Key Insights
The North America Diabetic Eye Disease Devices market is poised for significant expansion, projected to reach an estimated $12.65 billion in 2025. This robust growth is driven by the escalating prevalence of diabetes and its associated ocular complications across the United States, Canada, and Mexico. A substantial Compound Annual Growth Rate (CAGR) of 9.96% is anticipated over the forecast period (2025-2033), underscoring the increasing demand for advanced diagnostic and therapeutic solutions. Key drivers include technological advancements in ophthalmic lasers and intraocular lenses, alongside a growing awareness of early detection and intervention for conditions like diabetic retinopathy, glaucoma, and age-related macular degeneration. The market is segmented into sophisticated devices and targeted drug therapies, catering to a diverse range of patient needs.
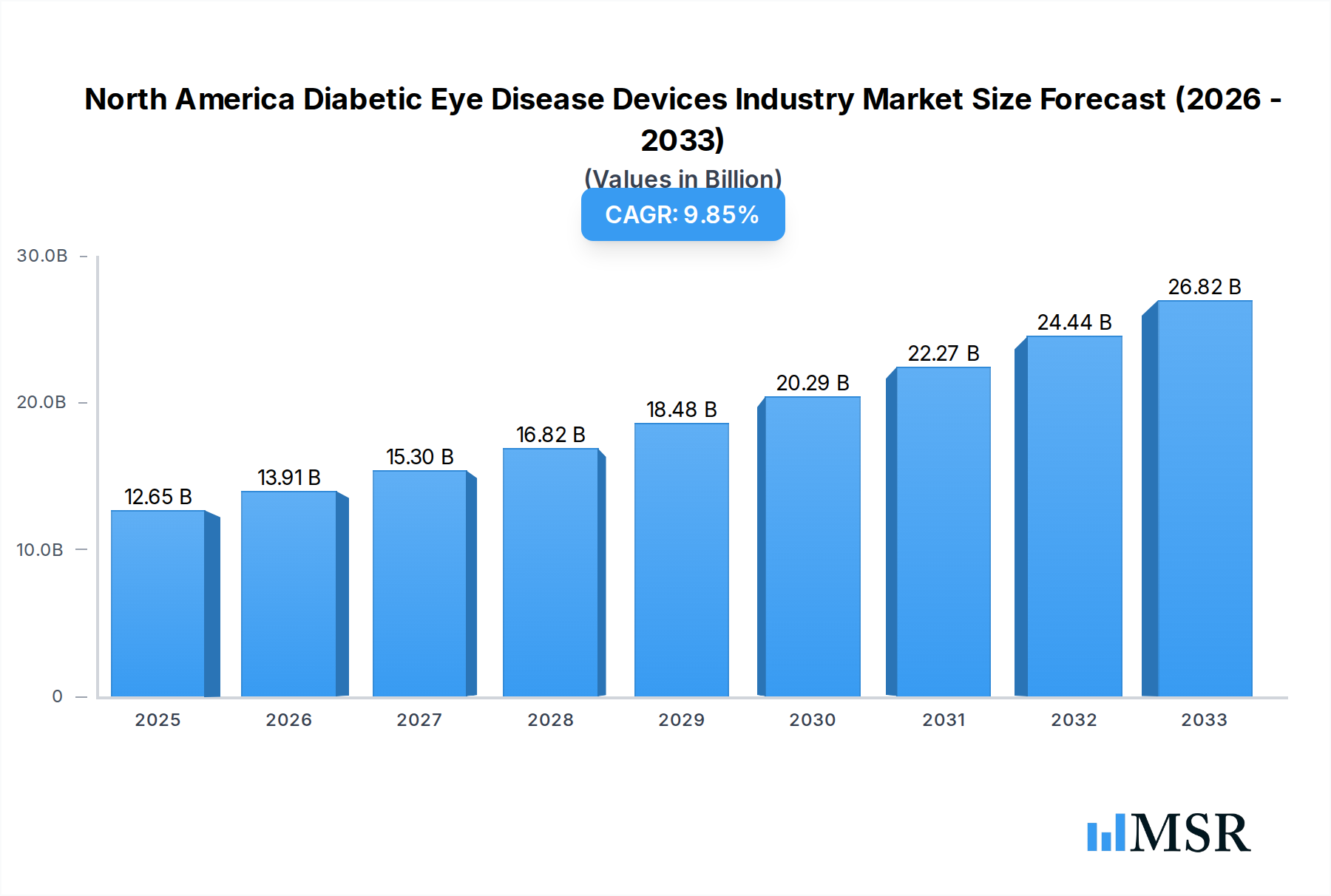
North America Diabetic Eye Disease Devices Industry Market Size (In Billion)

The competitive landscape features prominent players such as Alcon Inc., Johnson & Johnson, and Carl Zeiss Meditec AG, all vying for market share through innovation and strategic collaborations. Emerging trends such as the integration of artificial intelligence in diagnostic imaging and the development of minimally invasive surgical techniques are expected to further propel market expansion. While the market exhibits strong growth potential, challenges such as high treatment costs and limited access to specialized eye care in certain regions may present minor restraints. However, the overall outlook remains highly positive, driven by the continuous need for effective management of diabetic eye diseases and the relentless pursuit of improved patient outcomes through cutting-edge medical technology.
North America Diabetic Eye Disease Devices Industry Company Market Share

This comprehensive report provides an in-depth analysis of the North America Diabetic Eye Disease Devices Industry, offering actionable insights for stakeholders. The study encompasses a detailed examination of market dynamics, key segments, product developments, challenges, growth drivers, emerging opportunities, leading players, and strategic outlook, covering the period from 2019 to 2033, with a base and estimated year of 2025 and a forecast period of 2025–2033.
North America Diabetic Eye Disease Devices Industry Market Concentration & Dynamics
The North America Diabetic Eye Disease Devices Industry exhibits a moderate to high market concentration, characterized by a blend of established multinational corporations and emerging specialized companies. Key players such as Johnson & Johnson, Alcon Inc., and Carl Zeiss Meditec AG hold significant market shares, driven by their extensive product portfolios, robust R&D investments, and established distribution networks. Innovation is a critical determinant of competitive advantage, with ongoing advancements in diagnostic imaging technologies, minimally invasive surgical devices, and novel drug delivery systems. The regulatory landscape, primarily governed by the FDA in the United States and Health Canada, plays a crucial role in market entry and product approval, fostering a stringent environment for safety and efficacy. Substitute products, while present, often fall short in offering the specificity and efficacy required for managing complex diabetic eye conditions. End-user trends indicate a growing demand for early detection, personalized treatment strategies, and less invasive procedures. Merger and acquisition activities are observed as companies seek to expand their technological capabilities, market reach, and product offerings. For instance, strategic partnerships and acquisitions are common strategies to consolidate market position.
North America Diabetic Eye Disease Devices Industry Industry Insights & Trends
The North America Diabetic Eye Disease Devices Industry is poised for significant expansion, driven by a confluence of factors including the escalating prevalence of diabetes and its ocular complications. The market size is estimated to reach $XX billion by 2025, with a projected Compound Annual Growth Rate (CAGR) of XX% during the forecast period of 2025–2033. Technological disruptions are at the forefront of this growth, with innovations in artificial intelligence-powered diagnostic tools enabling earlier and more accurate detection of diabetic retinopathy, diabetic macular edema, and glaucoma. Advancements in ophthalmic lasers, such as femtosecond lasers for cataract surgery, and the development of sophisticated intraocular lenses (IOLs) offering enhanced visual outcomes are transforming surgical interventions. Furthermore, the evolution of drug delivery systems for retinal disorders and dry eye conditions is improving patient compliance and treatment efficacy. Evolving consumer behaviors are also shaping the market, with patients increasingly seeking minimally invasive procedures, faster recovery times, and personalized treatment plans. The growing awareness among diabetic patients about the importance of regular eye screenings and proactive management of eye health is a key market driver. The increasing healthcare expenditure and supportive government initiatives aimed at managing chronic diseases further bolster market growth. The integration of telemedicine and remote monitoring solutions is also gaining traction, offering greater accessibility to eye care services, particularly in underserved regions.
Key Markets & Segments Leading North America Diabetic Eye Disease Devices Industry
The United States stands as the dominant market within the North America Diabetic Eye Disease Devices Industry, driven by its large and aging population, high prevalence of diabetes, advanced healthcare infrastructure, and substantial investment in medical research and development.
Product Segment Dominance:
- Devices: The Surgical Devices segment is a major contributor, with Intraocular Lenses (IOLs) playing a crucial role due to the high incidence of cataracts, often exacerbated by diabetes. The demand for advanced IOLs, including toric and multifocal options, is robust.
- Drivers: Aging population, increasing cataract surgeries, patient demand for improved vision correction post-surgery.
- Diagnostic Devices: Within diagnostic devices, fundus cameras and optical coherence tomography (OCT) systems are critical for early detection and monitoring of diabetic retinopathy and macular edema.
- Drivers: Rising awareness of early detection, government initiatives for diabetic eye screening programs, technological advancements in imaging resolution and AI integration.
Disease Segment Dominance:
- Glaucoma: This segment is significant due to the higher risk of developing glaucoma in individuals with diabetes.
- Drivers: Increasing diabetic population, lack of early symptoms leading to delayed diagnosis, development of advanced glaucoma treatment devices and drugs.
- Cataract: As mentioned, cataract is a primary concern, often necessitating surgical intervention.
- Drivers: High prevalence in the aging diabetic population, availability of advanced surgical techniques and IOLs.
- Age-Related Macular Degeneration (AMD): While not solely diabetes-related, diabetes can accelerate or worsen AMD, making it a key disease area.
- Drivers: Growing elderly population, increasing understanding of the link between diabetes and macular health.
Geography:
- United States: Dominates due to market size, advanced healthcare systems, and high adoption rates of new technologies.
- Drivers: High disposable income for healthcare, robust reimbursement policies, extensive R&D infrastructure.
- Canada: Shows steady growth, supported by a universal healthcare system with increasing focus on chronic disease management.
- Drivers: Government investment in public health, growing diabetic population.
- Mexico: Presents a growing market with increasing awareness and improving healthcare accessibility.
- Drivers: Growing middle class, expanding private healthcare sector, increasing prevalence of diabetes.
North America Diabetic Eye Disease Devices Industry Product Developments
The North America Diabetic Eye Disease Devices Industry is witnessing a continuous stream of innovative product developments aimed at enhancing diagnosis, treatment, and patient outcomes. Advancements in diagnostic devices include the integration of artificial intelligence into OCT and fundus imaging systems for automated detection and grading of diabetic retinopathy, significantly improving diagnostic speed and accuracy. In surgical devices, next-generation intraocular lenses with enhanced optical performance and features to mitigate intraoperative complications are being introduced. Ophthalmic lasers are evolving towards greater precision and minimally invasive applications for procedures like laser photocoagulation and capsulotomy. Pharmaceutical companies are focusing on novel drug formulations and delivery systems for managing retinal diseases and dry eye, including sustained-release implants and targeted therapies. These developments collectively underscore the industry's commitment to addressing the complex needs of diabetic eye care through cutting-edge technology.
Challenges in the North America Diabetic Eye Disease Devices Industry Market
The North America Diabetic Eye Disease Devices Industry faces several significant challenges. High development and regulatory costs for new medical devices and pharmaceuticals can be a substantial barrier, with lengthy approval processes through agencies like the FDA often leading to delayed market entry. Reimbursement complexities and varying coverage policies across different healthcare systems can impact market access and adoption rates, especially for novel, high-cost technologies. Intense competition among established players and emerging innovators necessitates continuous investment in R&D to maintain a competitive edge. Supply chain disruptions, as witnessed in recent global events, can affect the availability of critical components and finished products.
Forces Driving North America Diabetic Eye Disease Devices Industry Growth
Several key forces are propelling the growth of the North America Diabetic Eye Disease Devices Industry. The rising global incidence of diabetes, a primary risk factor for various eye conditions, is a fundamental driver. Technological advancements in diagnostic imaging, laser technology, and surgical instrumentation are enabling earlier detection and more effective treatment options. Increasing healthcare expenditure and supportive government initiatives aimed at managing chronic diseases, including diabetes, are bolstering market demand. Furthermore, a growing awareness among patients and healthcare providers regarding the importance of regular eye screenings and proactive management of diabetic eye complications is fueling market expansion.
Challenges in the North America Diabetic Eye Disease Devices Industry Market
Long-term growth catalysts for the North America Diabetic Eye Disease Devices Industry lie in continued innovation and strategic market expansion. The development of preventative and regenerative therapies for diabetic eye diseases, moving beyond current treatment paradigms, represents a significant future opportunity. Increased adoption of digital health solutions, including telemedicine and AI-driven diagnostic platforms, will enhance accessibility and efficiency of eye care delivery. Partnerships and collaborations between device manufacturers, pharmaceutical companies, and research institutions will accelerate the development and commercialization of novel solutions. Furthermore, expanding into underserved markets and focusing on cost-effective solutions can unlock substantial growth potential.
Emerging Opportunities in North America Diabetic Eye Disease Devices Industry
Emerging opportunities within the North America Diabetic Eye Disease Devices Industry are multifaceted. The integration of artificial intelligence and machine learning in diagnostic imaging presents a significant avenue for improving diagnostic accuracy and workflow efficiency. The growing demand for minimally invasive and patient-centric treatments is driving innovation in surgical techniques and drug delivery systems, such as sustained-release implants and gene therapies. The expansion of teleophthalmology and remote patient monitoring offers a pathway to increase access to care, particularly in rural and remote areas. Furthermore, the development of personalized medicine approaches, tailored to individual patient needs and genetic profiles, holds immense promise for optimizing treatment outcomes for diabetic eye diseases.
Leading Players in the North America Diabetic Eye Disease Devices Industry Sector
- Bausch Health Companies Inc (Bausch + Lomb)
- Ziemer Group AG
- Carl Zeiss Meditec AG
- Johnson & Johnson
- Haag-Streit Group
- Topcon Corporation
- Essilor International SA
- Alcon Inc
- Nidek Co Ltd
Key Milestones in North America Diabetic Eye Disease Devices Industry Industry
- March 2022: Epipole, an emerging provider of world-class handheld fundus cameras, launched their new epiCam fundus camera to the United States Eye Care market at the Vision Expo East in New York City.
- January 2022: Aequus Pharmaceuticals Inc., a specialty pharmaceutical company, submitted a New Drug Submission (NDS) application to Health Canada for preservative-free bimatoprost 0.03% eye drops termed 'Zimed PF.' At this stage, Health Canada accepted the submission for Screening.
Strategic Outlook for North America Diabetic Eye Disease Devices Industry Market
The strategic outlook for the North America Diabetic Eye Disease Devices Industry is characterized by robust growth and continuous innovation. Key accelerators include the increasing focus on early detection and prevention of diabetic eye complications, driven by advancements in AI-powered diagnostic tools. The development of novel therapeutic agents and advanced surgical technologies, such as femtosecond laser technology and sophisticated intraocular lenses, will continue to reshape treatment protocols. Furthermore, the growing adoption of telemedicine and digital health platforms will enhance patient access and streamline care delivery. Strategic collaborations and mergers are expected to further consolidate the market and foster technological integration, paving the way for more personalized and effective management of diabetic eye diseases.
North America Diabetic Eye Disease Devices Industry Segmentation
-
1. Product
-
1.1. Devices
-
1.1.1. Surgical Devices
- 1.1.1.1. Intraocular Lenses
- 1.1.1.2. Ophthalmic Lasers
- 1.1.1.3. Other Surgical Devices
- 1.1.2. Diagnostic Devices
-
1.1.1. Surgical Devices
-
1.2. Drugs
- 1.2.1. Glaucoma Drugs
- 1.2.2. Retinal Disorder Drugs
- 1.2.3. Dry Eye Drugs
- 1.2.4. Allergic Conjunctivitis and Inflammation Drugs
- 1.2.5. Other Drugs
-
1.1. Devices
-
2. Disease
- 2.1. Glaucoma
- 2.2. Cataract
- 2.3. Age-Related Macular Degeneration
- 2.4. Inflammatory Diseases
- 2.5. Refractive Disorders
- 2.6. Other Diseases
-
3. Geography
- 3.1. United States
- 3.2. Canada
- 3.3. Mexico
North America Diabetic Eye Disease Devices Industry Segmentation By Geography
- 1. United States
- 2. Canada
- 3. Mexico
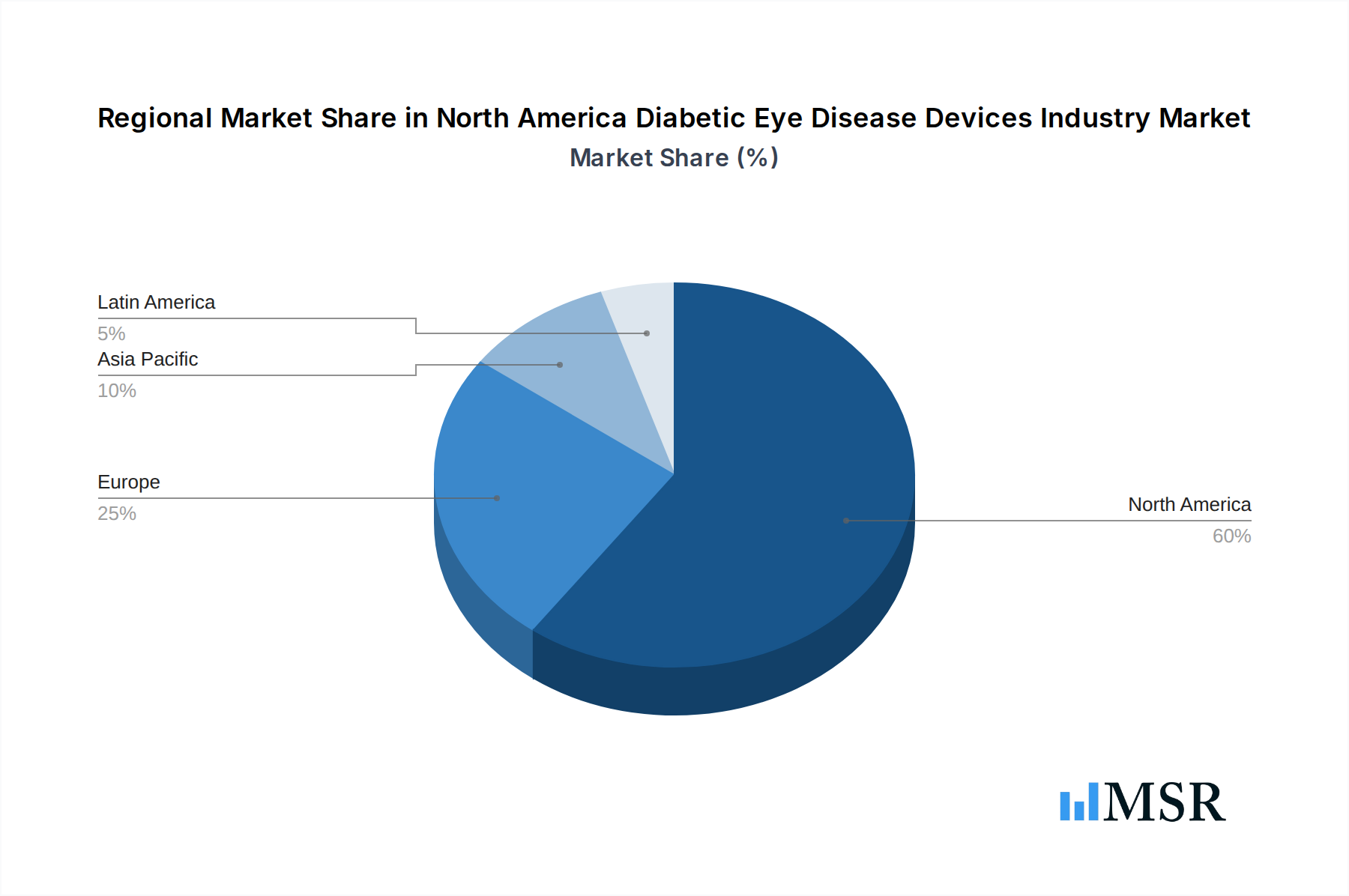
North America Diabetic Eye Disease Devices Industry Regional Market Share

Geographic Coverage of North America Diabetic Eye Disease Devices Industry
North America Diabetic Eye Disease Devices Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.96% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. MSR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product
- 5.1.1. Devices
- 5.1.1.1. Surgical Devices
- 5.1.1.1.1. Intraocular Lenses
- 5.1.1.1.2. Ophthalmic Lasers
- 5.1.1.1.3. Other Surgical Devices
- 5.1.1.2. Diagnostic Devices
- 5.1.1.1. Surgical Devices
- 5.1.2. Drugs
- 5.1.2.1. Glaucoma Drugs
- 5.1.2.2. Retinal Disorder Drugs
- 5.1.2.3. Dry Eye Drugs
- 5.1.2.4. Allergic Conjunctivitis and Inflammation Drugs
- 5.1.2.5. Other Drugs
- 5.1.1. Devices
- 5.2. Market Analysis, Insights and Forecast - by Disease
- 5.2.1. Glaucoma
- 5.2.2. Cataract
- 5.2.3. Age-Related Macular Degeneration
- 5.2.4. Inflammatory Diseases
- 5.2.5. Refractive Disorders
- 5.2.6. Other Diseases
- 5.3. Market Analysis, Insights and Forecast - by Geography
- 5.3.1. United States
- 5.3.2. Canada
- 5.3.3. Mexico
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. United States
- 5.4.2. Canada
- 5.4.3. Mexico
- 5.1. Market Analysis, Insights and Forecast - by Product
- 6. North America Diabetic Eye Disease Devices Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product
- 6.1.1. Devices
- 6.1.1.1. Surgical Devices
- 6.1.1.1.1. Intraocular Lenses
- 6.1.1.1.2. Ophthalmic Lasers
- 6.1.1.1.3. Other Surgical Devices
- 6.1.1.2. Diagnostic Devices
- 6.1.1.1. Surgical Devices
- 6.1.2. Drugs
- 6.1.2.1. Glaucoma Drugs
- 6.1.2.2. Retinal Disorder Drugs
- 6.1.2.3. Dry Eye Drugs
- 6.1.2.4. Allergic Conjunctivitis and Inflammation Drugs
- 6.1.2.5. Other Drugs
- 6.1.1. Devices
- 6.2. Market Analysis, Insights and Forecast - by Disease
- 6.2.1. Glaucoma
- 6.2.2. Cataract
- 6.2.3. Age-Related Macular Degeneration
- 6.2.4. Inflammatory Diseases
- 6.2.5. Refractive Disorders
- 6.2.6. Other Diseases
- 6.3. Market Analysis, Insights and Forecast - by Geography
- 6.3.1. United States
- 6.3.2. Canada
- 6.3.3. Mexico
- 6.1. Market Analysis, Insights and Forecast - by Product
- 7. United States North America Diabetic Eye Disease Devices Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product
- 7.1.1. Devices
- 7.1.1.1. Surgical Devices
- 7.1.1.1.1. Intraocular Lenses
- 7.1.1.1.2. Ophthalmic Lasers
- 7.1.1.1.3. Other Surgical Devices
- 7.1.1.2. Diagnostic Devices
- 7.1.1.1. Surgical Devices
- 7.1.2. Drugs
- 7.1.2.1. Glaucoma Drugs
- 7.1.2.2. Retinal Disorder Drugs
- 7.1.2.3. Dry Eye Drugs
- 7.1.2.4. Allergic Conjunctivitis and Inflammation Drugs
- 7.1.2.5. Other Drugs
- 7.1.1. Devices
- 7.2. Market Analysis, Insights and Forecast - by Disease
- 7.2.1. Glaucoma
- 7.2.2. Cataract
- 7.2.3. Age-Related Macular Degeneration
- 7.2.4. Inflammatory Diseases
- 7.2.5. Refractive Disorders
- 7.2.6. Other Diseases
- 7.3. Market Analysis, Insights and Forecast - by Geography
- 7.3.1. United States
- 7.3.2. Canada
- 7.3.3. Mexico
- 7.1. Market Analysis, Insights and Forecast - by Product
- 8. Canada North America Diabetic Eye Disease Devices Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product
- 8.1.1. Devices
- 8.1.1.1. Surgical Devices
- 8.1.1.1.1. Intraocular Lenses
- 8.1.1.1.2. Ophthalmic Lasers
- 8.1.1.1.3. Other Surgical Devices
- 8.1.1.2. Diagnostic Devices
- 8.1.1.1. Surgical Devices
- 8.1.2. Drugs
- 8.1.2.1. Glaucoma Drugs
- 8.1.2.2. Retinal Disorder Drugs
- 8.1.2.3. Dry Eye Drugs
- 8.1.2.4. Allergic Conjunctivitis and Inflammation Drugs
- 8.1.2.5. Other Drugs
- 8.1.1. Devices
- 8.2. Market Analysis, Insights and Forecast - by Disease
- 8.2.1. Glaucoma
- 8.2.2. Cataract
- 8.2.3. Age-Related Macular Degeneration
- 8.2.4. Inflammatory Diseases
- 8.2.5. Refractive Disorders
- 8.2.6. Other Diseases
- 8.3. Market Analysis, Insights and Forecast - by Geography
- 8.3.1. United States
- 8.3.2. Canada
- 8.3.3. Mexico
- 8.1. Market Analysis, Insights and Forecast - by Product
- 9. Mexico North America Diabetic Eye Disease Devices Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product
- 9.1.1. Devices
- 9.1.1.1. Surgical Devices
- 9.1.1.1.1. Intraocular Lenses
- 9.1.1.1.2. Ophthalmic Lasers
- 9.1.1.1.3. Other Surgical Devices
- 9.1.1.2. Diagnostic Devices
- 9.1.1.1. Surgical Devices
- 9.1.2. Drugs
- 9.1.2.1. Glaucoma Drugs
- 9.1.2.2. Retinal Disorder Drugs
- 9.1.2.3. Dry Eye Drugs
- 9.1.2.4. Allergic Conjunctivitis and Inflammation Drugs
- 9.1.2.5. Other Drugs
- 9.1.1. Devices
- 9.2. Market Analysis, Insights and Forecast - by Disease
- 9.2.1. Glaucoma
- 9.2.2. Cataract
- 9.2.3. Age-Related Macular Degeneration
- 9.2.4. Inflammatory Diseases
- 9.2.5. Refractive Disorders
- 9.2.6. Other Diseases
- 9.3. Market Analysis, Insights and Forecast - by Geography
- 9.3.1. United States
- 9.3.2. Canada
- 9.3.3. Mexico
- 9.1. Market Analysis, Insights and Forecast - by Product
- 10. Competitive Analysis
- 10.1. Company Profiles
- 10.1.1 Bausch Health Companies Inc (Bausch + Lomb)
- 10.1.1.1. Company Overview
- 10.1.1.2. Products
- 10.1.1.3. Company Financials
- 10.1.1.4. SWOT Analysis
- 10.1.2 Ziemer Group AG
- 10.1.2.1. Company Overview
- 10.1.2.2. Products
- 10.1.2.3. Company Financials
- 10.1.2.4. SWOT Analysis
- 10.1.3 Carl Zeiss Meditec AG
- 10.1.3.1. Company Overview
- 10.1.3.2. Products
- 10.1.3.3. Company Financials
- 10.1.3.4. SWOT Analysis
- 10.1.4 Johnson & Johnson
- 10.1.4.1. Company Overview
- 10.1.4.2. Products
- 10.1.4.3. Company Financials
- 10.1.4.4. SWOT Analysis
- 10.1.5 Haag-Streit Group
- 10.1.5.1. Company Overview
- 10.1.5.2. Products
- 10.1.5.3. Company Financials
- 10.1.5.4. SWOT Analysis
- 10.1.6 Topcon Corporation
- 10.1.6.1. Company Overview
- 10.1.6.2. Products
- 10.1.6.3. Company Financials
- 10.1.6.4. SWOT Analysis
- 10.1.7 Essilor International SA
- 10.1.7.1. Company Overview
- 10.1.7.2. Products
- 10.1.7.3. Company Financials
- 10.1.7.4. SWOT Analysis
- 10.1.8 Alcon Inc
- 10.1.8.1. Company Overview
- 10.1.8.2. Products
- 10.1.8.3. Company Financials
- 10.1.8.4. SWOT Analysis
- 10.1.9 Nidek Co Ltd
- 10.1.9.1. Company Overview
- 10.1.9.2. Products
- 10.1.9.3. Company Financials
- 10.1.9.4. SWOT Analysis
- 10.1.1 Bausch Health Companies Inc (Bausch + Lomb)
- 10.2. Market Entropy
- 10.2.1 Company's Key Areas Served
- 10.2.2 Recent Developments
- 10.3. Company Market Share Analysis 2025
- 10.3.1 Top 5 Companies Market Share Analysis
- 10.3.2 Top 3 Companies Market Share Analysis
- 10.4. List of Potential Customers
- 11. Research Methodology
List of Figures
- Figure 1: North America Diabetic Eye Disease Devices Industry Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: North America Diabetic Eye Disease Devices Industry Share (%) by Company 2025
List of Tables
- Table 1: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 2: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 3: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Disease 2020 & 2033
- Table 4: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Disease 2020 & 2033
- Table 5: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Geography 2020 & 2033
- Table 6: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Geography 2020 & 2033
- Table 7: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Region 2020 & 2033
- Table 8: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Region 2020 & 2033
- Table 9: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 10: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 11: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Disease 2020 & 2033
- Table 12: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Disease 2020 & 2033
- Table 13: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Geography 2020 & 2033
- Table 14: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Geography 2020 & 2033
- Table 15: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 16: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 17: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 18: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 19: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Disease 2020 & 2033
- Table 20: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Disease 2020 & 2033
- Table 21: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Geography 2020 & 2033
- Table 22: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Geography 2020 & 2033
- Table 23: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 24: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 25: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 26: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 27: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Disease 2020 & 2033
- Table 28: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Disease 2020 & 2033
- Table 29: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Geography 2020 & 2033
- Table 30: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Geography 2020 & 2033
- Table 31: North America Diabetic Eye Disease Devices Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 32: North America Diabetic Eye Disease Devices Industry Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the North America Diabetic Eye Disease Devices Industry?
The projected CAGR is approximately 9.96%.
2. Which companies are prominent players in the North America Diabetic Eye Disease Devices Industry?
Key companies in the market include Bausch Health Companies Inc (Bausch + Lomb), Ziemer Group AG, Carl Zeiss Meditec AG, Johnson & Johnson, Haag-Streit Group, Topcon Corporation, Essilor International SA, Alcon Inc, Nidek Co Ltd.
3. What are the main segments of the North America Diabetic Eye Disease Devices Industry?
The market segments include Product, Disease, Geography.
4. Can you provide details about the market size?
The market size is estimated to be USD 12.65 billion as of 2022.
5. What are some drivers contributing to market growth?
Demographic Shift and Impact of Prevalence of Eye Disease; Technological Advancements in the Field of Ophthalmology.
6. What are the notable trends driving market growth?
Cataract Segment is Expected to Show Better Growth in the Forecast Years.
7. Are there any restraints impacting market growth?
Higher Cost of the Ophthalmic Devices and Strict FDA Guidelines for Drug Approval.
8. Can you provide examples of recent developments in the market?
March 2022- Epipole, an emerging provider of world-class handheld fundus cameras, launched their new epiCam fundus camera to the United States Eye Care market at the Vision Expo East in New York City.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "North America Diabetic Eye Disease Devices Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the North America Diabetic Eye Disease Devices Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the North America Diabetic Eye Disease Devices Industry?
To stay informed about further developments, trends, and reports in the North America Diabetic Eye Disease Devices Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
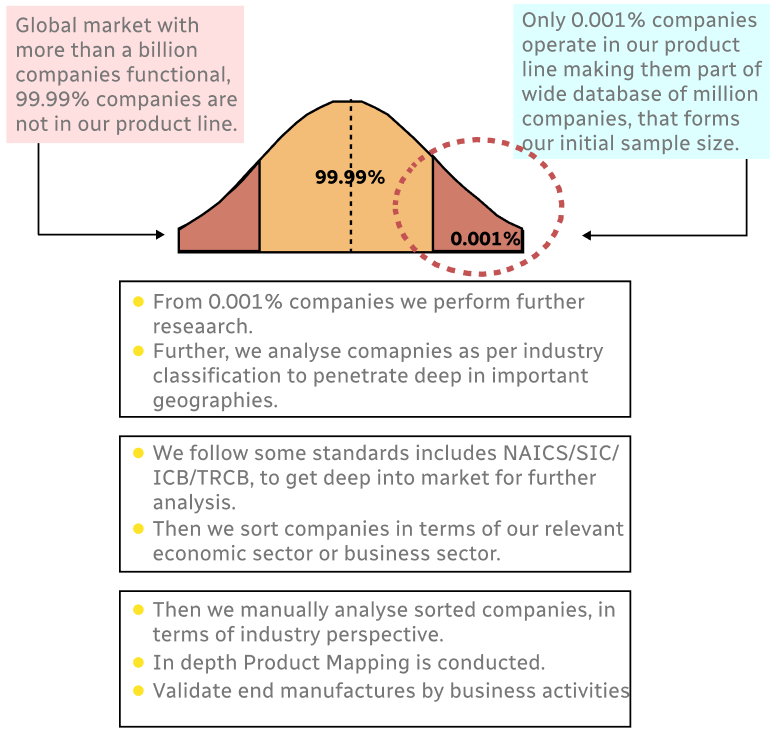
Step 1 - Identification of Relevant Samples Size from Population Database
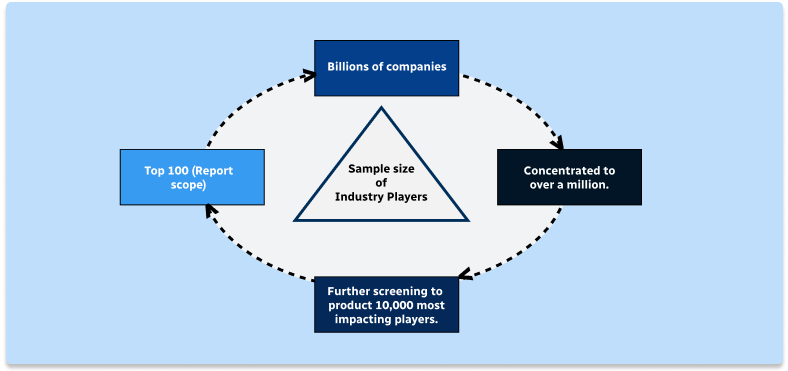
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
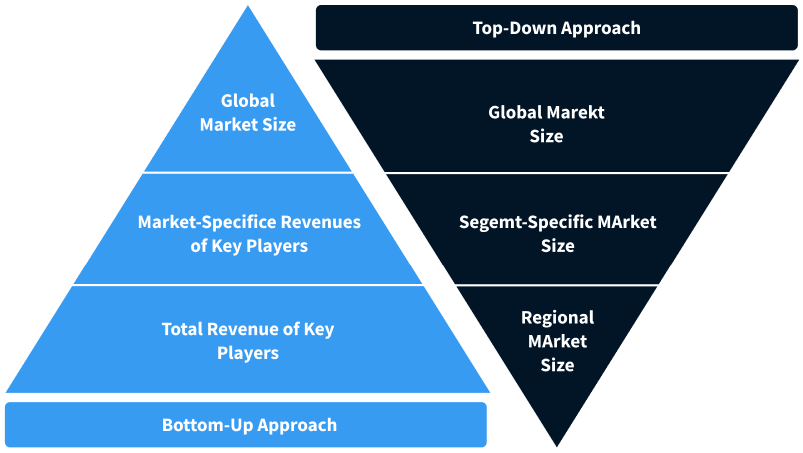
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


