Key Insights
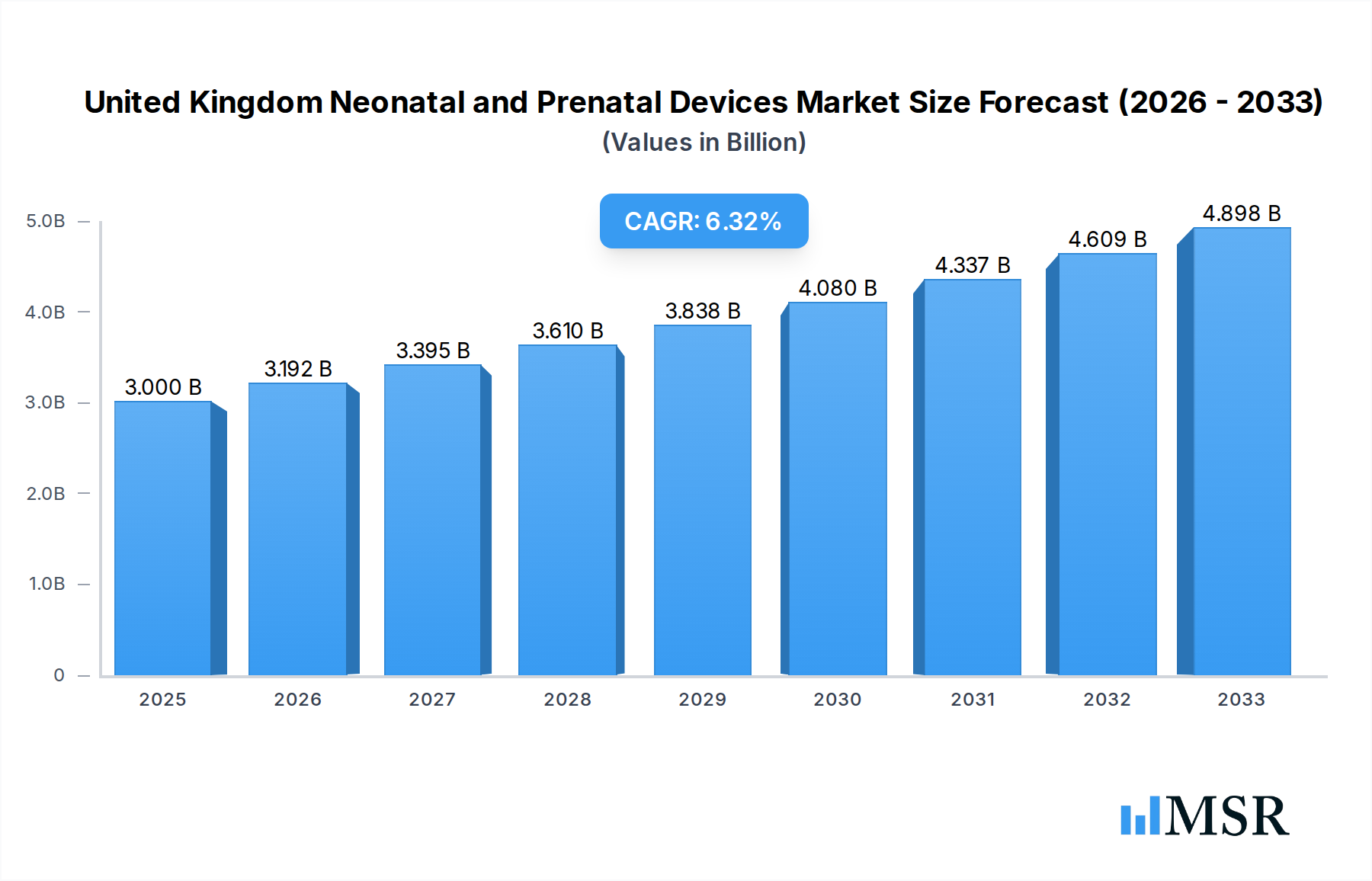
The United Kingdom's Neonatal and Prenatal Devices Market is poised for robust expansion, driven by increasing awareness of maternal and infant health, technological advancements in diagnostic and monitoring equipment, and a growing emphasis on early detection of fetal and neonatal complications. The market is projected to reach an estimated $3 billion in 2025, with a healthy Compound Annual Growth Rate (CAGR) of 6.4% anticipated to persist through 2033. This growth is significantly fueled by the escalating demand for advanced prenatal screening and diagnostic tools like high-resolution ultrasound devices and non-invasive fetal monitoring systems, which are instrumental in improving birth outcomes. Concurrently, the neonatal segment is witnessing increased adoption of sophisticated incubators, advanced respiratory support devices, and comprehensive monitoring solutions, all contributing to better care for premature and critically ill newborns. Government initiatives aimed at enhancing perinatal care infrastructure and private sector investments in innovative medical technologies further bolster market prospects.
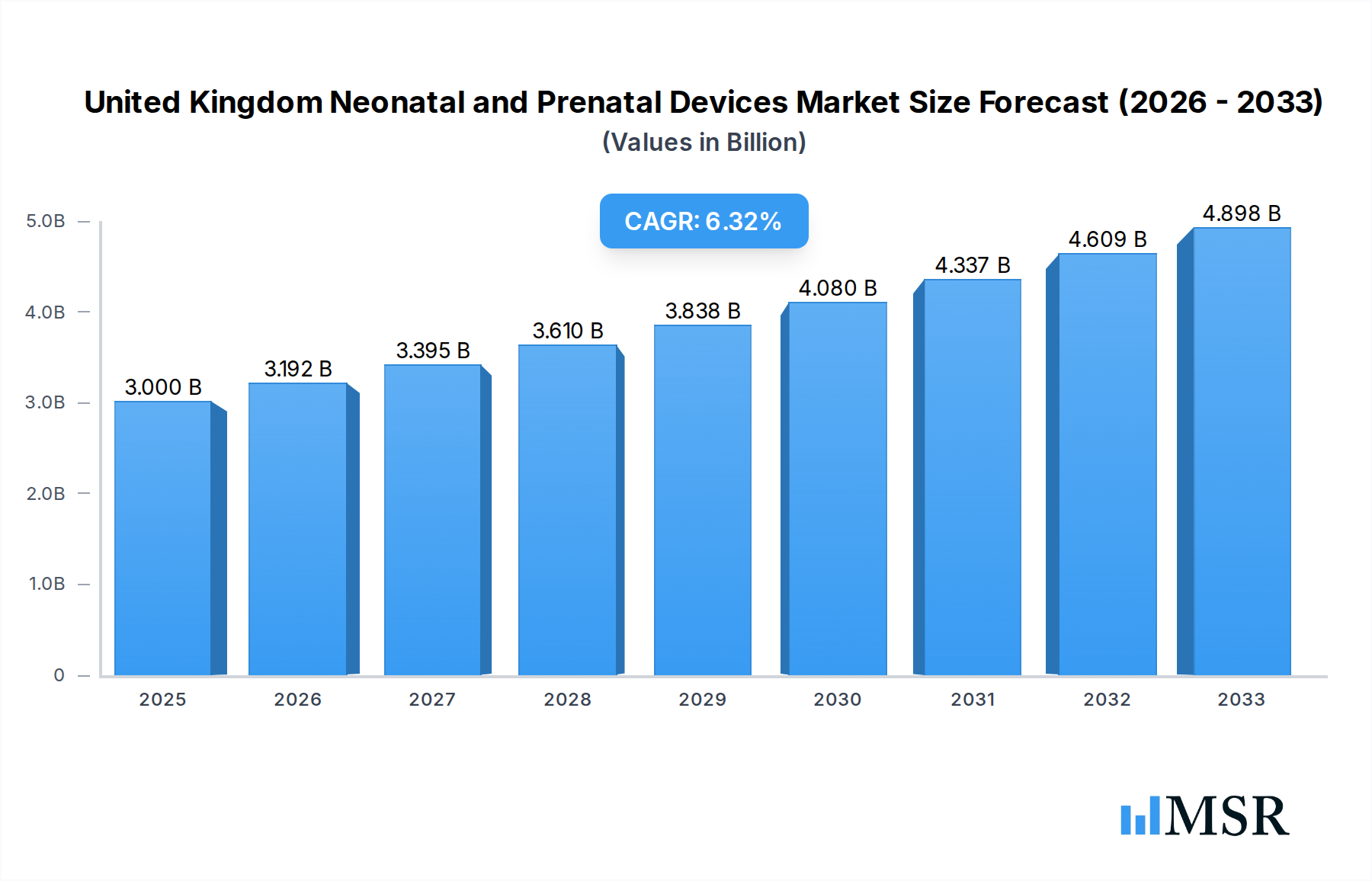
United Kingdom Neonatal and Prenatal Devices Market Market Size (In Billion)

The market's upward trajectory is further supported by evolving healthcare practices, including the move towards integrated care pathways for expectant mothers and newborns. The increasing prevalence of high-risk pregnancies and a greater focus on personalized medicine in both prenatal and neonatal care are creating a demand for specialized and technologically advanced devices. While the market demonstrates strong growth potential, certain factors such as the high cost of advanced equipment and the need for specialized training for healthcare professionals could present localized challenges. However, the overarching trend points towards a dynamic and expanding market, with companies continually innovating to offer solutions that enhance patient safety, improve diagnostic accuracy, and contribute to better long-term health for both mothers and infants in the United Kingdom. The strategic importance of these devices in reducing infant mortality rates and improving overall perinatal health underscores the sustained demand and market vitality.
United Kingdom Neonatal and Prenatal Devices Market Company Market Share

Unlock critical insights into the burgeoning United Kingdom neonatal and prenatal devices market, valued at an estimated $15.8 billion in 2025. This in-depth report, covering the historical period of 2019-2024 and extending to a forecast period of 2025-2033, provides an indispensable resource for manufacturers, distributors, investors, and healthcare providers navigating this dynamic sector. Discover market-leading technologies, emerging trends, and strategic opportunities in prenatal and neonatal care equipment.
United Kingdom Neonatal and Prenatal Devices Market Market Concentration & Dynamics
The United Kingdom neonatal and prenatal devices market exhibits a moderately concentrated landscape, characterized by the presence of both established global players and agile niche innovators. Key companies such as General Electric Company (GE Healthcare), Koninklijke Philips NV, and Medtronic PLC command significant market share through their extensive product portfolios and strong distribution networks. However, the innovation ecosystem is vibrant, fueled by increasing investments in research and development for advanced neonatal monitoring devices and sophisticated ultrasound devices. Regulatory frameworks, primarily governed by the Medicines and Healthcare products Regulatory Agency (MHRA), ensure product safety and efficacy, indirectly influencing market entry and competition. The threat of substitute products is relatively low due to the specialized nature of these medical devices. End-user trends are heavily influenced by the growing demand for non-invasive monitoring, remote patient care solutions, and cost-effective technologies that enhance neonatal survival rates and improve prenatal diagnostic accuracy. Mergers and acquisitions (M&A) activities, while not overtly frequent, play a crucial role in consolidating market share and expanding technological capabilities. For instance, acquisitions of smaller, specialized companies by larger corporations can significantly alter market dynamics. The overall M&A deal count in this segment is expected to remain steady, driven by strategic consolidations to address market gaps and leverage synergistic opportunities.
United Kingdom Neonatal and Prenatal Devices Market Industry Insights & Trends
The United Kingdom neonatal and prenatal devices market is poised for robust growth, driven by a confluence of technological advancements, increasing healthcare expenditure, and evolving demographic trends. The market size is projected to reach an estimated $24.5 billion by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately 5.8% from the base year 2025. Key growth drivers include the rising incidence of preterm births, which necessitates advanced neonatal intensive care unit (NICU) equipment, and the growing awareness surrounding prenatal diagnostics for early detection of fetal abnormalities. Technological disruptions are at the forefront, with continuous innovation in areas like AI-powered neonatal monitoring devices for predictive diagnostics, wearable biosensors for continuous maternal and fetal health tracking, and advanced ultrasound devices offering higher resolution and portability. Evolving consumer behaviors are also shaping the market, with a greater emphasis on personalized medicine, minimally invasive procedures, and home-based monitoring solutions for less critical cases. The demand for integrated healthcare solutions, where prenatal and neonatal devices seamlessly communicate with electronic health records, is also on the rise. Government initiatives focused on improving maternal and child health outcomes, coupled with increased private sector investment in healthcare infrastructure, further bolster market expansion. The development of more affordable and accessible prenatal and fetal equipment is also crucial in catering to a wider segment of the population. The overall sentiment is positive, with the market expected to witness sustained expansion driven by these multifaceted factors.
Key Markets & Segments Leading United Kingdom Neonatal and Prenatal Devices Market
The United Kingdom neonatal and prenatal devices market is dominated by the Neonatal Equipment segment, largely driven by the persistent need for advanced neonatal monitoring devices and critical respiratory assistance and monitoring devices. This dominance is further accentuated by the increasing rates of premature births and congenital conditions requiring intensive care. Within this segment, neonatal monitoring devices hold a substantial market share, encompassing vital signs monitors, oximeters, and neurophysiological monitoring systems. The growing sophistication of these devices, with capabilities like early detection of sepsis and neurological disorders, directly contributes to their market leadership. Furthermore, the respiratory assistance and monitoring devices segment, including ventilators, CPAP machines, and oxygen delivery systems, remains crucial due to the high incidence of respiratory distress syndrome in premature infants.
The Prenatal and Fetal Equipment segment also plays a pivotal role, with ultrasound devices being a significant contributor. Advancements in 3D and 4D ultrasound technology, coupled with the increasing adoption of portable and high-resolution ultrasound machines, are fueling growth in this area. These devices are indispensable for routine prenatal check-ups, anomaly scans, and early detection of fetal health issues. Fetal Doppler and fetal heart monitors also contribute to this segment’s growth, offering non-invasive methods for assessing fetal well-being throughout pregnancy.
Drivers for Neonatal Equipment Dominance:
- Rising Preterm Birth Rates: The UK, like many developed nations, faces a notable rate of preterm births, necessitating intensive neonatal care.
- Technological Advancements: Continuous innovation in monitoring and life support systems enhances infant survival rates and quality of life.
- Government Focus on Child Health: National health strategies prioritizing maternal and child welfare drive investment in neonatal care infrastructure and equipment.
- Increasing NICU Capacity: Expansion and upgrade of Neonatal Intensive Care Units (NICUs) across the UK directly translates to higher demand for specialized equipment.
Drivers for Prenatal and Fetal Equipment Growth:
- Early Detection and Diagnosis: Advanced prenatal tools enable early identification of potential complications, allowing for timely intervention and improved outcomes.
- Growing Maternal Age: An increasing trend of delayed childbearing can lead to higher-risk pregnancies, necessitating more frequent and sophisticated prenatal monitoring.
- Enhanced Diagnostic Accuracy: Improvements in imaging technologies like ultrasound provide clearer visualizations and more accurate diagnoses.
- Preventive Healthcare Emphasis: A greater focus on preventive healthcare measures during pregnancy contributes to the sustained demand for prenatal diagnostic tools.
United Kingdom Neonatal and Prenatal Devices Market Product Developments
The United Kingdom neonatal and prenatal devices market is witnessing rapid product innovation aimed at enhancing patient outcomes, improving clinician workflow, and reducing healthcare costs. Key advancements include the development of AI-powered neonatal monitoring devices capable of predicting adverse events like sepsis and apnea with greater accuracy. Furthermore, miniaturization and increased portability of devices like fetal dopplers and compact ultrasound devices are enabling their use in a wider range of clinical settings, including remote and home-care environments. Innovations in respiratory assistance and monitoring devices are focusing on less invasive ventilation strategies and smart alarms to reduce patient discomfort and clinician burden. These product developments are crucial in maintaining a competitive edge and addressing the evolving needs of the UK's healthcare system.
Challenges in the United Kingdom Neonatal and Prenatal Devices Market Market
Despite robust growth, the United Kingdom neonatal and prenatal devices market faces several challenges. High upfront costs associated with advanced neonatal intensive care unit (NICU) equipment and sophisticated prenatal and fetal equipment can be a barrier for smaller healthcare facilities. Stringent regulatory approval processes, while ensuring safety, can also lead to extended market entry timelines. Furthermore, supply chain disruptions, as evidenced in recent global events, can impact the availability and pricing of critical components and finished devices. Competitive pressures from both established players and new entrants also necessitate continuous innovation and cost optimization strategies.
Forces Driving United Kingdom Neonatal and Prenatal Devices Market Growth
Several forces are propelling the growth of the United Kingdom neonatal and prenatal devices market. Technological advancements are paramount, with continuous innovation in neonatal monitoring devices, ultrasound devices, and respiratory assistance and monitoring devices offering enhanced diagnostic capabilities and treatment efficacy. Increasing government initiatives and funding aimed at improving maternal and child health outcomes provide a strong impetus for market expansion. The rising incidence of preterm births and the growing awareness of the importance of early prenatal diagnostics further fuel demand for specialized equipment. Additionally, the expanding healthcare infrastructure and the increasing adoption of advanced medical technologies by healthcare providers are significant growth catalysts.
Challenges in the United Kingdom Neonatal and Prenatal Devices Market Market
Long-term growth in the United Kingdom neonatal and prenatal devices market will be significantly influenced by sustained investment in research and development of next-generation technologies. The increasing demand for integrated and connected healthcare solutions, where devices communicate seamlessly with electronic health records and support remote patient monitoring, presents a significant growth catalyst. Partnerships and collaborations between medical device manufacturers, healthcare providers, and academic institutions will be crucial for fostering innovation and accelerating the adoption of new products. Furthermore, market expansion into underserved regions within the UK and the development of more cost-effective solutions will be key to unlocking future growth potential.
Emerging Opportunities in United Kingdom Neonatal and Prenatal Devices Market
Emerging opportunities within the United Kingdom neonatal and prenatal devices market are abundant, driven by evolving healthcare paradigms. The burgeoning field of telehealth and remote patient monitoring presents a significant avenue for the deployment of advanced neonatal monitoring devices and portable prenatal and fetal equipment, allowing for continuous care outside traditional hospital settings. Artificial intelligence (AI) integration into diagnostic tools, such as AI-enhanced ultrasound devices for more accurate fetal anomaly detection, is a key trend. The growing focus on personalized medicine and the development of tailored treatment plans for high-risk pregnancies and neonates also opens new market segments. Furthermore, the increasing demand for user-friendly and intuitive devices for both clinicians and parents presents an opportunity for intuitive design and enhanced user experience.
Leading Players in the United Kingdom Neonatal and Prenatal Devices Market Sector
- Archimed SAS (Natus Medical Incorporated)
- Masimo Corporation
- Utah Medical Products Inc
- Ambu A/S
- Cardinal Health Inc
- Medtronic PLC
- Vyaire Medical Inc
- Phoenix Medical Systems Ltd
- Carl Bennet AB (Getinge AB)
- General Electric Company (GE Healthcare)
- Koninklijke Philips NV
- Dragerwerk AG & Co KGaA
Key Milestones in United Kingdom Neonatal and Prenatal Devices Market Industry
- July 2022: Maternova Inc. signed an agreement with BirthTech Lda to distribute its Preemie Test globally, including the United Kingdom. This test accurately assesses newborn gestational age, a critical factor for neonatal survival and protection of preterm newborns.
- April 2022: Embrace conducted a clinical trial for its Neonatal MRI system in 207 infants. This device has received a CE mark and is slated for availability in European countries, including the United Kingdom, marking a significant advancement in neonatal imaging capabilities.
Strategic Outlook for United Kingdom Neonatal and Prenatal Devices Market Market
The strategic outlook for the United Kingdom neonatal and prenatal devices market is highly positive, driven by ongoing technological advancements and a persistent demand for improved maternal and infant healthcare. Future growth will be accelerated by the continued integration of AI and machine learning into diagnostic and monitoring devices, enhancing predictive capabilities and clinical decision-making. The expansion of telehealth and remote monitoring solutions presents a significant opportunity to extend the reach of specialized neonatal and prenatal care. Strategic collaborations between industry players and healthcare institutions will be crucial for the development and adoption of innovative, cost-effective solutions. Furthermore, a focus on user-centric design and the development of interoperable systems will be key to navigating the evolving healthcare landscape and securing market leadership in the coming years.
United Kingdom Neonatal and Prenatal Devices Market Segmentation
-
1. Product Type
-
1.1. Prenatal and Fetal Equipment
- 1.1.1. Ultrasound Devices
- 1.1.2. Fetal Doppler
- 1.1.3. Fetal Heart Monitors
- 1.1.4. Other Prenatal and Fetal Equipment
-
1.2. Neonatal Equipment
- 1.2.1. Incubators
- 1.2.2. Neonatal Monitoring Devices
- 1.2.3. Respiratory Assistance and Monitoring Devices
- 1.2.4. Other Neonatal Care Equipment
- 1.3. Other Product Types
-
1.1. Prenatal and Fetal Equipment
United Kingdom Neonatal and Prenatal Devices Market Segmentation By Geography
- 1. United Kingdom
United Kingdom Neonatal and Prenatal Devices Market Regional Market Share

Geographic Coverage of United Kingdom Neonatal and Prenatal Devices Market
United Kingdom Neonatal and Prenatal Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. MSR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 5.1.1. Prenatal and Fetal Equipment
- 5.1.1.1. Ultrasound Devices
- 5.1.1.2. Fetal Doppler
- 5.1.1.3. Fetal Heart Monitors
- 5.1.1.4. Other Prenatal and Fetal Equipment
- 5.1.2. Neonatal Equipment
- 5.1.2.1. Incubators
- 5.1.2.2. Neonatal Monitoring Devices
- 5.1.2.3. Respiratory Assistance and Monitoring Devices
- 5.1.2.4. Other Neonatal Care Equipment
- 5.1.3. Other Product Types
- 5.1.1. Prenatal and Fetal Equipment
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. United Kingdom
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 6. United Kingdom Neonatal and Prenatal Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 6.1.1. Prenatal and Fetal Equipment
- 6.1.1.1. Ultrasound Devices
- 6.1.1.2. Fetal Doppler
- 6.1.1.3. Fetal Heart Monitors
- 6.1.1.4. Other Prenatal and Fetal Equipment
- 6.1.2. Neonatal Equipment
- 6.1.2.1. Incubators
- 6.1.2.2. Neonatal Monitoring Devices
- 6.1.2.3. Respiratory Assistance and Monitoring Devices
- 6.1.2.4. Other Neonatal Care Equipment
- 6.1.3. Other Product Types
- 6.1.1. Prenatal and Fetal Equipment
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 Archimed SAS (Natus Medical Incorporated)
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 Masimo Corporation
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 Utah Medical Products Inc
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 Ambu A/S
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Cardinal Health Inc
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Medtronic PLC
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 Vyaire Medical Inc
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 Phoenix Medical Systems Ltd
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.9 Carl Bennet AB (Getinge AB)
- 7.1.9.1. Company Overview
- 7.1.9.2. Products
- 7.1.9.3. Company Financials
- 7.1.9.4. SWOT Analysis
- 7.1.10 General Electric Company (GE Healthcare)
- 7.1.10.1. Company Overview
- 7.1.10.2. Products
- 7.1.10.3. Company Financials
- 7.1.10.4. SWOT Analysis
- 7.1.11 Koninklijke Philips NV
- 7.1.11.1. Company Overview
- 7.1.11.2. Products
- 7.1.11.3. Company Financials
- 7.1.11.4. SWOT Analysis
- 7.1.12 Dragerwerk AG & Co KGaA
- 7.1.12.1. Company Overview
- 7.1.12.2. Products
- 7.1.12.3. Company Financials
- 7.1.12.4. SWOT Analysis
- 7.1.1 Archimed SAS (Natus Medical Incorporated)
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: United Kingdom Neonatal and Prenatal Devices Market Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: United Kingdom Neonatal and Prenatal Devices Market Share (%) by Company 2025
List of Tables
- Table 1: United Kingdom Neonatal and Prenatal Devices Market Revenue billion Forecast, by Product Type 2020 & 2033
- Table 2: United Kingdom Neonatal and Prenatal Devices Market Volume K Units Forecast, by Product Type 2020 & 2033
- Table 3: United Kingdom Neonatal and Prenatal Devices Market Revenue billion Forecast, by Region 2020 & 2033
- Table 4: United Kingdom Neonatal and Prenatal Devices Market Volume K Units Forecast, by Region 2020 & 2033
- Table 5: United Kingdom Neonatal and Prenatal Devices Market Revenue billion Forecast, by Product Type 2020 & 2033
- Table 6: United Kingdom Neonatal and Prenatal Devices Market Volume K Units Forecast, by Product Type 2020 & 2033
- Table 7: United Kingdom Neonatal and Prenatal Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 8: United Kingdom Neonatal and Prenatal Devices Market Volume K Units Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the United Kingdom Neonatal and Prenatal Devices Market?
The projected CAGR is approximately 6.4%.
2. Which companies are prominent players in the United Kingdom Neonatal and Prenatal Devices Market?
Key companies in the market include Archimed SAS (Natus Medical Incorporated), Masimo Corporation, Utah Medical Products Inc, Ambu A/S, Cardinal Health Inc, Medtronic PLC, Vyaire Medical Inc, Phoenix Medical Systems Ltd, Carl Bennet AB (Getinge AB), General Electric Company (GE Healthcare), Koninklijke Philips NV, Dragerwerk AG & Co KGaA.
3. What are the main segments of the United Kingdom Neonatal and Prenatal Devices Market?
The market segments include Product Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 3 billion as of 2022.
5. What are some drivers contributing to market growth?
Growing Burden of Preterm Births; Increasing Awareness for Prenatal and Neonatal Care.
6. What are the notable trends driving market growth?
Incubators Segment is Expected to Witness a Significant Growth Over the Forecast Period..
7. Are there any restraints impacting market growth?
Low Birth Rates.
8. Can you provide examples of recent developments in the market?
In July 2022, Maternova Inc. reported that they signed an agreement with BirthTech Lda to distribute its Preemie Test in multiple geographies worldwide, including the United Kingdom. The Preemie Test is a medical device clinically proven to accurately assess the gestational age of a newborn, which is the major marker of neonatal survival and protects preterm newborns.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K Units.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "United Kingdom Neonatal and Prenatal Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the United Kingdom Neonatal and Prenatal Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the United Kingdom Neonatal and Prenatal Devices Market?
To stay informed about further developments, trends, and reports in the United Kingdom Neonatal and Prenatal Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
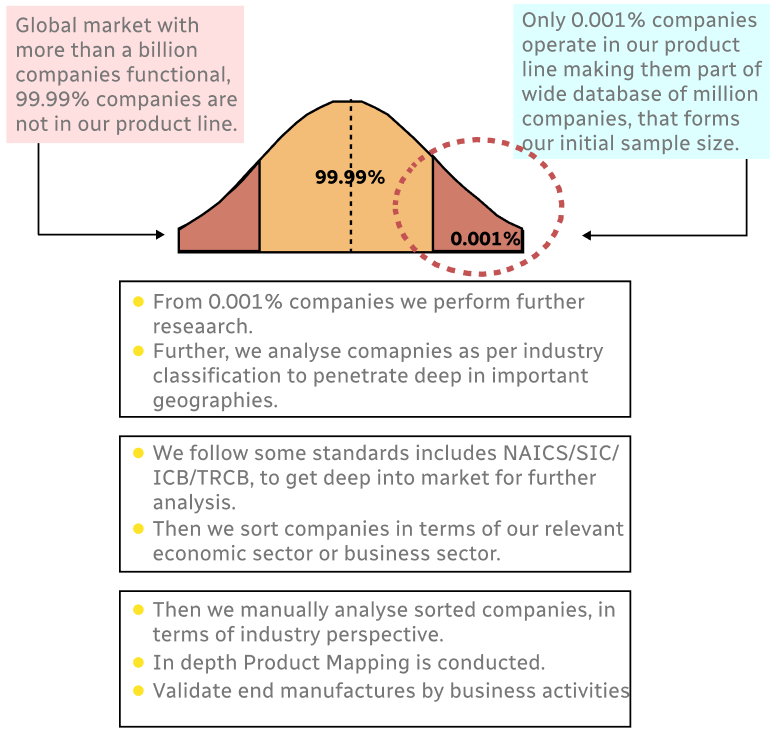
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
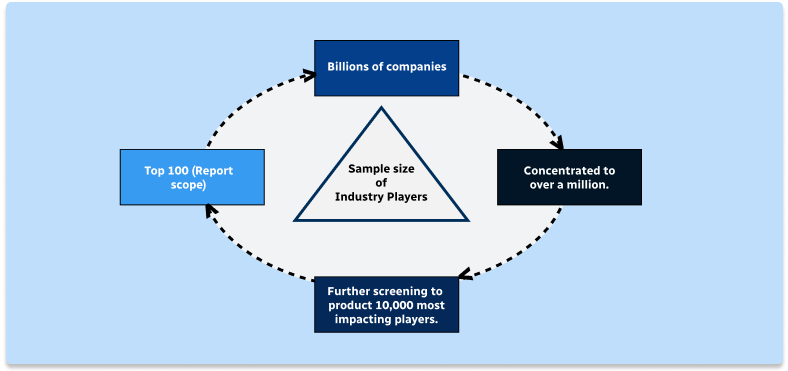
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
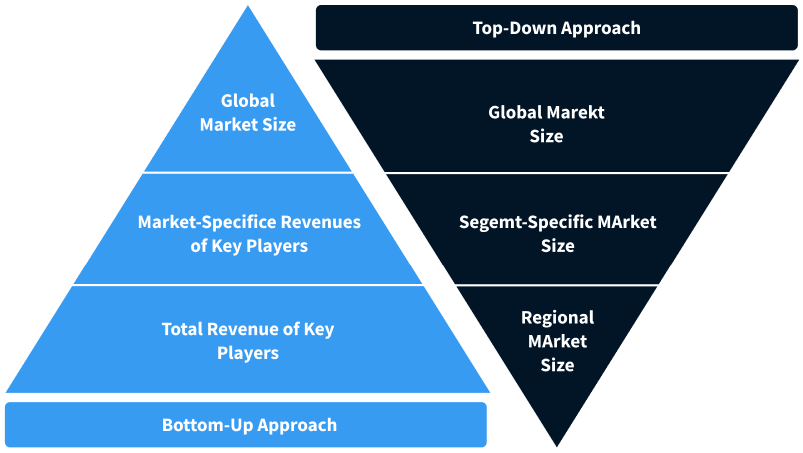
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


