Key Insights
The Melphalan Hydrochloride API market is poised for significant expansion, projecting a robust market size of $0.32 billion in 2025, with an anticipated Compound Annual Growth Rate (CAGR) of 6% from 2019 to 2033. This growth is primarily propelled by the increasing prevalence of specific cancers, most notably multiple myeloma and ovarian cancer, for which Melphalan Hydrochloride is a cornerstone treatment. Advances in pharmaceutical research and development are further driving demand, as ongoing studies explore novel therapeutic applications and combination therapies involving Melphalan Hydrochloride. The market’s expansion is also influenced by the growing global cancer burden, especially in emerging economies, where access to advanced cancer therapies is steadily improving. Furthermore, a heightened focus on patient-centric treatment approaches and the development of more targeted drug delivery systems are expected to create new avenues for market growth. The demand for high-purity Melphalan Hydrochloride API, particularly those exceeding Purity ≥98%, is also on the rise, reflecting stringent regulatory requirements and the need for efficacious and safe pharmaceutical formulations. The primary application segment for Melphalan Hydrochloride API is expected to be Injection, given its established role in intravenous chemotherapy regimens.
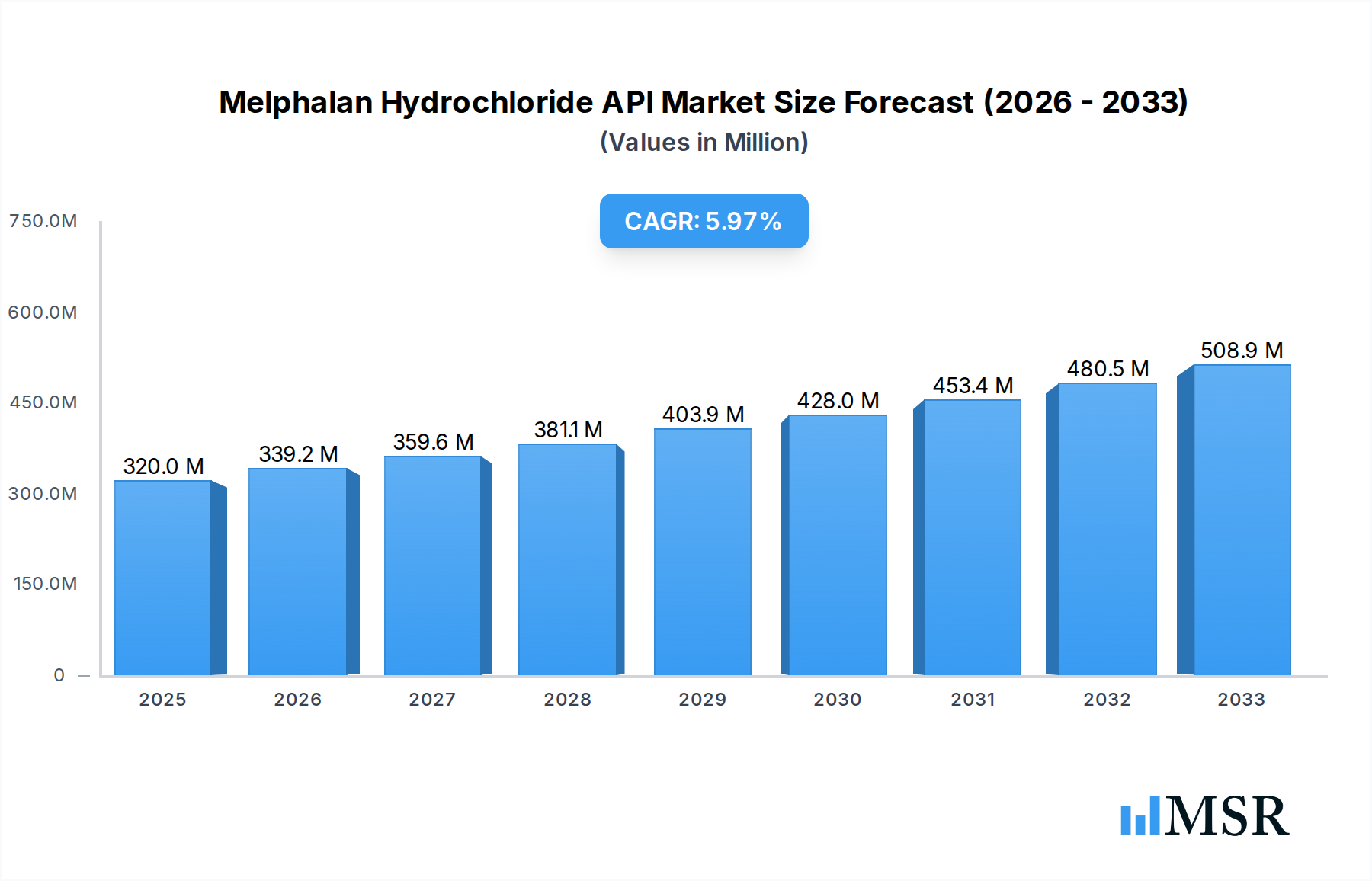
Melphalan Hydrochloride API Market Size (In Million)

While the market demonstrates strong growth potential, certain factors may present challenges. The production of Melphalan Hydrochloride API is complex and requires specialized manufacturing facilities and adherence to stringent quality control measures, which can lead to higher production costs. Additionally, the availability and cost of raw materials, coupled with evolving regulatory landscapes and patent expirations of existing formulations, could influence market dynamics. However, the enduring therapeutic efficacy of Melphalan Hydrochloride in treating life-threatening conditions, alongside continuous efforts to optimize its delivery and expand its therapeutic scope, ensures its continued relevance and drives the market's upward trajectory. The market's segmentation by purity, with a clear preference for Purity ≥98%, highlights the critical importance of API quality in pharmaceutical manufacturing, directly impacting drug efficacy and patient safety. The Injection application segment is expected to dominate, underscoring its established clinical utility in oncology.
Melphalan Hydrochloride API Company Market Share

Melphalan Hydrochloride API Market Concentration & Dynamics
The Melphalan Hydrochloride API market exhibits a moderate to high concentration, with key players like AUROBINDO PHARMA LTD, GRANULES INDIA LTD, HETERO LABS LTD, SHILPA MEDICARE LTD, and OLON SPA holding significant market shares. This concentration is further intensified by ongoing Mergers & Acquisitions (M&A) activities, with an estimated 10-15 M&A deals recorded during the historical period (2019-2024), aimed at consolidating market presence and expanding product portfolios. The innovation ecosystem is characterized by consistent research and development efforts, particularly in enhancing API purity and exploring novel delivery mechanisms. Regulatory frameworks, primarily driven by agencies like the FDA and EMA, play a crucial role in shaping market entry and product approvals, with 20-25 new drug master files (DMFs) submitted annually. The threat of substitute products remains low due to the specialized nature of Melphalan Hydrochloride API in oncological treatments. End-user trends are heavily influenced by the demand for advanced cancer therapies and the increasing prevalence of target diseases.
- Market Concentration: Moderate to High, with a few dominant players.
- M&A Activity: Estimated 10-15 deals (2019-2024), indicating consolidation.
- Innovation Focus: API purity enhancement and novel delivery systems.
- Regulatory Impact: Strict approvals by FDA, EMA, driving compliance.
- Substitute Products: Low threat due to specialized oncological applications.
- End-User Trends: Driven by demand for advanced cancer treatments.
Melphalan Hydrochloride API Industry Insights & Trends
The Melphalan Hydrochloride API market is poised for robust growth, driven by an increasing global cancer burden and advancements in pharmaceutical manufacturing. The global market size for Melphalan Hydrochloride API is projected to reach an impressive USD 1.5 billion by the base year 2025, with a projected Compound Annual Growth Rate (CAGR) of 6.5% during the forecast period of 2025-2033. This growth is underpinned by several key factors, including an expanding patient population diagnosed with myelodysplastic syndromes, multiple myeloma, and ovarian cancer, for which Melphalan Hydrochloride is a critical treatment component. Technological disruptions in API synthesis and purification technologies are leading to higher purity standards, with Purity ≥98% becoming the industry benchmark, driving demand for premium-grade APIs. Evolving consumer behaviors, specifically the rising preference for targeted therapies and personalized medicine, are indirectly fueling the demand for APIs like Melphalan Hydrochloride that form the backbone of such treatments. Furthermore, the increasing investment in oncology drug research and development by pharmaceutical giants and emerging biotechs is a significant market catalyst. The development of more efficient and cost-effective manufacturing processes by key manufacturers such as AUROBINDO PHARMA LTD, GRANULES INDIA LTD, and HETERO LABS LTD is also contributing to market expansion. The regulatory landscape, while stringent, is also facilitating growth by encouraging high-quality API production. The increasing accessibility of advanced healthcare infrastructure in emerging economies is further broadening the market reach for Melphalan Hydrochloride API. The global market is expected to witness a sustained upward trajectory, supported by ongoing clinical trials exploring new indications and combinations of Melphalan Hydrochloride. The ability of manufacturers to consistently deliver high-quality APIs at competitive prices will be paramount in capturing market share. The market dynamics are also shaped by the development of new drug formulations and delivery systems, which may enhance the therapeutic efficacy and patient compliance, thereby boosting API consumption. The rising expenditure on healthcare globally, coupled with government initiatives to combat cancer, are also contributing to the optimistic market outlook. The intellectual property landscape, with patents on manufacturing processes and specific formulations, also plays a role in market dynamics, influencing competition and pricing strategies. The consistent demand from established therapeutic areas, coupled with the exploration of potential new applications, ensures a steady growth path for Melphalan Hydrochloride API manufacturers. The commitment to quality and regulatory compliance by leading companies like SHILPA MEDICARE LTD and EMCURE PHARMACEUTICALS LTD further solidifies market trust and expansion.
Key Markets & Segments Leading Melphalan Hydrochloride API
The Injection segment dominates the Melphalan Hydrochloride API market, driven by its primary application in intravenous chemotherapy. Within this segment, Purity ≥98% is the prevailing standard, reflecting the stringent quality requirements for injectable pharmaceuticals. Geographically, North America and Europe currently lead in market share due to established healthcare infrastructures and a high prevalence of oncological diseases. However, the Asia-Pacific region is emerging as a significant growth driver, fueled by increasing healthcare spending, a growing patient population, and expanding pharmaceutical manufacturing capabilities in countries like India and China. The "Others" application segment, encompassing oral formulations and potential future uses, is also expected to witness steady growth.
- Dominant Application Segment: Injection
- Leading Purity Type: Purity ≥98%
- Key Geographic Markets: North America, Europe, and rapidly growing Asia-Pacific.
Melphalan Hydrochloride API Product Developments
Recent product developments in Melphalan Hydrochloride API focus on enhancing purity, stability, and bioavailability. Manufacturers are investing in advanced synthesis and purification techniques to achieve higher grades of Purity ≥98%, crucial for minimizing side effects and maximizing therapeutic efficacy. Innovations are also geared towards developing more stable formulations for improved shelf-life and easier handling. The market relevance of these developments lies in their ability to meet evolving regulatory standards and physician demands for safer and more effective cancer treatments. Companies like BIOPHORE INDIA PHARMACEUTICALS PVT LTD and FARMABIOS SPA are actively contributing to these advancements, aiming to provide a competitive edge in the pharmaceutical ingredient landscape.
Challenges in the Melphalan Hydrochloride API Market
The Melphalan Hydrochloride API market faces several critical challenges that impact its growth trajectory. Stringent regulatory approvals and the need for extensive documentation for drug master files represent a significant hurdle, demanding substantial investment and time. Supply chain disruptions, as evidenced by recent global events, can lead to raw material shortages and price volatility, affecting production timelines and costs. Intense competition among API manufacturers, particularly those adhering to Purity ≥98%, can put pressure on profit margins. Furthermore, the high cost of specialized manufacturing equipment and adherence to Good Manufacturing Practices (GMP) add to the operational expenses, acting as a barrier to entry for smaller players.
- Regulatory Hurdles: Lengthy and complex approval processes.
- Supply Chain Vulnerabilities: Risk of raw material shortages and price fluctuations.
- Competitive Pressures: Impact on pricing and profitability.
- High Manufacturing Costs: Investment in specialized equipment and compliance.
Forces Driving Melphalan Hydrochloride API Growth
The Melphalan Hydrochloride API market is propelled by several potent growth drivers. The increasing global incidence of cancer, particularly myelodysplastic syndromes, multiple myeloma, and ovarian cancer, directly translates to a higher demand for this critical therapeutic agent. Technological advancements in API synthesis and purification technologies are enabling manufacturers to produce higher purity APIs, such as Purity ≥98%, meeting the stringent requirements of the pharmaceutical industry. Government initiatives and increasing healthcare expenditure globally are also bolstering the market by improving access to advanced cancer treatments. Furthermore, the expanding pharmaceutical research and development activities focusing on oncology drugs are continuously creating new opportunities for API suppliers.
Challenges in the Melphalan Hydrochloride API Market
While growth is evident, long-term sustainability in the Melphalan Hydrochloride API market hinges on addressing inherent challenges. The patent expiries of blockbuster cancer drugs that utilize Melphalan Hydrochloride can lead to increased generic competition, potentially impacting pricing power. The development of novel therapeutic modalities like immunotherapy and targeted gene therapies, while promising for cancer treatment, could eventually reduce reliance on traditional chemotherapy agents like Melphalan Hydrochloride. Geopolitical instability and trade barriers can further disrupt global supply chains and impact the cost-effectiveness of API manufacturing and distribution. Companies must remain agile and invest in diversified production capabilities and strategic partnerships to mitigate these long-term risks.
Emerging Opportunities in Melphalan Hydrochloride API
The Melphalan Hydrochloride API market is ripe with emerging opportunities for forward-thinking stakeholders. The growing demand for high-purity APIs (Purity ≥98%) in emerging economies presents a significant untapped market. Technological advancements in continuous manufacturing processes offer the potential for increased efficiency and reduced production costs, leading to competitive advantages. Furthermore, the exploration of novel drug delivery systems for Melphalan Hydrochloride, such as liposomal formulations, could enhance therapeutic outcomes and create new market niches. Strategic collaborations and partnerships with pharmaceutical companies developing next-generation oncology treatments can unlock substantial growth avenues.
Leading Players in the Melphalan Hydrochloride API Sector
- AUROBINDO PHARMA LTD
- GRANULES INDIA LTD
- NAVINTA LLC
- HETERO LABS LTD
- SHILPA MEDICARE LTD
- BIOPHORE INDIA PHARMACEUTICALS PVT LTD
- NERPHARMA SRL
- FARMABIOS SPA
- EMCURE PHARMACEUTICALS LTD
- OLON SPA
Key Milestones in Melphalan Hydrochloride API Industry
- 2019: Increased regulatory scrutiny on API impurities, driving demand for Purity ≥98%.
- 2020: Global supply chain disruptions due to the pandemic, highlighting the need for resilient manufacturing.
- 2021: Emergence of advanced purification techniques, improving Melphalan Hydrochloride API quality.
- 2022: Several key players initiated capacity expansions to meet rising demand.
- 2023: Focus on sustainable manufacturing practices within the API industry.
- 2024: Continued investment in R&D for novel delivery systems of Melphalan Hydrochloride.
Strategic Outlook for Melphalan Hydrochloride API Market
The strategic outlook for the Melphalan Hydrochloride API market remains positive, driven by sustained demand in established oncological applications and emerging market expansion. Key growth accelerators include continued investment in R&D for higher purity standards (Purity ≥98%) and the exploration of novel delivery mechanisms. Strategic partnerships between API manufacturers and oncology drug developers will be crucial for navigating the evolving therapeutic landscape. Furthermore, focusing on supply chain resilience and adherence to stringent regulatory requirements will ensure long-term market competitiveness. The market's trajectory is expected to be shaped by innovation in manufacturing processes and an increasing focus on patient-centric drug development, promising sustained growth throughout the forecast period.
Melphalan Hydrochloride API Segmentation
-
1. Application
- 1.1. Injection
- 1.2. Others
-
2. Types
- 2.1. Purity≥98%
- 2.2. Purity<98%
Melphalan Hydrochloride API Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
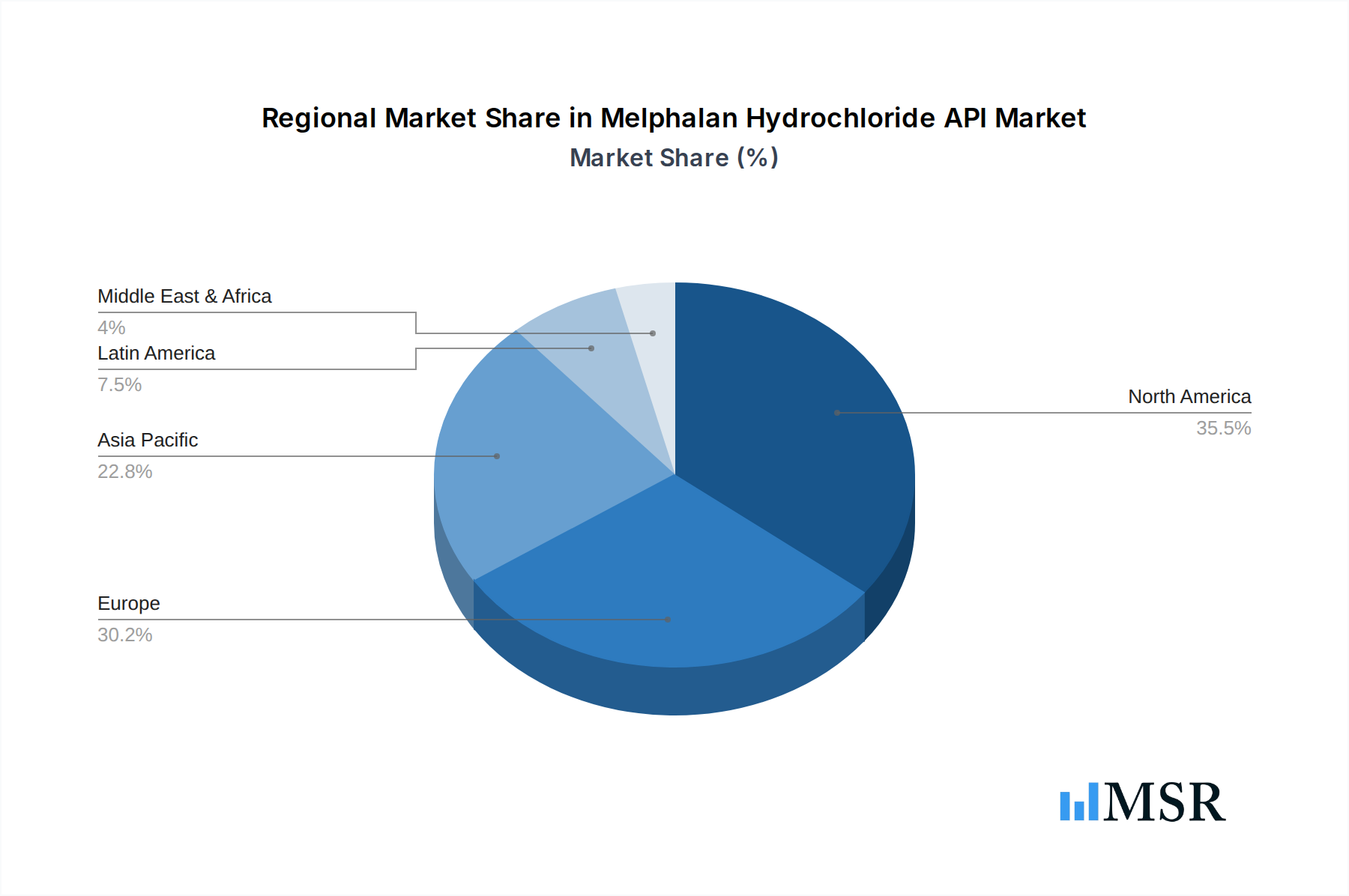
Melphalan Hydrochloride API Regional Market Share

Geographic Coverage of Melphalan Hydrochloride API
Melphalan Hydrochloride API REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Melphalan Hydrochloride API Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Injection
- 5.1.2. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Purity≥98%
- 5.2.2. Purity<98%
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Melphalan Hydrochloride API Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Injection
- 6.1.2. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Purity≥98%
- 6.2.2. Purity<98%
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Melphalan Hydrochloride API Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Injection
- 7.1.2. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Purity≥98%
- 7.2.2. Purity<98%
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Melphalan Hydrochloride API Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Injection
- 8.1.2. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Purity≥98%
- 8.2.2. Purity<98%
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Melphalan Hydrochloride API Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Injection
- 9.1.2. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Purity≥98%
- 9.2.2. Purity<98%
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Melphalan Hydrochloride API Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Injection
- 10.1.2. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Purity≥98%
- 10.2.2. Purity<98%
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 AUROBINDO PHARMA LTD
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 GRANULES INDIA LTD
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 NAVINTA LLC
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 HETERO LABS LTD
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 SHILPA MEDICARE LTD
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 BIOPHORE INDIA PHARMACEUTICALS PVT LTD
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 NERPHARMA SRL
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 FARMABIOS SPA
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 EMCURE PHARMACEUTICALS LTD
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 OLON SPA
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.1 AUROBINDO PHARMA LTD
List of Figures
- Figure 1: Global Melphalan Hydrochloride API Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Melphalan Hydrochloride API Revenue (billion), by Application 2025 & 2033
- Figure 3: North America Melphalan Hydrochloride API Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Melphalan Hydrochloride API Revenue (billion), by Types 2025 & 2033
- Figure 5: North America Melphalan Hydrochloride API Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Melphalan Hydrochloride API Revenue (billion), by Country 2025 & 2033
- Figure 7: North America Melphalan Hydrochloride API Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Melphalan Hydrochloride API Revenue (billion), by Application 2025 & 2033
- Figure 9: South America Melphalan Hydrochloride API Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Melphalan Hydrochloride API Revenue (billion), by Types 2025 & 2033
- Figure 11: South America Melphalan Hydrochloride API Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Melphalan Hydrochloride API Revenue (billion), by Country 2025 & 2033
- Figure 13: South America Melphalan Hydrochloride API Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Melphalan Hydrochloride API Revenue (billion), by Application 2025 & 2033
- Figure 15: Europe Melphalan Hydrochloride API Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Melphalan Hydrochloride API Revenue (billion), by Types 2025 & 2033
- Figure 17: Europe Melphalan Hydrochloride API Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Melphalan Hydrochloride API Revenue (billion), by Country 2025 & 2033
- Figure 19: Europe Melphalan Hydrochloride API Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Melphalan Hydrochloride API Revenue (billion), by Application 2025 & 2033
- Figure 21: Middle East & Africa Melphalan Hydrochloride API Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Melphalan Hydrochloride API Revenue (billion), by Types 2025 & 2033
- Figure 23: Middle East & Africa Melphalan Hydrochloride API Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Melphalan Hydrochloride API Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East & Africa Melphalan Hydrochloride API Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Melphalan Hydrochloride API Revenue (billion), by Application 2025 & 2033
- Figure 27: Asia Pacific Melphalan Hydrochloride API Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Melphalan Hydrochloride API Revenue (billion), by Types 2025 & 2033
- Figure 29: Asia Pacific Melphalan Hydrochloride API Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Melphalan Hydrochloride API Revenue (billion), by Country 2025 & 2033
- Figure 31: Asia Pacific Melphalan Hydrochloride API Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Melphalan Hydrochloride API Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global Melphalan Hydrochloride API Revenue billion Forecast, by Types 2020 & 2033
- Table 3: Global Melphalan Hydrochloride API Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global Melphalan Hydrochloride API Revenue billion Forecast, by Application 2020 & 2033
- Table 5: Global Melphalan Hydrochloride API Revenue billion Forecast, by Types 2020 & 2033
- Table 6: Global Melphalan Hydrochloride API Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global Melphalan Hydrochloride API Revenue billion Forecast, by Application 2020 & 2033
- Table 11: Global Melphalan Hydrochloride API Revenue billion Forecast, by Types 2020 & 2033
- Table 12: Global Melphalan Hydrochloride API Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Brazil Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: Argentina Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Global Melphalan Hydrochloride API Revenue billion Forecast, by Application 2020 & 2033
- Table 17: Global Melphalan Hydrochloride API Revenue billion Forecast, by Types 2020 & 2033
- Table 18: Global Melphalan Hydrochloride API Revenue billion Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 20: Germany Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 21: France Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 22: Italy Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Spain Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: Russia Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Benelux Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Nordics Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global Melphalan Hydrochloride API Revenue billion Forecast, by Application 2020 & 2033
- Table 29: Global Melphalan Hydrochloride API Revenue billion Forecast, by Types 2020 & 2033
- Table 30: Global Melphalan Hydrochloride API Revenue billion Forecast, by Country 2020 & 2033
- Table 31: Turkey Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Israel Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: GCC Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: North Africa Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 35: South Africa Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 37: Global Melphalan Hydrochloride API Revenue billion Forecast, by Application 2020 & 2033
- Table 38: Global Melphalan Hydrochloride API Revenue billion Forecast, by Types 2020 & 2033
- Table 39: Global Melphalan Hydrochloride API Revenue billion Forecast, by Country 2020 & 2033
- Table 40: China Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 41: India Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: Japan Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 43: South Korea Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 45: Oceania Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Melphalan Hydrochloride API Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Melphalan Hydrochloride API?
The projected CAGR is approximately 6%.
2. Which companies are prominent players in the Melphalan Hydrochloride API?
Key companies in the market include AUROBINDO PHARMA LTD, GRANULES INDIA LTD, NAVINTA LLC, HETERO LABS LTD, SHILPA MEDICARE LTD, BIOPHORE INDIA PHARMACEUTICALS PVT LTD, NERPHARMA SRL, FARMABIOS SPA, EMCURE PHARMACEUTICALS LTD, OLON SPA.
3. What are the main segments of the Melphalan Hydrochloride API?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 0.32 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Melphalan Hydrochloride API," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Melphalan Hydrochloride API report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Melphalan Hydrochloride API?
To stay informed about further developments, trends, and reports in the Melphalan Hydrochloride API, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
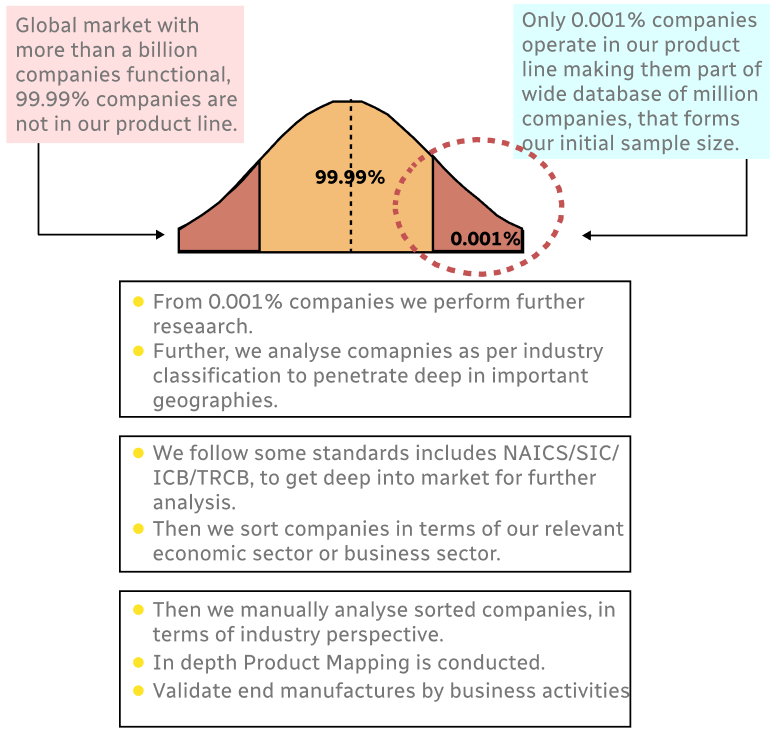
Step 1 - Identification of Relevant Samples Size from Population Database
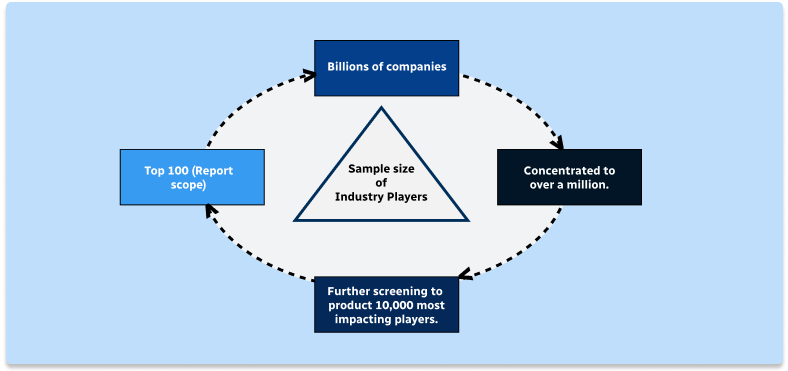
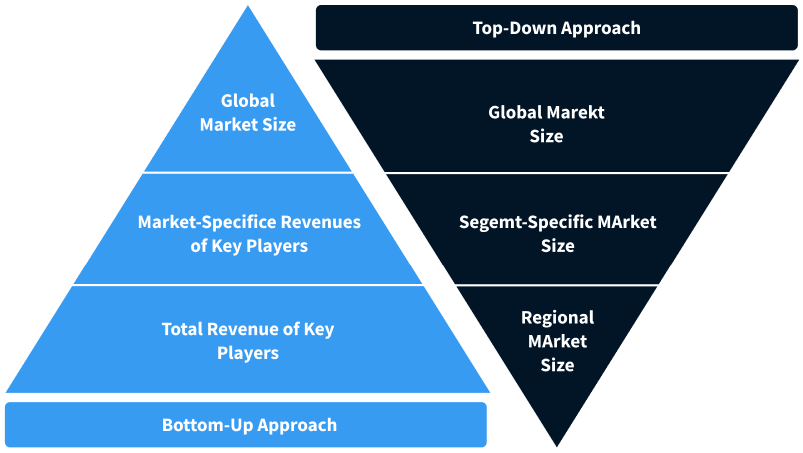
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
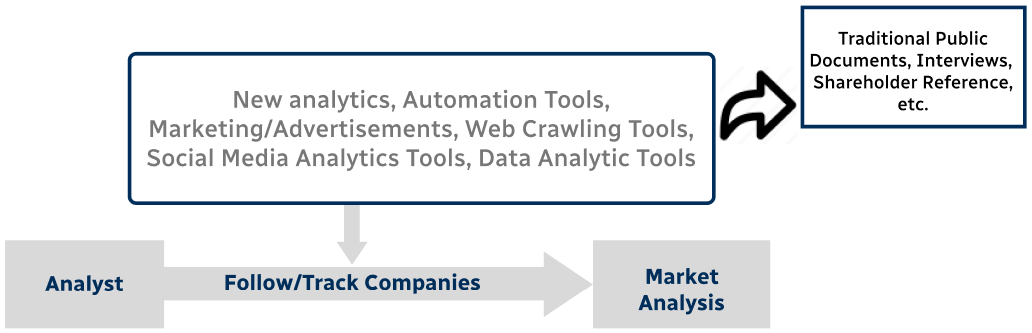
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


