Key Insights
The In Vivo Contract Research Organization (CRO) Market is poised for significant expansion, driven by the increasing complexity of drug discovery and development and the growing demand for specialized preclinical research services. The market is estimated to reach USD 7.67 billion in 2025, with a projected Compound Annual Growth Rate (CAGR) of 8.7% during the forecast period of 2025-2033. This robust growth is underpinned by critical drivers such as the rising prevalence of chronic diseases, necessitating innovative therapeutic solutions, and the escalating investments in pharmaceutical R&D. Furthermore, the increasing outsourcing trend by pharmaceutical and biotechnology companies, seeking cost-effectiveness, efficiency, and access to advanced technologies, is a primary catalyst. The market is segmented by indications including Blood Cancer, Solid Tumors, and Other Indications, with Syngeneic, Xenograft, and Patient-Derived Xenograft (PDX) models forming key platforms for preclinical efficacy and safety assessments.
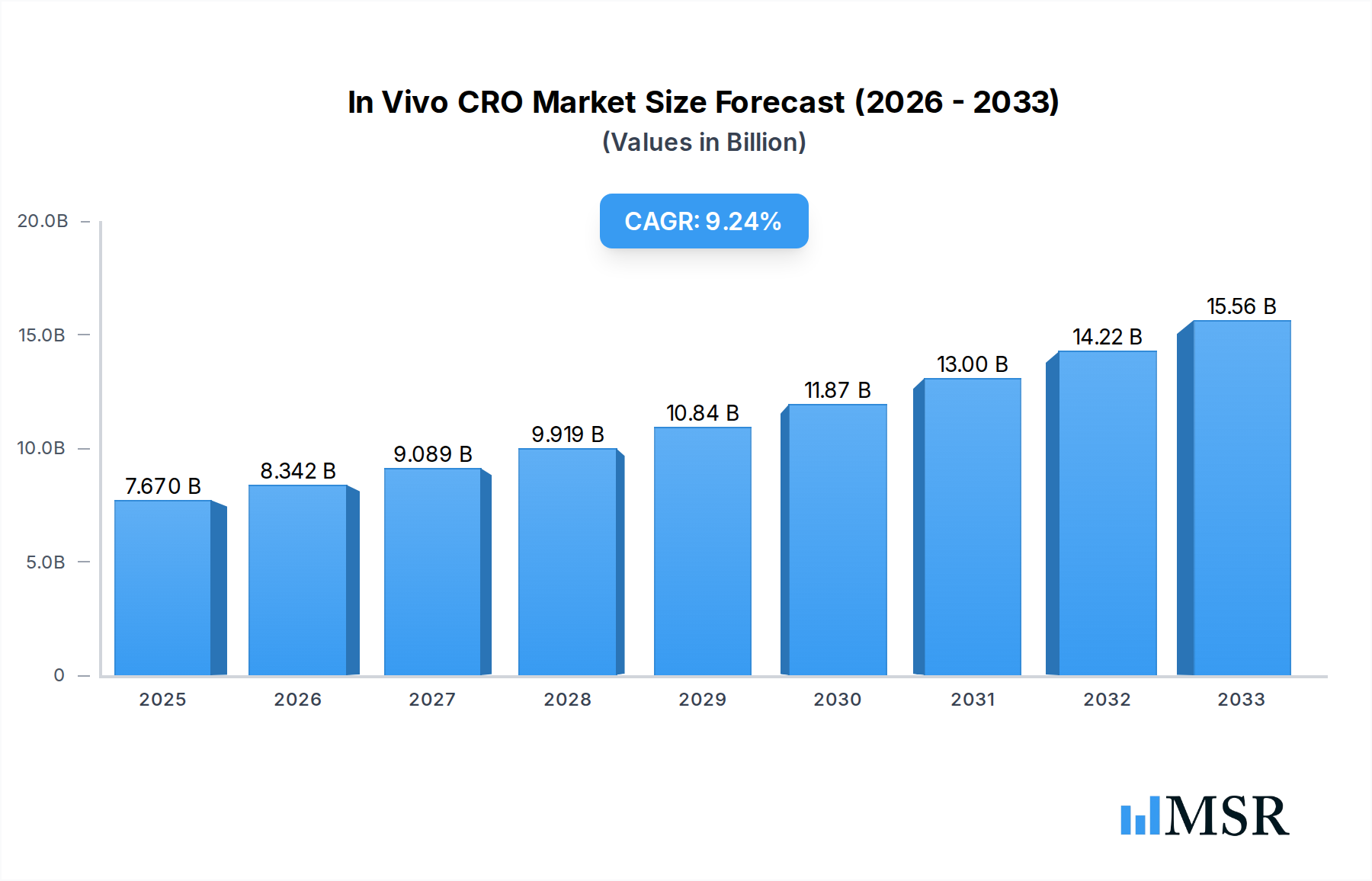
In Vivo CRO Market Market Size (In Billion)

The In Vivo CRO Market is experiencing a dynamic shift, with a notable trend towards the adoption of advanced preclinical models, particularly Patient-Derived Xenografts (PDX), which offer greater translational relevance to human disease. This focus on more predictive models is crucial for de-risking drug development pipelines. Key restraints, however, include the high cost associated with conducting in vivo studies and stringent regulatory compliance requirements. Despite these challenges, the market is characterized by the presence of leading companies such as Charles River Laboratory Inc., Champions Oncology Inc., and The Jackson Laboratory, actively engaged in strategic collaborations and service expansions. Geographically, North America is expected to lead the market, followed by Europe and the Asia Pacific, with China emerging as a significant growth hub due to its expanding biopharmaceutical ecosystem and increasing R&D investments.
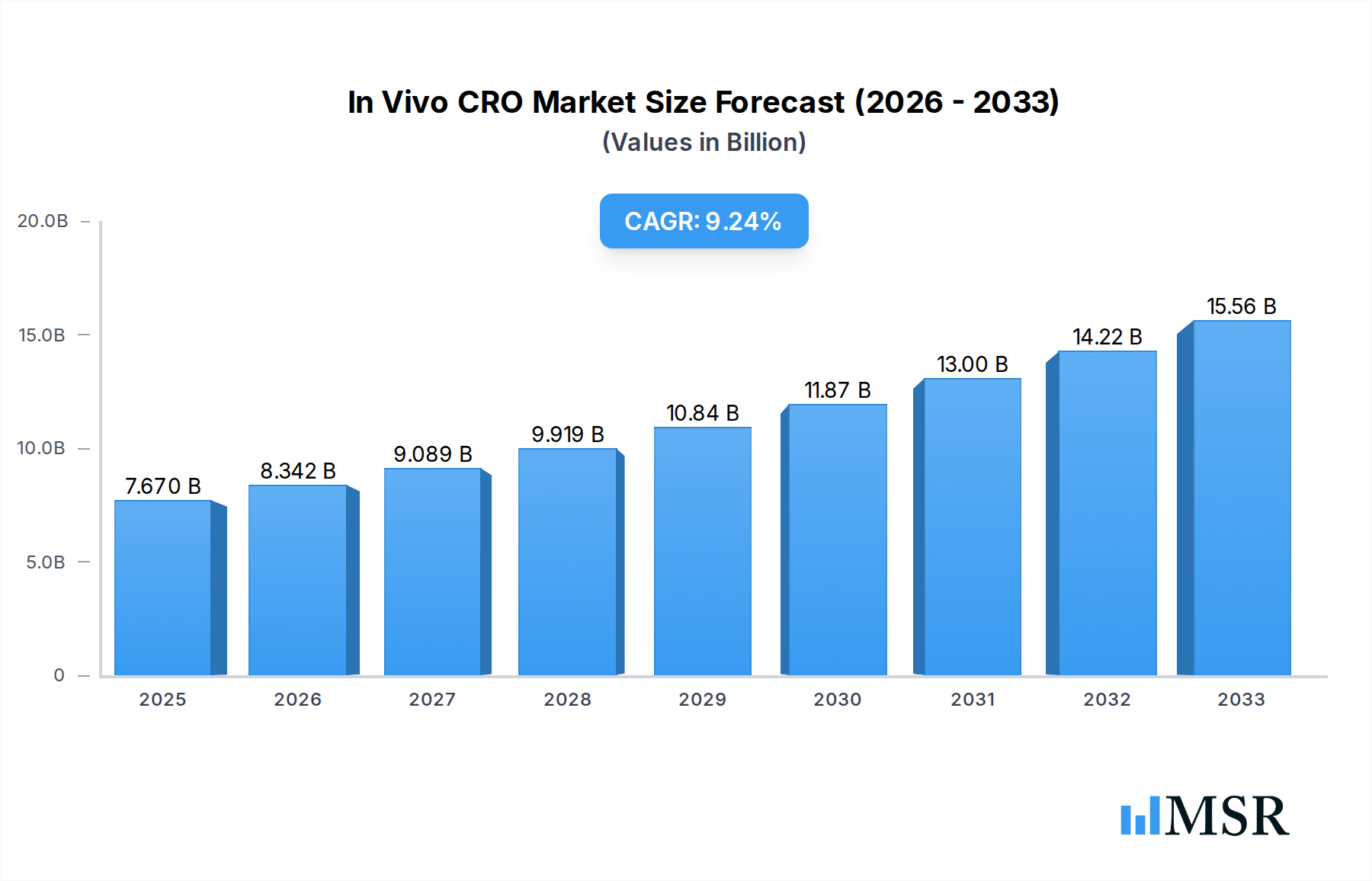
In Vivo CRO Market Company Market Share

Here is an SEO-optimized and engaging report description for the In Vivo CRO Market, incorporating the requested details and structure.
Report Title: In Vivo CRO Market: Size, Share, Trends, and Forecasts 2019-2033 – Driving Pre-Clinical Drug Development
Report Description:
Uncover the dynamic landscape of the In Vivo Contract Research Organization (CRO) Market, a critical sector underpinning pre-clinical drug development and oncology research. This comprehensive report, spanning the historical period of 2019–2024 and projecting to 2033, provides unparalleled insights into market size, growth drivers, and emerging opportunities. With a base year of 2025, the report meticulously analyzes the market's trajectory, driven by innovations in cancer vaccine development, RNA-based therapeutics, and advanced pre-clinical models.
The In Vivo CRO Market is experiencing robust expansion, fueled by increasing R&D investments in biotechnology and pharmaceuticals. This report delves into the key segments, including Blood Cancer, Solid Tumors, and Other Indications, alongside crucial in vivo models such as Syngeneic, Xenograft, and Patient Derived Xenograft (PDX). Gain a competitive edge with deep dives into market concentration, industry trends, product developments, and the strategic initiatives of leading players.
Key Stakeholders: Pharmaceutical companies, biotechnology firms, academic institutions, venture capitalists, and CROs will find actionable intelligence to inform strategic decisions, identify partnership opportunities, and navigate the complexities of the global in vivo research services market.
In Vivo CRO Market Market Concentration & Dynamics
The In Vivo CRO Market exhibits a moderate to high concentration, characterized by the presence of established global players alongside specialized regional entities. Market leaders are investing heavily in expanding their service portfolios, technological capabilities, and geographical reach to capture a larger share of this rapidly evolving sector. Innovation ecosystems are thriving, driven by the urgent need for efficient and reliable pre-clinical testing for novel therapeutics, particularly in oncology. Regulatory frameworks, while stringent, are adapting to accommodate new modalities like RNA-based therapeutics, creating both challenges and opportunities for CROs. The threat of substitute products, such as advanced in vitro models and computational approaches, is present but remains limited for comprehensive efficacy and toxicity evaluations that require live biological systems. End-user trends are heavily influenced by the increasing complexity of drug candidates, the demand for personalized medicine approaches like Patient Derived Xenograft (PDX) models, and the growing outsourcing propensity of pharmaceutical giants. Merger and acquisition (M&A) activities are a significant aspect of market dynamics, with companies seeking to consolidate capabilities, acquire innovative technologies, or gain access to new markets. These M&A deals, expected to be in the double digits annually, are crucial for shaping the competitive landscape and driving market consolidation. The market share of the top five players is estimated to be between 45-60%, indicating a significant but not fully monopolized industry structure.
In Vivo CRO Market Industry Insights & Trends
The In Vivo CRO Market is poised for substantial growth, projected to reach a market size of approximately $25.5 billion by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of around 9.8% during the forecast period of 2025–2033. This impressive expansion is propelled by a confluence of factors, primarily the escalating global burden of chronic diseases, especially cancer, and the subsequent surge in research and development (R&D) activities by pharmaceutical and biotechnology companies. The increasing complexity of drug candidates, including biologics, cell and gene therapies, and innovative cancer vaccines, necessitates sophisticated and reliable in vivo testing to assess efficacy, safety, and pharmacokinetic profiles. Technological disruptions are playing a pivotal role, with advancements in animal imaging techniques, the development of more genetically engineered rodent models, and the refinement of Patient Derived Xenograft (PDX) models enhancing the predictive power of pre-clinical studies. Furthermore, the growing trend towards personalized medicine is driving demand for CROs capable of conducting studies with patient-derived materials, such as PDX models, to tailor treatments for individual patient profiles. Evolving consumer behaviors within the pharmaceutical industry, including a greater emphasis on outsourcing non-core R&D activities to specialized CROs for cost-efficiency and access to specialized expertise, are also significant growth catalysts. The demand for robust pre-clinical drug development services is further amplified by the pipeline of novel drug candidates, particularly in therapeutic areas like oncology, immunology, and neurology. The increasing regulatory scrutiny and the need for high-quality data to support regulatory submissions compel companies to partner with experienced CROs. The market is also influenced by the global expansion of the pharmaceutical industry and the rising number of clinical trials, which in turn necessitate a strong foundation of pre-clinical in vivo studies.
Key Markets & Segments Leading In Vivo CRO Market
The In Vivo CRO Market is characterized by strong leadership within specific indications and preclinical models, reflecting the current priorities in drug development.
Dominant Indications:
- Solid Tumors: This segment is a primary driver of growth within the In Vivo CRO Market. The persistent high incidence and mortality rates associated with solid tumors across various cancer types, including lung, breast, colorectal, and prostate cancers, necessitate extensive pre-clinical research. Pharmaceutical and biotechnology companies are heavily investing in the development of novel targeted therapies, immunotherapies, and combination treatments for these challenging malignancies. The demand for robust in vivo efficacy and toxicity studies for these diverse solid tumor indications consistently fuels outsourcing to CROs.
- Blood Cancer: While solid tumors often dominate, hematological malignancies, including leukemias, lymphomas, and multiple myeloma, represent a significant and growing segment. Advancements in understanding the complex biology of blood cancers, coupled with the development of novel therapeutic agents like CAR-T therapies and targeted small molecules, are increasing the need for specialized in vivo models and testing services. CROs with expertise in xenotransplantation of hematological cell lines and primary patient samples are in high demand.
- Other Indications: This broad category encompasses a diverse range of therapeutic areas beyond oncology, including infectious diseases, neurological disorders, cardiovascular diseases, and metabolic disorders. As research into these areas expands and new therapeutic modalities emerge, the demand for in vivo CRO services in these segments is steadily increasing, contributing to overall market growth.
Dominant Models:
- Patient Derived Xenograft (PDX) Models: PDX models are emerging as the gold standard in oncology pre-clinical research due to their superior ability to recapitulate human tumor heterogeneity and patient-specific responses. The increasing focus on personalized medicine and precision oncology has significantly boosted the demand for PDX models, as they allow for the testing of drug efficacy in a more clinically relevant context. CROs offering comprehensive PDX model generation, characterization, and implantation services are at the forefront of this trend.
- Xenograft Models: Standard xenograft models, utilizing human cancer cell lines implanted into immunocompromised animals, remain a workhorse for early-stage drug screening and validation. Their scalability, established protocols, and cost-effectiveness make them indispensable for initial compound evaluation. The continuous development of new cell line-derived xenografts and their application in various oncology research studies ensures their sustained importance in the In Vivo CRO Market.
- Syngeneic Models: Syngeneic models, involving the implantation of tumor cells and tumor microenvironment from the same genetic strain of mice, are crucial for studying immuno-oncology and evaluating the efficacy of immunotherapies. These models allow for the investigation of tumor-host immune interactions and the assessment of how experimental therapies modulate the immune response within the tumor microenvironment. The surge in immuno-oncology drug development has made syngeneic models a critical component of the In Vivo CRO Market.
The dominance of these segments is driven by factors such as increasing R&D investments in these specific disease areas, the pipeline of novel drug candidates, the demand for clinically relevant and predictive preclinical data, and the specialized expertise and infrastructure required for conducting studies with these models.
In Vivo CRO Market Product Developments
Product developments in the In Vivo CRO Market are centered on enhancing the predictive power and efficiency of pre-clinical drug testing. Innovations include the development of novel genetically engineered mouse models (GEMMs) that more accurately mimic human diseases, advancements in imaging technologies for non-invasive monitoring of tumor growth and drug response, and the refinement of PDX model platforms for greater biological relevance. CROs are also investing in specialized platforms for evaluating complex modalities like gene therapies and antibody-drug conjugates. The integration of AI and machine learning for data analysis and predictive modeling further streamlines the drug discovery process, offering clients accelerated timelines and more robust insights, thereby providing a significant competitive edge.
Challenges in the In Vivo CRO Market Market
The In Vivo CRO Market faces several significant challenges that can impede its growth trajectory. Regulatory hurdles, including the evolving ethical considerations for animal research and the stringent requirements for data validation and reporting across different global jurisdictions, necessitate continuous adaptation and investment in compliance. Supply chain disruptions, particularly for specialized reagents, animal models, and critical raw materials, can lead to project delays and increased operational costs. Intense competitive pressures from both established global players and emerging regional CROs can lead to price erosion and a constant need for service differentiation. Furthermore, the high cost associated with establishing and maintaining state-of-the-art in vivo research facilities, coupled with the scarcity of highly skilled scientific personnel, presents a significant barrier to entry and expansion.
Forces Driving In Vivo CRO Market Growth
Several potent forces are driving the significant growth of the In Vivo CRO Market. Technologically, advancements in genomics, proteomics, and CRISPR-Cas9 gene editing are enabling the creation of more sophisticated and human-relevant animal models, particularly for complex diseases like cancer. Economically, the escalating R&D expenditure by pharmaceutical and biotechnology companies, driven by the need to address unmet medical needs and a robust pipeline of novel drug candidates, directly translates into increased demand for in vivo CRO services. Regulatory factors also play a crucial role; while stringent, the need for high-quality, reproducible in vivo data to support regulatory submissions compels companies to partner with expert CROs. The growing trend towards outsourcing non-core R&D activities to achieve cost efficiencies and access specialized expertise further amplifies these growth drivers.
Challenges in the In Vivo CRO Market Market
The long-term growth catalysts for the In Vivo CRO Market are deeply intertwined with continuous innovation and strategic market expansion. The increasing focus on personalized medicine and precision oncology will continue to fuel demand for specialized Patient Derived Xenograft (PDX) models and companion diagnostic testing services. Partnerships between CROs and academic institutions will foster the development of novel animal models and cutting-edge research methodologies. Market expansions into emerging economies with growing pharmaceutical R&D footprints will also contribute significantly. Furthermore, the growing complexity of drug modalities, such as cell and gene therapies, will create opportunities for CROs that can develop specialized in vivo testing capabilities.
Emerging Opportunities in In Vivo CRO Market
Emerging opportunities within the In Vivo CRO Market are abundant, driven by scientific advancements and evolving healthcare landscapes. The burgeoning field of immuno-oncology presents a substantial avenue for growth, with increasing demand for syngeneic models and services that evaluate combination therapies and immune checkpoint inhibitors. The development of novel cancer vaccines, as exemplified by initiatives like Moderna and Merck's PCV, is creating new avenues for in vivo testing services related to immunogenicity and efficacy. The growing interest in RNA-based therapeutics and lipid nanoparticle (LNP) formulation development, highlighted by eTheRNA immunotherapies' service introduction, opens up opportunities for CROs to support the pre-clinical evaluation of these innovative modalities. Furthermore, the expanding application of in vivo research beyond oncology into areas like neurodegenerative diseases and rare genetic disorders offers untapped market potential.
Leading Players in the In Vivo CRO Market Sector
- Charles River Laboratory Inc
- Champions Oncology Inc
- The Jackson Laboratory
- Eurofins Scientific
- XenTech
- Taconic Biosciences Inc
- EVOTEC
- Crown Bioscience Inc
- ICON Plc
- Labcorp Drug Development (Laboratory Corporation of America Holdings)
- Living Tumor Laboratory
- Wuxi AppTec
Key Milestones in In Vivo CRO Market Industry
- October 2022: Moderna, Inc., and Merck reported that Merck had exercised its option to develop and commercialize personalized cancer vaccine (PCV) jointly mRNA-4157/V940 under the terms of its existing Collaboration and License Agreement, underscoring the growing importance of in vivo evaluation for novel cancer immunotherapies.
- March 2022: eTheRNA immunotherapies introduced a new Lipid Nanoparticle (LNP) formulation development and production service to support the discovery and early pre-clinical development of RNA-based therapeutics and vaccines, signaling an expansion of CRO capabilities to cater to emerging therapeutic modalities.
Strategic Outlook for In Vivo CRO Market Market
The strategic outlook for the In Vivo CRO Market is overwhelmingly positive, driven by a sustained increase in pharmaceutical R&D pipelines and the persistent need for robust pre-clinical validation. Future market potential will be significantly shaped by the continued adoption of advanced technologies such as artificial intelligence in data analysis and predictive modeling for in vivo studies, as well as the development of more predictive and human-relevant animal models. Strategic opportunities lie in expanding service offerings to encompass niche therapeutic areas and novel drug modalities, such as cell and gene therapies and personalized vaccines. Strengthening partnerships with pharmaceutical and biotechnology companies, alongside strategic acquisitions to enhance specialized capabilities, will be crucial for sustained growth and market leadership. The global expansion of healthcare infrastructure and increasing R&D investments in emerging markets also present significant avenues for strategic market penetration.
In Vivo CRO Market Segmentation
-
1. Indication
- 1.1. Blood Cancer
- 1.2. Solid Tumors
- 1.3. Other Indications
-
2. Model
- 2.1. Syngeneic
- 2.2. Xenograft
- 2.3. Patient Derived Xenograft (PDX)
In Vivo CRO Market Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
- 4. Middle East
-
5. GCC
- 5.1. South Africa
- 5.2. Rest of Middle East
-
6. South America
- 6.1. Brazil
- 6.2. Argentina
- 6.3. Rest of South America
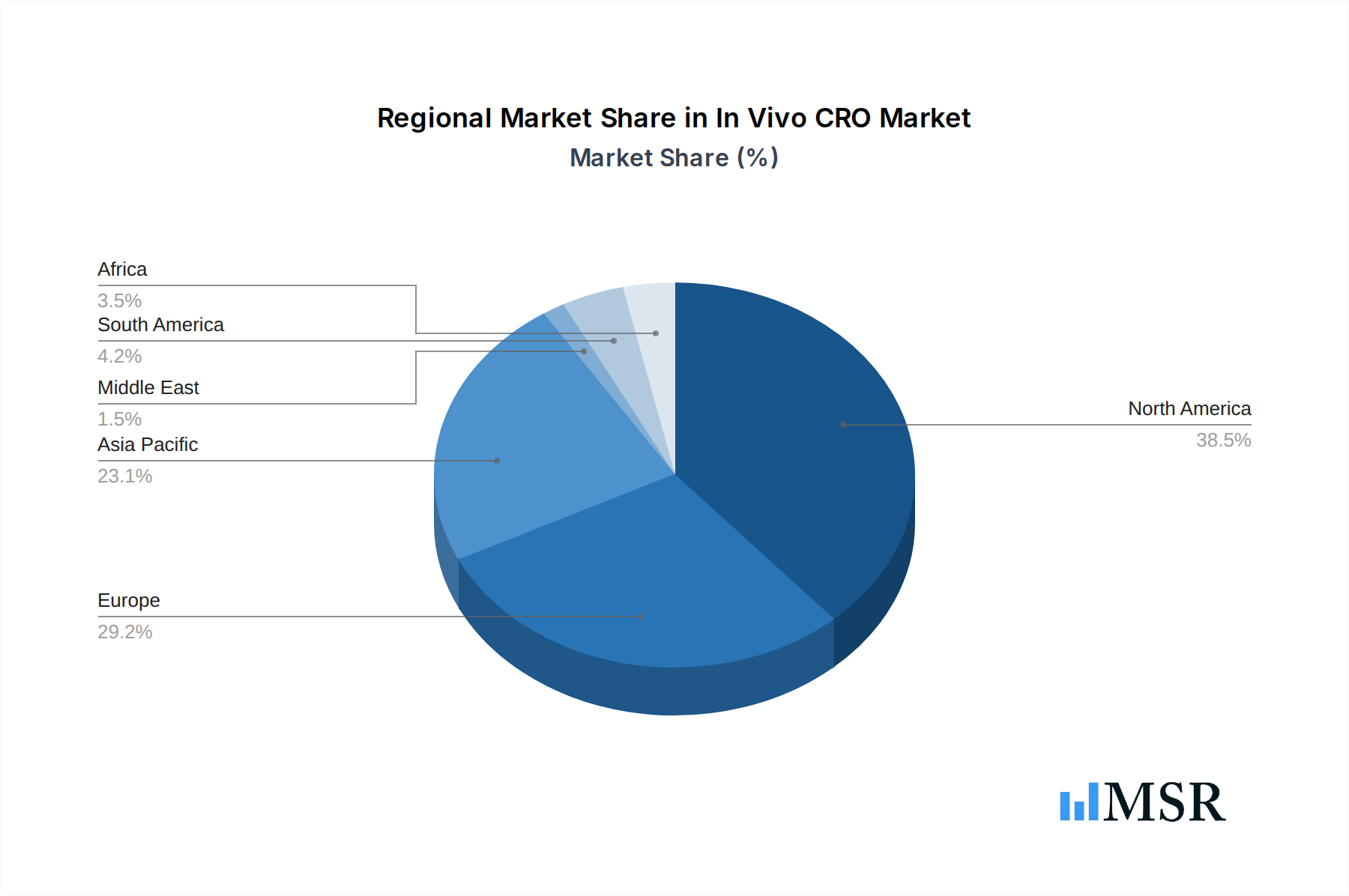
In Vivo CRO Market Regional Market Share

Geographic Coverage of In Vivo CRO Market
In Vivo CRO Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.7% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. MSR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Indication
- 5.1.1. Blood Cancer
- 5.1.2. Solid Tumors
- 5.1.3. Other Indications
- 5.2. Market Analysis, Insights and Forecast - by Model
- 5.2.1. Syngeneic
- 5.2.2. Xenograft
- 5.2.3. Patient Derived Xenograft (PDX)
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. Europe
- 5.3.3. Asia Pacific
- 5.3.4. Middle East
- 5.3.5. GCC
- 5.3.6. South America
- 5.1. Market Analysis, Insights and Forecast - by Indication
- 6. Global In Vivo CRO Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Indication
- 6.1.1. Blood Cancer
- 6.1.2. Solid Tumors
- 6.1.3. Other Indications
- 6.2. Market Analysis, Insights and Forecast - by Model
- 6.2.1. Syngeneic
- 6.2.2. Xenograft
- 6.2.3. Patient Derived Xenograft (PDX)
- 6.1. Market Analysis, Insights and Forecast - by Indication
- 7. North America In Vivo CRO Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Indication
- 7.1.1. Blood Cancer
- 7.1.2. Solid Tumors
- 7.1.3. Other Indications
- 7.2. Market Analysis, Insights and Forecast - by Model
- 7.2.1. Syngeneic
- 7.2.2. Xenograft
- 7.2.3. Patient Derived Xenograft (PDX)
- 7.1. Market Analysis, Insights and Forecast - by Indication
- 8. Europe In Vivo CRO Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Indication
- 8.1.1. Blood Cancer
- 8.1.2. Solid Tumors
- 8.1.3. Other Indications
- 8.2. Market Analysis, Insights and Forecast - by Model
- 8.2.1. Syngeneic
- 8.2.2. Xenograft
- 8.2.3. Patient Derived Xenograft (PDX)
- 8.1. Market Analysis, Insights and Forecast - by Indication
- 9. Asia Pacific In Vivo CRO Market Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Indication
- 9.1.1. Blood Cancer
- 9.1.2. Solid Tumors
- 9.1.3. Other Indications
- 9.2. Market Analysis, Insights and Forecast - by Model
- 9.2.1. Syngeneic
- 9.2.2. Xenograft
- 9.2.3. Patient Derived Xenograft (PDX)
- 9.1. Market Analysis, Insights and Forecast - by Indication
- 10. Middle East In Vivo CRO Market Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Indication
- 10.1.1. Blood Cancer
- 10.1.2. Solid Tumors
- 10.1.3. Other Indications
- 10.2. Market Analysis, Insights and Forecast - by Model
- 10.2.1. Syngeneic
- 10.2.2. Xenograft
- 10.2.3. Patient Derived Xenograft (PDX)
- 10.1. Market Analysis, Insights and Forecast - by Indication
- 11. GCC In Vivo CRO Market Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Indication
- 11.1.1. Blood Cancer
- 11.1.2. Solid Tumors
- 11.1.3. Other Indications
- 11.2. Market Analysis, Insights and Forecast - by Model
- 11.2.1. Syngeneic
- 11.2.2. Xenograft
- 11.2.3. Patient Derived Xenograft (PDX)
- 11.1. Market Analysis, Insights and Forecast - by Indication
- 12. South America In Vivo CRO Market Analysis, Insights and Forecast, 2020-2032
- 12.1. Market Analysis, Insights and Forecast - by Indication
- 12.1.1. Blood Cancer
- 12.1.2. Solid Tumors
- 12.1.3. Other Indications
- 12.2. Market Analysis, Insights and Forecast - by Model
- 12.2.1. Syngeneic
- 12.2.2. Xenograft
- 12.2.3. Patient Derived Xenograft (PDX)
- 12.1. Market Analysis, Insights and Forecast - by Indication
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 Charles River Laboratory Inc
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 Champions Oncology Inc
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 The Jackson Laboratory
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 Eurofins Scientific
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 XenTech
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 Taconic Biosciences Inc
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.7 EVOTEC
- 13.1.7.1. Company Overview
- 13.1.7.2. Products
- 13.1.7.3. Company Financials
- 13.1.7.4. SWOT Analysis
- 13.1.8 Crown Bioscience Inc
- 13.1.8.1. Company Overview
- 13.1.8.2. Products
- 13.1.8.3. Company Financials
- 13.1.8.4. SWOT Analysis
- 13.1.9 ICON Plc
- 13.1.9.1. Company Overview
- 13.1.9.2. Products
- 13.1.9.3. Company Financials
- 13.1.9.4. SWOT Analysis
- 13.1.10 Labcorp Drug Development (Laboratory Corporation of America Holdings)
- 13.1.10.1. Company Overview
- 13.1.10.2. Products
- 13.1.10.3. Company Financials
- 13.1.10.4. SWOT Analysis
- 13.1.11 Living Tumor Laboratory
- 13.1.11.1. Company Overview
- 13.1.11.2. Products
- 13.1.11.3. Company Financials
- 13.1.11.4. SWOT Analysis
- 13.1.12 Wuxi AppTec
- 13.1.12.1. Company Overview
- 13.1.12.2. Products
- 13.1.12.3. Company Financials
- 13.1.12.4. SWOT Analysis
- 13.1.1 Charles River Laboratory Inc
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Global In Vivo CRO Market Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America In Vivo CRO Market Revenue (billion), by Indication 2025 & 2033
- Figure 3: North America In Vivo CRO Market Revenue Share (%), by Indication 2025 & 2033
- Figure 4: North America In Vivo CRO Market Revenue (billion), by Model 2025 & 2033
- Figure 5: North America In Vivo CRO Market Revenue Share (%), by Model 2025 & 2033
- Figure 6: North America In Vivo CRO Market Revenue (billion), by Country 2025 & 2033
- Figure 7: North America In Vivo CRO Market Revenue Share (%), by Country 2025 & 2033
- Figure 8: Europe In Vivo CRO Market Revenue (billion), by Indication 2025 & 2033
- Figure 9: Europe In Vivo CRO Market Revenue Share (%), by Indication 2025 & 2033
- Figure 10: Europe In Vivo CRO Market Revenue (billion), by Model 2025 & 2033
- Figure 11: Europe In Vivo CRO Market Revenue Share (%), by Model 2025 & 2033
- Figure 12: Europe In Vivo CRO Market Revenue (billion), by Country 2025 & 2033
- Figure 13: Europe In Vivo CRO Market Revenue Share (%), by Country 2025 & 2033
- Figure 14: Asia Pacific In Vivo CRO Market Revenue (billion), by Indication 2025 & 2033
- Figure 15: Asia Pacific In Vivo CRO Market Revenue Share (%), by Indication 2025 & 2033
- Figure 16: Asia Pacific In Vivo CRO Market Revenue (billion), by Model 2025 & 2033
- Figure 17: Asia Pacific In Vivo CRO Market Revenue Share (%), by Model 2025 & 2033
- Figure 18: Asia Pacific In Vivo CRO Market Revenue (billion), by Country 2025 & 2033
- Figure 19: Asia Pacific In Vivo CRO Market Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East In Vivo CRO Market Revenue (billion), by Indication 2025 & 2033
- Figure 21: Middle East In Vivo CRO Market Revenue Share (%), by Indication 2025 & 2033
- Figure 22: Middle East In Vivo CRO Market Revenue (billion), by Model 2025 & 2033
- Figure 23: Middle East In Vivo CRO Market Revenue Share (%), by Model 2025 & 2033
- Figure 24: Middle East In Vivo CRO Market Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East In Vivo CRO Market Revenue Share (%), by Country 2025 & 2033
- Figure 26: GCC In Vivo CRO Market Revenue (billion), by Indication 2025 & 2033
- Figure 27: GCC In Vivo CRO Market Revenue Share (%), by Indication 2025 & 2033
- Figure 28: GCC In Vivo CRO Market Revenue (billion), by Model 2025 & 2033
- Figure 29: GCC In Vivo CRO Market Revenue Share (%), by Model 2025 & 2033
- Figure 30: GCC In Vivo CRO Market Revenue (billion), by Country 2025 & 2033
- Figure 31: GCC In Vivo CRO Market Revenue Share (%), by Country 2025 & 2033
- Figure 32: South America In Vivo CRO Market Revenue (billion), by Indication 2025 & 2033
- Figure 33: South America In Vivo CRO Market Revenue Share (%), by Indication 2025 & 2033
- Figure 34: South America In Vivo CRO Market Revenue (billion), by Model 2025 & 2033
- Figure 35: South America In Vivo CRO Market Revenue Share (%), by Model 2025 & 2033
- Figure 36: South America In Vivo CRO Market Revenue (billion), by Country 2025 & 2033
- Figure 37: South America In Vivo CRO Market Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global In Vivo CRO Market Revenue billion Forecast, by Indication 2020 & 2033
- Table 2: Global In Vivo CRO Market Revenue billion Forecast, by Model 2020 & 2033
- Table 3: Global In Vivo CRO Market Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global In Vivo CRO Market Revenue billion Forecast, by Indication 2020 & 2033
- Table 5: Global In Vivo CRO Market Revenue billion Forecast, by Model 2020 & 2033
- Table 6: Global In Vivo CRO Market Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global In Vivo CRO Market Revenue billion Forecast, by Indication 2020 & 2033
- Table 11: Global In Vivo CRO Market Revenue billion Forecast, by Model 2020 & 2033
- Table 12: Global In Vivo CRO Market Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Germany In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: United Kingdom In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: France In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Italy In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 17: Spain In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 18: Rest of Europe In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 19: Global In Vivo CRO Market Revenue billion Forecast, by Indication 2020 & 2033
- Table 20: Global In Vivo CRO Market Revenue billion Forecast, by Model 2020 & 2033
- Table 21: Global In Vivo CRO Market Revenue billion Forecast, by Country 2020 & 2033
- Table 22: China In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Japan In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: India In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Australia In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: South Korea In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Asia Pacific In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global In Vivo CRO Market Revenue billion Forecast, by Indication 2020 & 2033
- Table 29: Global In Vivo CRO Market Revenue billion Forecast, by Model 2020 & 2033
- Table 30: Global In Vivo CRO Market Revenue billion Forecast, by Country 2020 & 2033
- Table 31: Global In Vivo CRO Market Revenue billion Forecast, by Indication 2020 & 2033
- Table 32: Global In Vivo CRO Market Revenue billion Forecast, by Model 2020 & 2033
- Table 33: Global In Vivo CRO Market Revenue billion Forecast, by Country 2020 & 2033
- Table 34: South Africa In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 35: Rest of Middle East In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 36: Global In Vivo CRO Market Revenue billion Forecast, by Indication 2020 & 2033
- Table 37: Global In Vivo CRO Market Revenue billion Forecast, by Model 2020 & 2033
- Table 38: Global In Vivo CRO Market Revenue billion Forecast, by Country 2020 & 2033
- Table 39: Brazil In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 40: Argentina In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
- Table 41: Rest of South America In Vivo CRO Market Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the In Vivo CRO Market?
The projected CAGR is approximately 8.7%.
2. Which companies are prominent players in the In Vivo CRO Market?
Key companies in the market include Charles River Laboratory Inc, Champions Oncology Inc, The Jackson Laboratory, Eurofins Scientific, XenTech, Taconic Biosciences Inc, EVOTEC, Crown Bioscience Inc, ICON Plc, Labcorp Drug Development (Laboratory Corporation of America Holdings), Living Tumor Laboratory, Wuxi AppTec.
3. What are the main segments of the In Vivo CRO Market?
The market segments include Indication, Model.
4. Can you provide details about the market size?
The market size is estimated to be USD 5.11 billion as of 2022.
5. What are some drivers contributing to market growth?
Rising incidence of Cancer Globally; Increasing Initiatives by Market Players.
6. What are the notable trends driving market growth?
Solid Tumor Segment is Expected to Hold the Largest Market Share in the Oncology Based In-Vivo CRO Market.
7. Are there any restraints impacting market growth?
Availability of Alternatives and Quality issues with CRO.
8. Can you provide examples of recent developments in the market?
In October 2022, Moderna, Inc., and Merck reported that Merck had exercised its option to develop and commercialize personalized cancer vaccine (PCV) jointly mRNA-4157/V940 under the terms of its existing Collaboration and License Agreement.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "In Vivo CRO Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the In Vivo CRO Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the In Vivo CRO Market?
To stay informed about further developments, trends, and reports in the In Vivo CRO Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
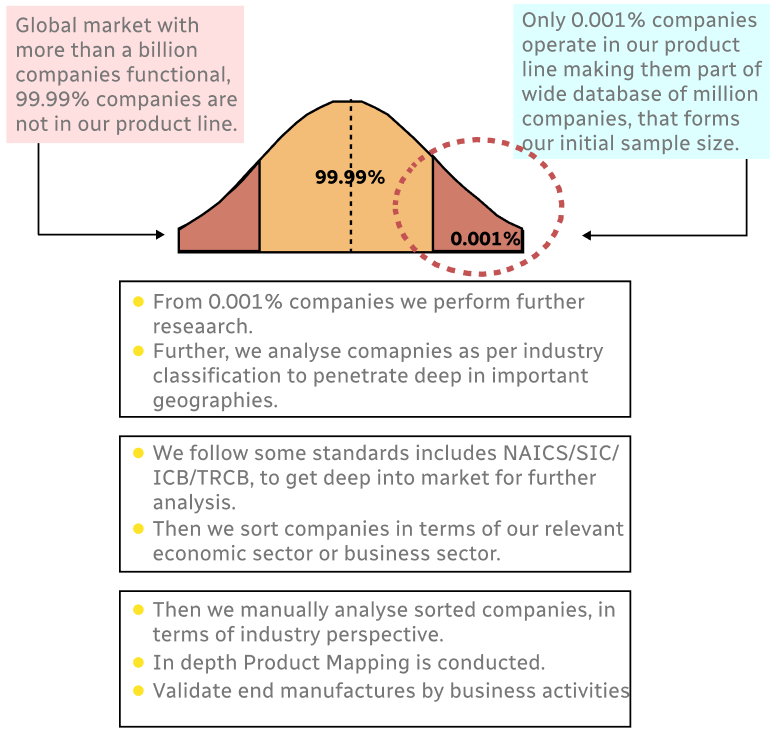
Step 1 - Identification of Relevant Samples Size from Population Database
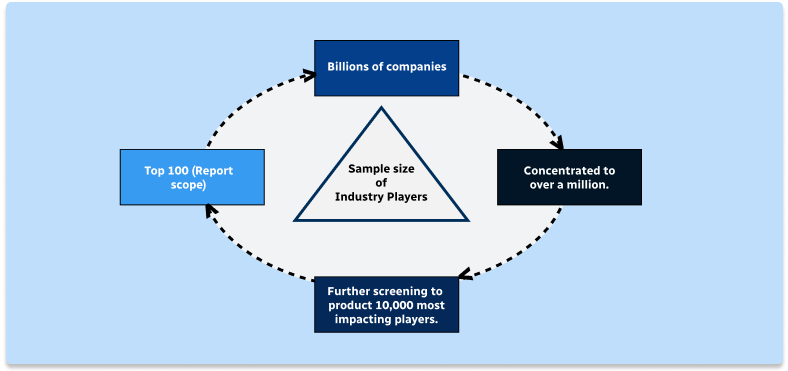
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
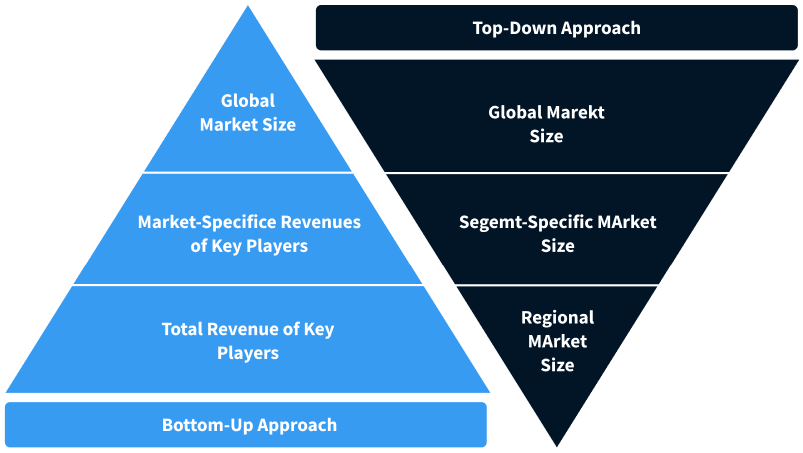
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
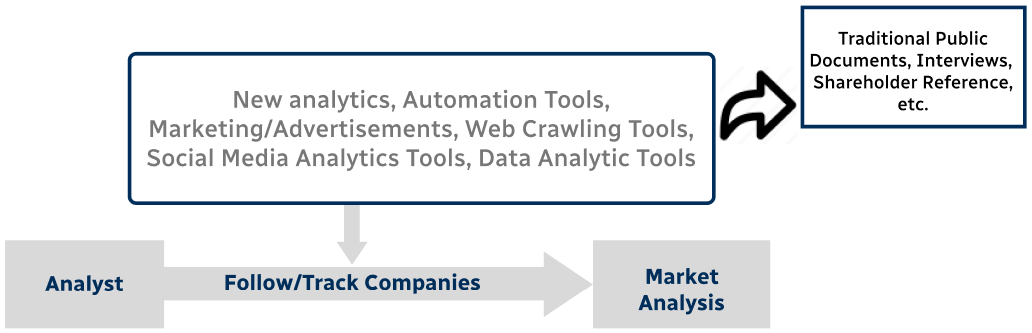
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


