monitoring Devices
-
1.2. Therapeutic Devices
- 1.2.1. Ventilators
- 1.2.2. Inhalers
- 1.2.3. CPAP Devices
- 1.2.4. Oxygen Concentrators
- 1.2.5. Other Therapeutic Devices
-
1.3. Disposables
- 1.3.1. Masks
- 1.3.2. Breathing Circuits
- 1.3.3. Other Disposables
-
1.1. Diagnostic and Monitoring Devices
Japan Respiratory Devices Industry Segmentation By Geography
- 1. Japan
Japan Respiratory Devices Industry Regional Market Share

Geographic Coverage of Japan Respiratory Devices Industry
Japan Respiratory Devices Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.3% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. MSR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Type
- 5.1.1. Diagnostic and Monitoring Devices
- 5.1.1.1. Spirometers
- 5.1.1.2. Sleep Test Devices
- 5.1.1.3. Peak Flow Meters
- 5.1.1.4. Other Diagnostic and Monitoring Devices
- 5.1.2. Therapeutic Devices
- 5.1.2.1. Ventilators
- 5.1.2.2. Inhalers
- 5.1.2.3. CPAP Devices
- 5.1.2.4. Oxygen Concentrators
- 5.1.2.5. Other Therapeutic Devices
- 5.1.3. Disposables
- 5.1.3.1. Masks
- 5.1.3.2. Breathing Circuits
- 5.1.3.3. Other Disposables
- 5.1.1. Diagnostic and Monitoring Devices
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. Japan
- 5.1. Market Analysis, Insights and Forecast - by Type
- 6. Japan Respiratory Devices Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Type
- 6.1.1. Diagnostic and Monitoring Devices
- 6.1.1.1. Spirometers
- 6.1.1.2. Sleep Test Devices
- 6.1.1.3. Peak Flow Meters
- 6.1.1.4. Other Diagnostic and Monitoring Devices
- 6.1.2. Therapeutic Devices
- 6.1.2.1. Ventilators
- 6.1.2.2. Inhalers
- 6.1.2.3. CPAP Devices
- 6.1.2.4. Oxygen Concentrators
- 6.1.2.5. Other Therapeutic Devices
- 6.1.3. Disposables
- 6.1.3.1. Masks
- 6.1.3.2. Breathing Circuits
- 6.1.3.3. Other Disposables
- 6.1.1. Diagnostic and Monitoring Devices
- 6.1. Market Analysis, Insights and Forecast - by Type
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 CHEST M I Inc
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 Terumo Corporation
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 GE Healthcare
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 Medtronic PLC
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Getinge AB
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Metran Co Ltd*List Not Exhaustive
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 Koninklijke Philips NV
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 ResMed Inc
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.9 Dragerwerk AG
- 7.1.9.1. Company Overview
- 7.1.9.2. Products
- 7.1.9.3. Company Financials
- 7.1.9.4. SWOT Analysis
- 7.1.10 Fisher & Paykel Healthcare Ltd
- 7.1.10.1. Company Overview
- 7.1.10.2. Products
- 7.1.10.3. Company Financials
- 7.1.10.4. SWOT Analysis
- 7.1.1 CHEST M I Inc
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: Japan Respiratory Devices Industry Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: Japan Respiratory Devices Industry Share (%) by Company 2025
List of Tables
- Table 1: Japan Respiratory Devices Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 2: Japan Respiratory Devices Industry Revenue billion Forecast, by Region 2020 & 2033
- Table 3: Japan Respiratory Devices Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 4: Japan Respiratory Devices Industry Revenue billion Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Japan Respiratory Devices Industry?
The projected CAGR is approximately 7.3%.
2. Which companies are prominent players in the Japan Respiratory Devices Industry?
Key companies in the market include CHEST M I Inc, Terumo Corporation, GE Healthcare, Medtronic PLC, Getinge AB, Metran Co Ltd*List Not Exhaustive, Koninklijke Philips NV, ResMed Inc, Dragerwerk AG, Fisher & Paykel Healthcare Ltd.
3. What are the main segments of the Japan Respiratory Devices Industry?
The market segments include Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 23.6 billion as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence of Respiratory Disorders; Technological Advancements in the Devices.
6. What are the notable trends driving market growth?
Inhalers Segment is Expected to Witness Strong Growth Over the Forecast Period.
7. Are there any restraints impacting market growth?
High Cost of Equipment.
8. Can you provide examples of recent developments in the market?
Feb 2022: Aptar Pharma announced the launch of HeroTracker Sense, a novel digital respiratory health solution that transforms a standard metered dose inhaler (pMDI) into a smart, connected healthcare device.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Japan Respiratory Devices Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Japan Respiratory Devices Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Japan Respiratory Devices Industry?
To stay informed about further developments, trends, and reports in the Japan Respiratory Devices Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
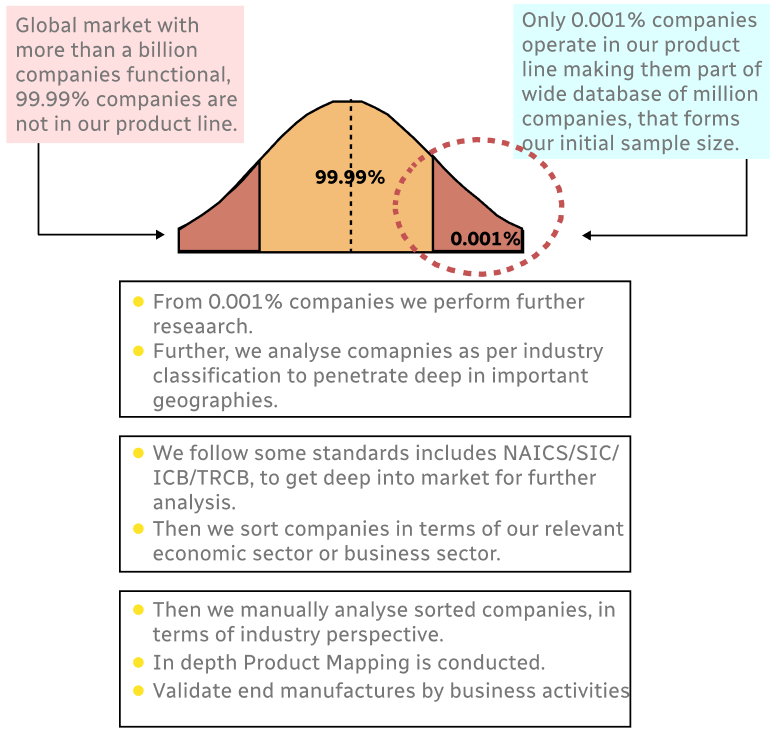
Step 1 - Identification of Relevant Samples Size from Population Database
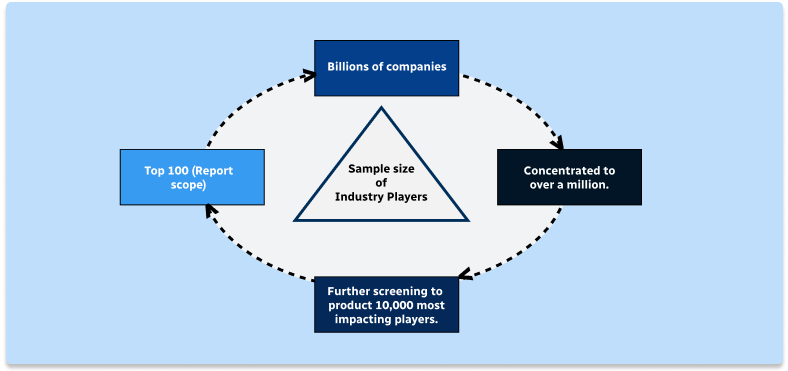
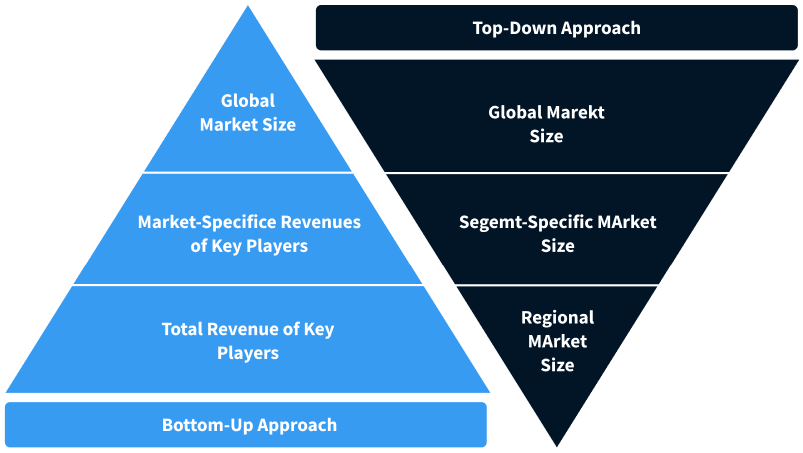
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


