Key Insights
The Manual Incentive Spirometer market is poised for significant expansion, projected to reach USD 1377.7 million by 2025, driven by a robust CAGR of 9.5% through 2033. This growth trajectory is fueled by an increasing prevalence of respiratory diseases, a growing aging population, and a greater emphasis on post-operative respiratory care. The rising awareness among healthcare providers and patients regarding the benefits of incentive spirometry in preventing and managing lung conditions, such as pneumonia and atelectasis, is a primary catalyst. Furthermore, the expanding healthcare infrastructure in emerging economies and the increasing adoption of homecare solutions for chronic respiratory ailments are expected to contribute substantially to market expansion. The demand for hand-held spirometers, favored for their portability and ease of use in various settings including homecare, is particularly strong.
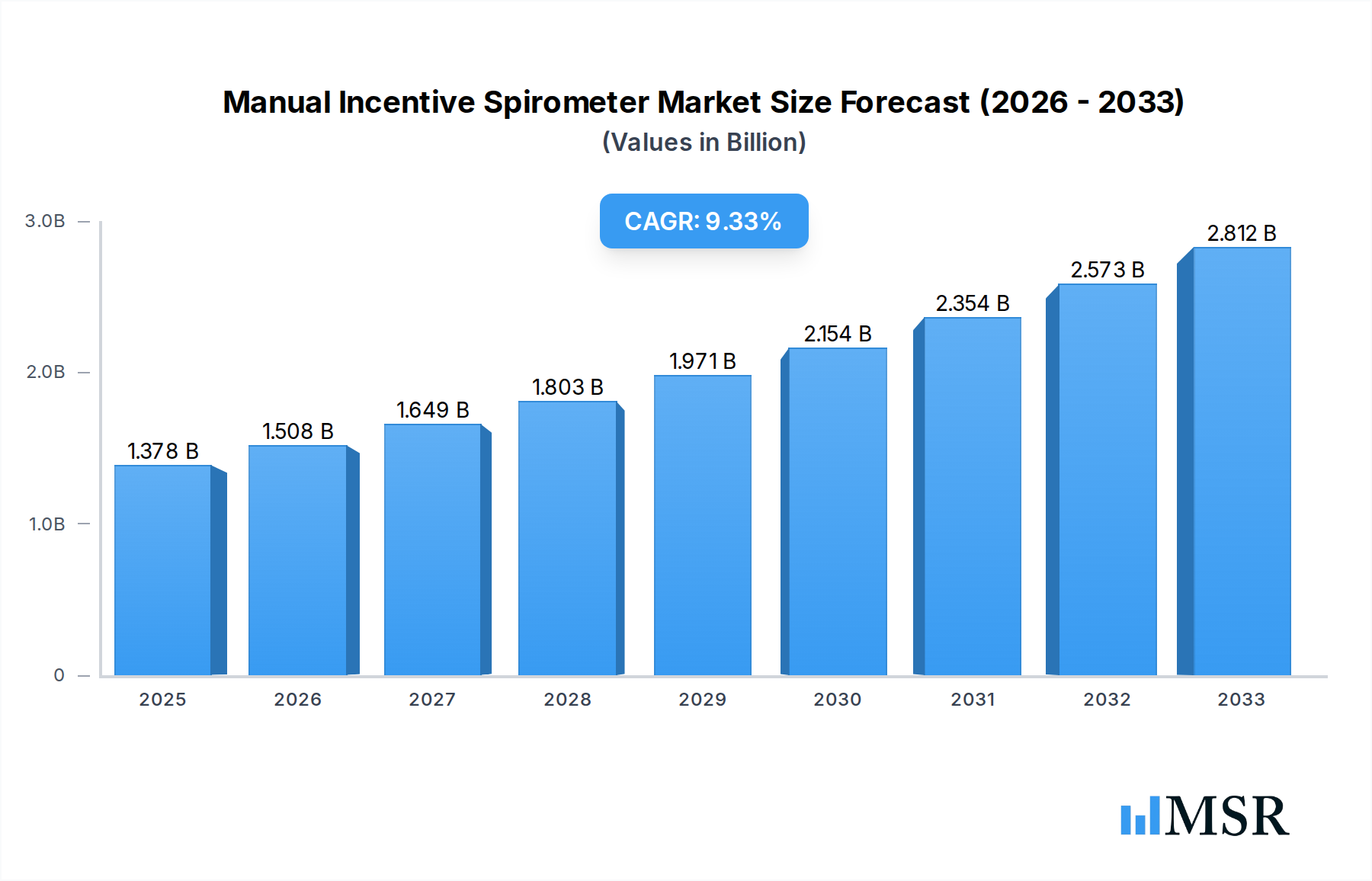
Manual Incentive Spirometer Market Size (In Billion)

The market landscape is characterized by a competitive environment with established players like BD (CareFusion), Schiller, and Welch Allyn, alongside emerging companies focusing on technological advancements and cost-effective solutions. While the market is robust, potential restraints include stringent regulatory approvals for new devices and reimbursement challenges in certain healthcare systems. However, these are being offset by innovations leading to user-friendly designs, integration with digital health platforms, and a growing focus on pulmonary rehabilitation programs. The Asia Pacific region is anticipated to witness the fastest growth due to its large population, increasing healthcare expenditure, and rising incidence of respiratory illnesses, while North America and Europe are expected to maintain their dominance due to advanced healthcare infrastructure and high adoption rates of respiratory care devices.
Manual Incentive Spirometer Company Market Share

Here is the SEO-optimized report description for the Manual Incentive Spirometer market.
Unlock the Future of Respiratory Care: Comprehensive Manual Incentive Spirometer Market Report (2019-2033)
Gain unparalleled insights into the dynamic Manual Incentive Spirometer market with this in-depth report. Covering the historical period of 2019-2024, base and estimated year of 2025, and a robust forecast period extending to 2033, this comprehensive analysis is your definitive guide to understanding market concentration, key trends, competitive landscapes, and future growth trajectories. Essential for respiratory therapists, pulmonologists, device manufacturers, distributors, investors, and healthcare administrators, this report provides actionable intelligence to navigate the evolving respiratory health sector.
Manual Incentive Spirometer Market Concentration & Dynamics
The global Manual Incentive Spirometer market is characterized by a moderate concentration, with a few key players holding significant market share, estimated to be over $500 million in the base year. Innovation ecosystems are actively developing, driven by advancements in materials science and user-interface design, leading to an increased number of patent filings by leading companies like BD (CareFusion), Schiller, and Welch Allyn. Regulatory frameworks, primarily governed by bodies like the FDA in the US and CE marking in Europe, ensure product safety and efficacy, impacting market entry for new manufacturers such as MGC and Futuremed. The threat of substitute products, while present from advanced digital spirometers, remains limited due to the cost-effectiveness and simplicity of manual devices in many clinical and homecare settings. End-user trends highlight a growing preference for portable and easy-to-use devices, particularly in homecare segments, pushing companies like MIR and Vitalograph to refine their offerings. Mergers and acquisitions (M&A) activity, with an estimated 10-15 deals in the past five years valued at over $20 million collectively, indicate consolidation and strategic expansion by larger entities, including CHEST. MI. and SDI Diagnostics, to broaden their product portfolios and market reach.
- Market Share Dominance: Leading players like BD (CareFusion) and CHEST. MI. command a combined market share of approximately 35%.
- Innovation Focus: Increased R&D investment in ergonomic designs and improved airflow indicators.
- Regulatory Impact: Stringent approval processes continue to shape market dynamics for new entrants.
- Substitute Threat: Low to moderate, primarily in specialized clinical settings.
- End-User Preference: Emphasis on simplicity, affordability, and portability for patient adherence.
- M&A Activity: Driven by the desire for expanded product lines and market penetration, with an average of 2-3 significant deals annually.
Manual Incentive Spirometer Industry Insights & Trends
The Manual Incentive Spirometer industry is poised for significant growth, projected to expand from an estimated market size of over $1,000 million in the base year 2025 to over $1,800 million by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately 7.5%. This growth is propelled by a confluence of factors including the increasing prevalence of respiratory diseases such as COPD, asthma, and post-operative respiratory complications, which necessitate consistent pulmonary rehabilitation. Technological disruptions, while more pronounced in digital spirometry, are influencing manual device design through enhanced material durability and improved visual feedback mechanisms, benefiting manufacturers like Welch Allyn and Fukuda Sangyo. Evolving consumer behaviors, particularly the rising trend of self-management of chronic conditions and the growing adoption of homecare solutions, are creating substantial demand for user-friendly and cost-effective respiratory aids. The aging global population is also a significant contributor, as older individuals are more susceptible to respiratory ailments and often prefer simpler, non-intrusive medical devices. Furthermore, growing awareness campaigns by healthcare organizations and government initiatives promoting respiratory health education are further bolstering market penetration, especially in emerging economies where access to advanced medical equipment may be limited. The emphasis on preventative healthcare and early intervention also plays a crucial role, as incentive spirometers are recommended for patients at risk of developing respiratory issues or as part of post-surgical recovery protocols. The development of integrated educational materials and patient support programs by companies like Vitalograph and NDD is also enhancing user engagement and compliance, thereby driving market expansion. The consistent demand for these devices in hospitals and clinics for pre- and post-operative care, alongside their increasing use in homecare settings for long-term management, solidifies their crucial role in the respiratory care ecosystem. The affordability and ease of use of manual incentive spirometers ensure their continued relevance and adoption across diverse healthcare settings and patient demographics, contributing to a stable and upward market trajectory.
Key Markets & Segments Leading Manual Incentive Spirometer
The Hospital segment, projected to account for over 50% of the total market revenue by 2033, is the dominant force in the Manual Incentive Spirometer market. This leadership is driven by its widespread use in pre- and post-operative care to prevent atelectasis and improve lung function, a critical component of patient recovery protocols in surgical wards. The sheer volume of surgical procedures performed globally, estimated at over 300 million annually, directly translates into a consistent demand for these devices. Within the hospital segment, Hand-held Spirometers are the most prevalent type, estimated to capture over 60% of the segment's market share. Their portability, ease of use for both patients and clinicians, and cost-effectiveness make them ideal for widespread deployment across various hospital departments.
- Drivers in the Hospital Segment:
- High Volume of Surgical Procedures: Essential for post-operative pulmonary rehabilitation.
- Prevalence of Respiratory Illnesses: Used for diagnosis and monitoring of conditions like pneumonia.
- Cost-Effectiveness: Lower acquisition and maintenance costs compared to advanced digital alternatives.
- Clinical Guidelines & Protocols: Mandated use in many recovery pathways.
The Clinic segment represents the second-largest application, with a projected market share of approximately 30% by 2033. Clinics utilize manual incentive spirometers for routine respiratory assessments, patient education on lung exercises, and long-term management of chronic respiratory conditions. The growing number of outpatient respiratory care facilities and pulmonology clinics globally contributes to this segment's expansion. Here, Table-top Spirometers also find significant application due to their slightly larger size allowing for more precise measurements in a stable clinical environment.
- Drivers in the Clinic Segment:
- Chronic Disease Management: Crucial for ongoing pulmonary therapy for asthma and COPD patients.
- Patient Education & Training: Facilitates learning of proper breathing techniques.
- Accessibility: Readily available in community health centers and private practices.
The Homecare segment, though smaller at approximately 20% market share, is experiencing the fastest growth, projected at a CAGR of over 8%. This surge is fueled by the increasing shift towards decentralized healthcare, aging populations requiring continuous monitoring, and the desire for patient autonomy in managing their health. Hand-held Spirometers are overwhelmingly preferred in this segment due to their portability, simplicity, and affordability, enabling patients to continue their therapy independently at home.
- Drivers in the Homecare Segment:
- Aging Population Demographics: Increased incidence of respiratory issues in elderly individuals.
- Telehealth Adoption: Integration with remote patient monitoring systems.
- Patient Empowerment: Desire for self-management of chronic conditions.
- Healthcare Cost Containment: Shifting care from hospitals to home settings.
Geographically, North America and Europe currently dominate the market due to advanced healthcare infrastructure and high awareness of respiratory health. However, the Asia-Pacific region is emerging as a high-growth market, driven by rising disposable incomes, increasing healthcare expenditure, and a growing prevalence of respiratory diseases.
Manual Incentive Spirometer Product Developments
Recent product developments in the Manual Incentive Spirometer market focus on enhancing user experience and clinical utility. Innovations include the integration of clearer visual indicators for inhalation depth, improved mouthpiece designs for better seal and comfort, and the use of more durable, medical-grade plastics. Companies like CHEST. MI. and SDI Diagnostics are introducing models with enhanced airflow chambers for more consistent performance across varying user efforts. Furthermore, some manufacturers are exploring biodegradable materials for greater environmental sustainability. These advancements aim to improve patient compliance, provide more reliable feedback for therapists, and ensure the long-term effectiveness of pulmonary exercises, thereby offering a competitive edge in an evolving market.
Challenges in the Manual Incentive Spirometer Market
The Manual Incentive Spirometer market faces several challenges that could impede its growth. Regulatory hurdles, including stringent approval processes in certain regions, can delay product launches and increase development costs, impacting smaller manufacturers like Medikro. Supply chain disruptions, as witnessed in recent global events, can lead to material shortages and increased production costs, affecting the availability and pricing of essential components. Intense competitive pressure from digital spirometers, which offer advanced data analytics and connectivity, poses a threat, particularly in sophisticated healthcare settings. Additionally, lack of widespread patient education on the proper use and benefits of incentive spirometry in some developing regions can limit market penetration. The overall impact of these challenges could lead to a slowdown in market expansion by an estimated 1-2% if not adequately addressed.
Forces Driving Manual Incentive Spirometer Growth
Several forces are driving the sustained growth of the Manual Incentive Spirometer market. Technological advancements in material science and ergonomic design are making devices more user-friendly and durable, benefiting companies like Futuremed and Anhui Electronics Scientific Institute. Economic factors, such as increasing healthcare expenditure globally and the rising disposable incomes in emerging economies, are enhancing affordability and accessibility. Regulatory support and public health initiatives promoting respiratory health awareness and preventative care are creating a more favorable market environment. The growing prevalence of chronic respiratory diseases like COPD and asthma, coupled with an aging global population, continues to be a primary demand driver, ensuring a consistent need for these essential devices.
Challenges in the Manual Incentive Spirometer Market
Long-term growth catalysts for the Manual Incentive Spirometer market lie in continuous innovation and strategic market expansion. The development of spirometers with improved patient engagement features, such as gamified elements or integration with simple tracking apps, can foster better adherence. Partnerships between device manufacturers and healthcare providers, including hospitals and respiratory therapy clinics, can lead to wider adoption and integrated care pathways. Expanding market reach into underserved regions with growing healthcare needs, through targeted distribution strategies and localized marketing efforts by companies like Contec and Geratherm, presents significant untapped potential. Furthermore, focusing on cost-effective solutions that cater to both institutional and individual patient needs will solidify their position in the evolving healthcare landscape.
Emerging Opportunities in Manual Incentive Spirometer
Emerging opportunities in the Manual Incentive Spirometer market are abundant, driven by new trends and unmet needs. The burgeoning homecare segment, fueled by telehealth adoption and a preference for remote patient management, offers a substantial growth avenue for portable and user-friendly devices. The development of integrated digital features that complement manual spirometry, such as basic Bluetooth connectivity for data logging without the full complexity of digital spirometers, could bridge the gap between traditional and advanced devices. Furthermore, exploring emerging markets in Asia, Africa, and Latin America, where healthcare infrastructure is developing and the demand for affordable respiratory solutions is high, presents significant untapped potential for manufacturers like Fukuda Sangyo and SDI Diagnostics. Increased focus on preventative respiratory health and early intervention strategies in schools and workplaces also opens new avenues for market penetration.
Leading Players in the Manual Incentive Spirometer Sector
- BD (CareFusion)
- Schiller
- Welch Allyn
- CHEST. MI.
- MIR
- Vitalograph
- MGC
- Futuremed
- Fukuda Sangyo
- NDD
- SDI Diagnostics
- Geratherm
- Cosmed
- Medikro
- Anhui Electronics Scientific Institute
- Contec
Key Milestones in Manual Incentive Spirometer Industry
- 2019: Introduction of advanced airflow resistance mechanisms in select models.
- 2020: Increased demand due to heightened awareness of respiratory health post-pandemic.
- 2021: Emergence of eco-friendly material options in some product lines.
- 2022: Significant investment in R&D for improved patient adherence features.
- 2023: Growing adoption in homecare settings driven by telehealth trends.
- 2024: Expansion of distribution networks into emerging markets.
Strategic Outlook for Manual Incentive Spirometer Market
The strategic outlook for the Manual Incentive Spirometer market is one of steady and promising growth, driven by an enduring need for accessible and effective respiratory rehabilitation. Key growth accelerators include the continued expansion of the homecare sector, leveraging advancements in telehealth and remote monitoring. Strategic partnerships between manufacturers and healthcare institutions will be crucial for integrating these devices into comprehensive patient care pathways. Furthermore, capitalizing on the expanding healthcare infrastructure and increasing disposable incomes in emerging economies presents a significant opportunity for market penetration. By focusing on product innovation that enhances user experience, affordability, and durability, while actively engaging in market education and expanding distribution channels, the Manual Incentive Spirometer market is well-positioned for sustained success in the coming years.
Manual Incentive Spirometer Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Clinic
- 1.3. Homecare
-
2. Types
- 2.1. Hand-held Spirometer
- 2.2. Table-top Spirometer
- 2.3. Desktop (PC) Spirometer
Manual Incentive Spirometer Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
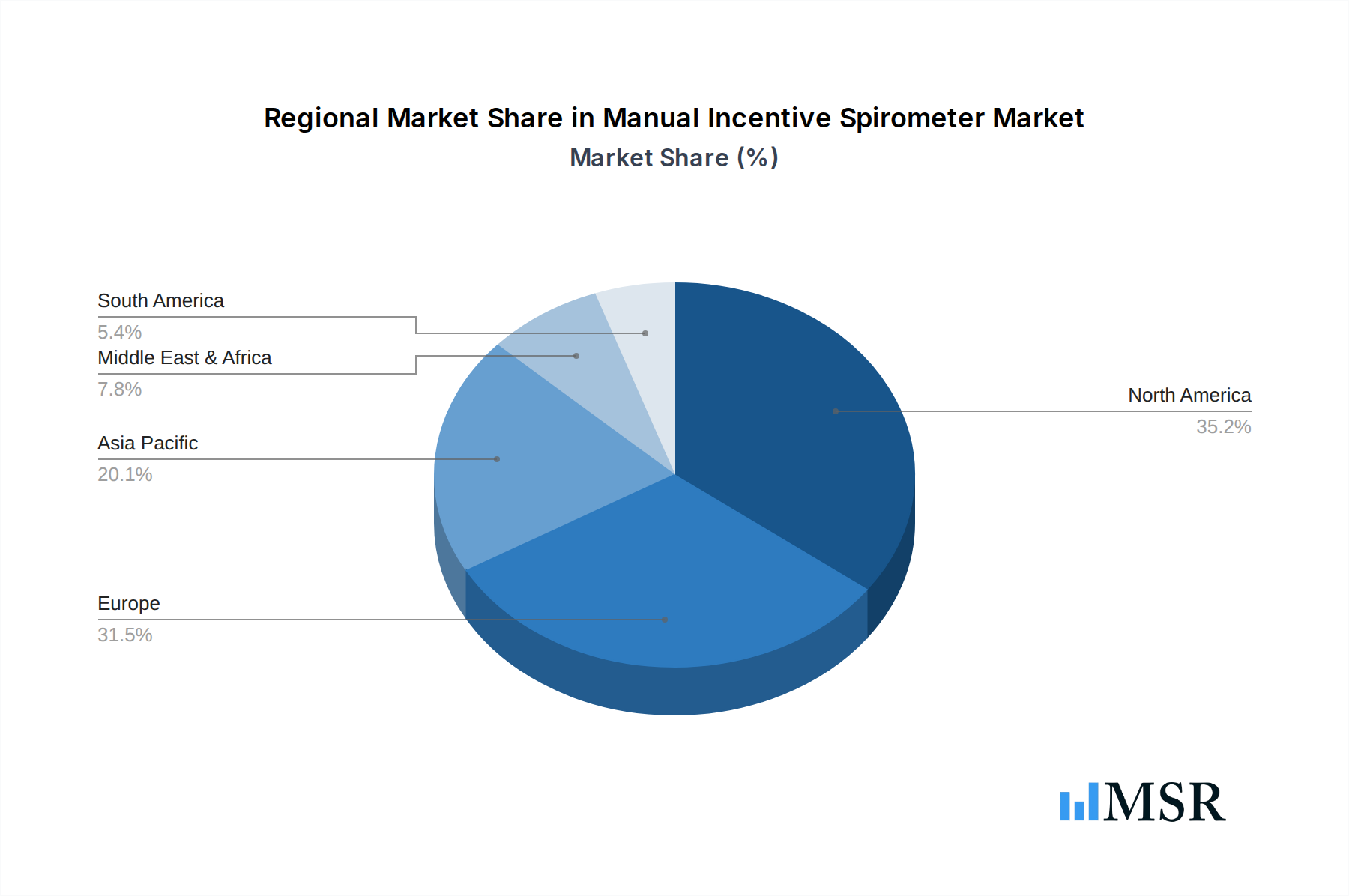
Manual Incentive Spirometer Regional Market Share

Geographic Coverage of Manual Incentive Spirometer
Manual Incentive Spirometer REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.5% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Manual Incentive Spirometer Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Clinic
- 5.1.3. Homecare
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Hand-held Spirometer
- 5.2.2. Table-top Spirometer
- 5.2.3. Desktop (PC) Spirometer
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Manual Incentive Spirometer Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Clinic
- 6.1.3. Homecare
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Hand-held Spirometer
- 6.2.2. Table-top Spirometer
- 6.2.3. Desktop (PC) Spirometer
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Manual Incentive Spirometer Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Clinic
- 7.1.3. Homecare
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Hand-held Spirometer
- 7.2.2. Table-top Spirometer
- 7.2.3. Desktop (PC) Spirometer
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Manual Incentive Spirometer Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Clinic
- 8.1.3. Homecare
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Hand-held Spirometer
- 8.2.2. Table-top Spirometer
- 8.2.3. Desktop (PC) Spirometer
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Manual Incentive Spirometer Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Clinic
- 9.1.3. Homecare
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Hand-held Spirometer
- 9.2.2. Table-top Spirometer
- 9.2.3. Desktop (PC) Spirometer
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Manual Incentive Spirometer Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Clinic
- 10.1.3. Homecare
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Hand-held Spirometer
- 10.2.2. Table-top Spirometer
- 10.2.3. Desktop (PC) Spirometer
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 BD (CareFusion)
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Schiller
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Welch Allyn
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 CHEST. MI.
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 MIR
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Vitalograph
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 MGC
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Futuremed
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Fukuda Sangyo
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 NDD
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 SDI Diagnostics
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Geratherm
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Cosmed
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Medikro
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Anhui Electronics Scientific Institute
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Contec
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.1 BD (CareFusion)
List of Figures
- Figure 1: Global Manual Incentive Spirometer Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Manual Incentive Spirometer Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Manual Incentive Spirometer Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Manual Incentive Spirometer Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Manual Incentive Spirometer Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Manual Incentive Spirometer Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Manual Incentive Spirometer Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Manual Incentive Spirometer Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Manual Incentive Spirometer Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Manual Incentive Spirometer Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Manual Incentive Spirometer Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Manual Incentive Spirometer Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Manual Incentive Spirometer Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Manual Incentive Spirometer Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Manual Incentive Spirometer Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Manual Incentive Spirometer Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Manual Incentive Spirometer Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Manual Incentive Spirometer Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Manual Incentive Spirometer Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Manual Incentive Spirometer Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Manual Incentive Spirometer Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Manual Incentive Spirometer Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Manual Incentive Spirometer Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Manual Incentive Spirometer Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Manual Incentive Spirometer Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Manual Incentive Spirometer Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Manual Incentive Spirometer Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Manual Incentive Spirometer Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Manual Incentive Spirometer Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Manual Incentive Spirometer Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Manual Incentive Spirometer Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Manual Incentive Spirometer Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Manual Incentive Spirometer Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Manual Incentive Spirometer Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Manual Incentive Spirometer Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Manual Incentive Spirometer Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Manual Incentive Spirometer Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Manual Incentive Spirometer Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Manual Incentive Spirometer Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Manual Incentive Spirometer Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Manual Incentive Spirometer Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Manual Incentive Spirometer Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Manual Incentive Spirometer Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Manual Incentive Spirometer Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Manual Incentive Spirometer Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Manual Incentive Spirometer Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Manual Incentive Spirometer Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Manual Incentive Spirometer Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Manual Incentive Spirometer Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Manual Incentive Spirometer Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Manual Incentive Spirometer?
The projected CAGR is approximately 9.5%.
2. Which companies are prominent players in the Manual Incentive Spirometer?
Key companies in the market include BD (CareFusion), Schiller, Welch Allyn, CHEST. MI., MIR, Vitalograph, MGC, Futuremed, Fukuda Sangyo, NDD, SDI Diagnostics, Geratherm, Cosmed, Medikro, Anhui Electronics Scientific Institute, Contec.
3. What are the main segments of the Manual Incentive Spirometer?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Manual Incentive Spirometer," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Manual Incentive Spirometer report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Manual Incentive Spirometer?
To stay informed about further developments, trends, and reports in the Manual Incentive Spirometer, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
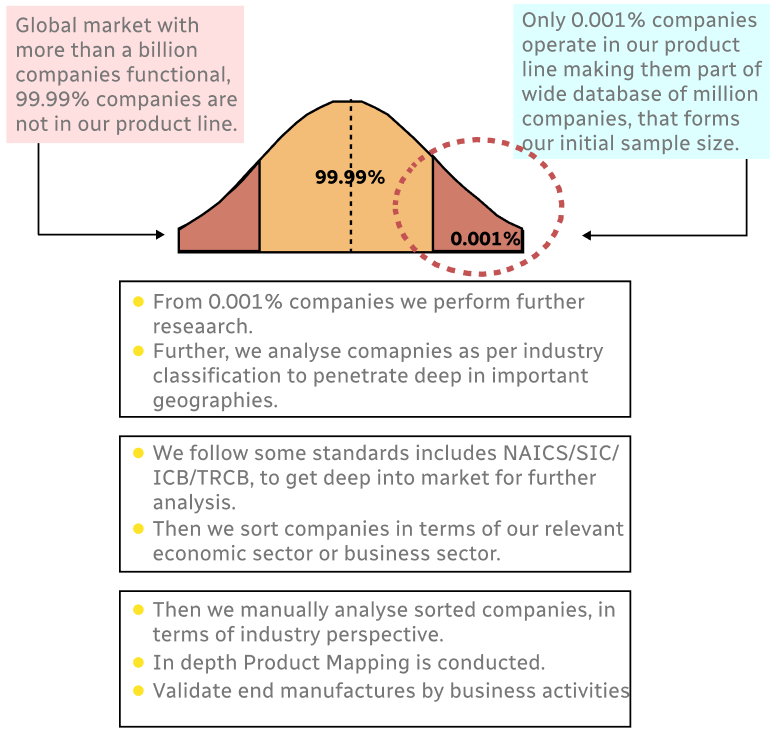
Step 1 - Identification of Relevant Samples Size from Population Database
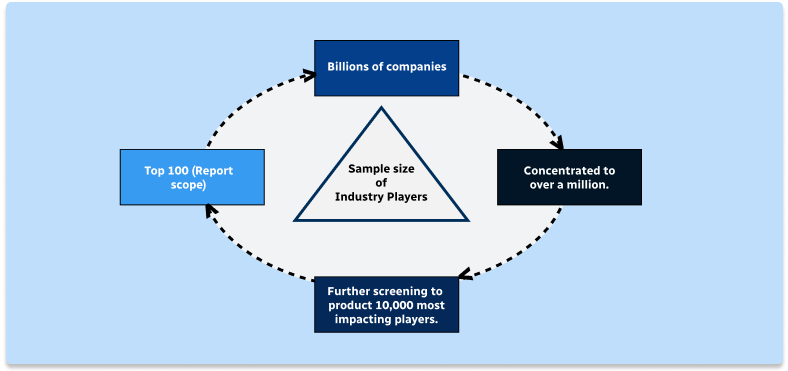
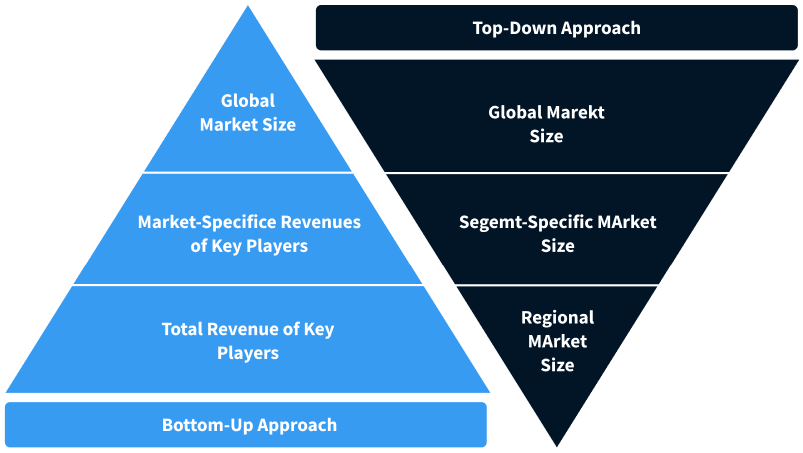
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
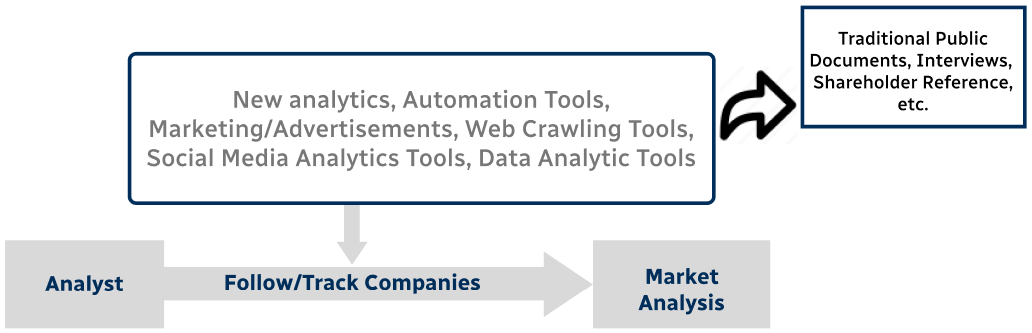
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


