Key Insights
The global market for Rabies Virus Monoclonal Antibodies is poised for significant expansion, projected to reach USD 9.28 billion by 2025, exhibiting a robust CAGR of 13.33% through 2033. This growth is primarily fueled by the increasing prevalence of rabies in developing nations, coupled with rising awareness and demand for more effective post-exposure prophylaxis (PEP) treatments. The development of novel, highly specific monoclonal antibodies offers a promising alternative or adjunct to traditional rabies immunoglobulin, boasting enhanced efficacy and potentially reduced side effects. Key applications are centered around Category II and Category III exposure scenarios, where rapid and potent neutralization of the rabies virus is critical for preventing disease onset. The market is witnessing a surge in demand for various vial sizes, including 100IU, 200IU, 250IU, 600IU, and 1500IU, catering to diverse clinical needs and patient demographics. This increasing demand for advanced rabies treatment solutions is a testament to the evolving landscape of infectious disease management and the continuous pursuit of improved patient outcomes.
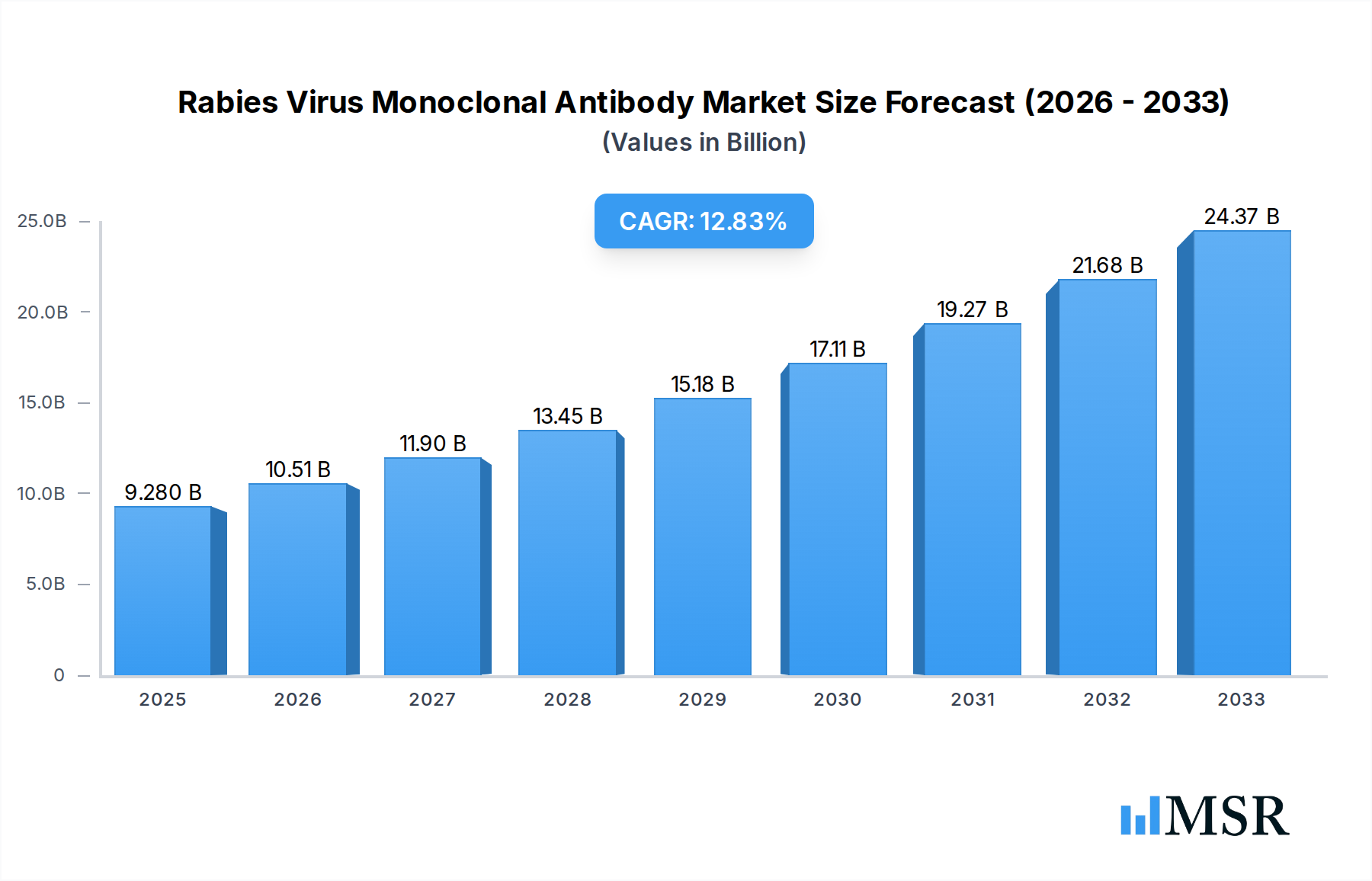
Rabies Virus Monoclonal Antibody Market Size (In Billion)

Several factors are driving this upward trajectory. Advancements in biotechnology and recombinant DNA technology are enabling the more efficient and cost-effective production of these complex antibodies. Government initiatives and public health campaigns aimed at rabies eradication further bolster market growth by increasing diagnostic rates and treatment accessibility. Leading players like the Serum Institute of India, Zydus Lifesciences, North China Pharmaceutical, and Xingmeng Biopharmaceuticals (Suzhou) are actively investing in research and development to expand their product portfolios and geographical reach. While the market is largely driven by the need for effective rabies prevention, potential restraints could include stringent regulatory hurdles for new biological products, high manufacturing costs, and the availability of existing cost-effective vaccines. However, the inherent advantages of monoclonal antibodies in terms of specificity and therapeutic potential are expected to outweigh these challenges, positioning the market for sustained and accelerated growth in the coming years.
Rabies Virus Monoclonal Antibody Company Market Share

Rabies Virus Monoclonal Antibody Market Analysis: Comprehensive Insights and Forecast 2019-2033
This in-depth market report provides a detailed analysis of the global Rabies Virus Monoclonal Antibody market, encompassing historical trends, current dynamics, and future projections. Driven by increasing global rabies awareness, advancements in biopharmaceutical research, and a growing demand for effective post-exposure prophylaxis, the market is poised for significant expansion. Our comprehensive study covers market concentration, key industry trends, segmentation analysis, product developments, challenges, growth drivers, emerging opportunities, leading players, and strategic outlooks, offering actionable insights for industry stakeholders. The report utilizes the Study Period: 2019–2033, with Base Year: 2025, Estimated Year: 2025, and Forecast Period: 2025–2033, providing a robust understanding of market trajectories. Historical data from 2019–2024 sets the context for future projections.
Rabies Virus Monoclonal Antibody Market Concentration & Dynamics
The Rabies Virus Monoclonal Antibody market exhibits a moderate to high concentration, with a few dominant global players alongside a growing number of regional and niche manufacturers. Innovation plays a crucial role, driven by continuous research into improved efficacy, longer shelf-life, and more cost-effective production methods. Regulatory frameworks, such as those established by the FDA, EMA, and WHO, significantly influence market entry and product approvals, ensuring patient safety and product quality. Substitute products, primarily traditional rabies immunoglobulin (RIG) derived from human or equine sources, are gradually being complemented and, in some cases, replaced by monoclonal antibodies due to their higher specificity and reduced risk of adverse reactions. End-user trends are increasingly favoring these advanced biologics for their predictable pharmacokinetic profiles and reduced immunogenicity. Mergers and acquisitions (M&A) activities are anticipated to increase as larger pharmaceutical companies seek to expand their biologics portfolios and gain market share. M&A deal counts are projected to rise by approximately 15% over the forecast period, reflecting strategic consolidation. Market share for leading players is estimated to range between 10% and 25% for the top three entities.
Rabies Virus Monoclonal Antibody Industry Insights & Trends
The global Rabies Virus Monoclonal Antibody market is experiencing robust growth, fueled by several interconnected factors. A primary growth driver is the escalating global burden of rabies, a zoonotic disease that remains a significant public health threat, particularly in developing nations. The World Health Organization's intensified efforts to eliminate rabies by 2030, coupled with increased public awareness campaigns, are directly translating into higher demand for effective post-exposure prophylaxis (PEP). The market size was approximately $1.2 billion in 2024 and is projected to reach $3.5 billion by 2033, demonstrating a Compound Annual Growth Rate (CAGR) of approximately 12.5% during the forecast period. Technological disruptions, such as advancements in recombinant DNA technology and single-use bioreactor systems, are enabling more efficient and scalable production of monoclonal antibodies, thereby reducing manufacturing costs and improving accessibility. Evolving consumer behaviors, influenced by greater access to health information and a preference for evidence-based medical interventions, are steering healthcare providers and patients towards newer, more targeted therapies like monoclonal antibodies. The development of novel antibody formats, such as bispecific antibodies designed to neutralize multiple viral strains or enhance immune responses, represents a significant trend. Furthermore, the increasing adoption of PEP regimens that include monoclonal antibodies, particularly for Category III exposures, is a critical factor bolstering market expansion. The overall industry is witnessing a paradigm shift towards personalized medicine and targeted therapies, with rabies virus monoclonal antibodies standing at the forefront of this evolution.
Key Markets & Segments Leading Rabies Virus Monoclonal Antibody
The dominance in the Rabies Virus Monoclonal Antibody market is multifaceted, with specific regions and application segments driving significant demand. North America and Europe currently lead the market, owing to well-established healthcare infrastructures, higher disposable incomes, advanced research and development capabilities, and proactive public health initiatives for rabies prevention. However, the Asia-Pacific region is emerging as a high-growth market, driven by a rising incidence of rabies, increasing healthcare expenditure, and improving access to advanced medical treatments.
Dominant Application Segments:
- Category III Exposure: This segment is a primary revenue generator. Exposure incidents involving severe bites, multiple wounds, or bites on the head or face, which carry a higher risk of rabies transmission, necessitate immediate and effective PEP. The use of monoclonal antibodies in these critical situations is increasingly becoming the standard of care due to their rapid onset of action and high neutralization capacity. The market for Category III Exposure applications is expected to grow at a CAGR of 13.8% during the forecast period, driven by greater awareness and improved diagnostic capabilities leading to timely intervention.
- Category II Exposure: This segment, involving less severe exposures like single or minor bites through clothing, also contributes significantly. As awareness of rabies prevention grows, the demand for effective PEP, including monoclonal antibodies, in these categories is also rising. The market for Category II Exposure is projected to see a CAGR of 11.5%.
Dominant Product Type Segments:
- 1500IU per Vial: This high-potency formulation is crucial for severe exposures and pediatric cases, offering a concentrated dose for rapid neutralization of the rabies virus. Its market share is substantial due to its critical role in life-saving interventions.
- 600IU per Vial: This versatile dosage is widely used for a range of exposure scenarios, offering a balance between efficacy and cost-effectiveness, contributing to its significant market presence.
- 200IU per Vial & 250IU per Vial: These intermediate dosages cater to specific patient profiles and exposure severities, providing flexibility in treatment regimens.
- 100IU per Vial: While representing a smaller segment, this dosage is valuable for specific pediatric applications or milder exposure scenarios, ensuring precise dosing.
Economic growth in emerging economies, coupled with government investments in public health infrastructure and vaccine programs, are key drivers. The increasing prevalence of stray animal populations and the associated risk of animal bites in densely populated urban areas further fuel demand. Infrastructure development, including improved cold chain logistics for biologics, is also crucial for market expansion.
Rabies Virus Monoclonal Antibody Product Developments
The Rabies Virus Monoclonal Antibody market is witnessing continuous innovation focused on enhancing therapeutic efficacy and patient convenience. Companies are developing novel antibody variants with improved binding affinity and longer half-lives, aiming to reduce the frequency of administration and improve patient compliance. Research is also being directed towards developing fully humanized monoclonal antibodies to minimize immunogenicity and potential allergic reactions. Furthermore, advancements in manufacturing processes, including continuous bioprocessing and advanced purification techniques, are leading to more cost-effective production, making these advanced therapies more accessible globally. The development of combination therapies, integrating monoclonal antibodies with existing vaccine protocols, is another promising area of product development, aiming for more robust and long-lasting immunity against rabies.
Challenges in the Rabies Virus Monoclonal Antibody Market
The Rabies Virus Monoclonal Antibody market, while promising, faces several significant challenges. High manufacturing costs associated with biologics can lead to premium pricing, limiting accessibility in resource-constrained regions. Stringent regulatory approval processes across different countries, though essential for safety, can prolong time-to-market and increase development expenditures. Supply chain complexities, including the need for specialized cold chain logistics, present logistical hurdles, especially in remote or underserved areas. Furthermore, the availability and established use of traditional rabies immunoglobulins can create inertia in adoption, requiring significant market education and physician outreach. Competitive pressures from biosimilar development and evolving treatment guidelines also necessitate continuous innovation and cost optimization strategies.
Forces Driving Rabies Virus Monoclonal Antibody Growth
The growth of the Rabies Virus Monoclonal Antibody market is propelled by a confluence of critical forces. A fundamental driver is the persistent global threat of rabies, which necessitates effective and rapid post-exposure prophylaxis. The increasing emphasis on public health initiatives and disease eradication programs by international organizations like the WHO directly translates to higher demand for advanced prophylactic treatments. Technological advancements in biotechnology and biopharmaceutical manufacturing are crucial, enabling the development of more potent, specific, and cost-effective monoclonal antibodies. Evolving clinical guidelines and growing physician awareness regarding the benefits of monoclonal antibodies, such as their predictable pharmacokinetic profiles and reduced immunogenicity compared to traditional immunoglobulins, are also significant growth accelerators. Rising healthcare expenditure and improving access to advanced medical treatments in emerging economies are further expanding the market reach.
Challenges in the Rabies Virus Monoclonal Antibody Market
The long-term growth of the Rabies Virus Monoclonal Antibody market is contingent on overcoming persistent challenges. The high cost of production and subsequent pricing of monoclonal antibodies remains a primary barrier to widespread adoption, especially in low and middle-income countries where rabies incidence is highest. Complex and often lengthy regulatory approval pathways in different geographical regions can stifle market penetration and necessitate substantial investment in compliance. Ensuring a robust and reliable global supply chain, including specialized cold chain management for these sensitive biologics, poses logistical challenges that can impact product availability. Furthermore, competition from established, lower-cost traditional rabies immunoglobulins requires continuous market education and demonstration of superior clinical and economic value. The development of potential resistance or reduced efficacy against emerging viral strains would necessitate ongoing research and adaptation of antibody therapies.
Emerging Opportunities in Rabies Virus Monoclonal Antibody
The Rabies Virus Monoclonal Antibody market is ripe with emerging opportunities, driven by ongoing research and evolving healthcare landscapes. A key opportunity lies in the development of next-generation monoclonal antibodies with enhanced neutralization capabilities, longer half-lives, and potentially broader spectrum activity against different rabies virus variants. The expansion into previously underserved emerging markets, particularly in Asia and Africa, presents substantial growth potential, requiring tailored market strategies and potentially more affordable product offerings. Strategic partnerships and collaborations between antibody developers, vaccine manufacturers, and public health organizations can accelerate product development, facilitate market access, and improve global rabies prevention efforts. Furthermore, the exploration of novel delivery methods and combination therapies, such as co-formulating antibodies with vaccines for simplified administration, offers significant untapped potential. The growing focus on one-health approaches to disease prevention also opens avenues for integrated rabies control strategies that leverage advanced biological interventions.
Leading Players in the Rabies Virus Monoclonal Antibody Sector
- Serum Institute of India
- Zydus Lifesciences
- North China Pharmaceutical
- Xingmeng Biopharmaceuticals (Suzhou)
Key Milestones in Rabies Virus Monoclonal Antibody Industry
- 2019: Initiation of Phase II clinical trials for novel recombinant rabies virus monoclonal antibodies by leading research institutions.
- 2020: Increased global focus on zoonotic disease prevention and control, boosting investment in advanced rabies prophylaxis research.
- 2021: Regulatory submission for a new rabies virus monoclonal antibody in key emerging markets, signaling potential for broader access.
- 2022: Significant advancements in manufacturing technologies, leading to improved yields and reduced production costs for monoclonal antibodies.
- 2023: Expansion of clinical trials to include larger patient cohorts and diverse geographical regions, strengthening the evidence base for efficacy.
- 2024: Strategic collaborations announced between major pharmaceutical companies and research consortia to accelerate development and commercialization.
Strategic Outlook for Rabies Virus Monoclonal Antibody Market
The strategic outlook for the Rabies Virus Monoclonal Antibody market is exceptionally positive, driven by sustained demand and ongoing innovation. Key growth accelerators include the continued development of more potent and cost-effective monoclonal antibodies, enhanced by advancements in bioprocessing. Strategic market expansion into high-incidence regions in Asia and Africa, coupled with targeted public health campaigns, will unlock significant revenue potential. Furthermore, fostering collaborations for integrated rabies control programs, combining advanced prophylactics with robust vaccination strategies, represents a critical pathway for long-term market success. The increasing focus on personalized medicine and the demonstrated superiority of monoclonal antibodies in specific exposure scenarios will continue to drive their adoption as the standard of care, ensuring robust market growth through 2033 and beyond.
Rabies Virus Monoclonal Antibody Segmentation
-
1. Application
- 1.1. Category II Exposure
- 1.2. Category III Exposure
-
2. Types
- 2.1. 100IU per Vial
- 2.2. 200IU per Vial
- 2.3. 250IU per Vial
- 2.4. 600IU per Vial
- 2.5. 1500IU per Vial
Rabies Virus Monoclonal Antibody Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
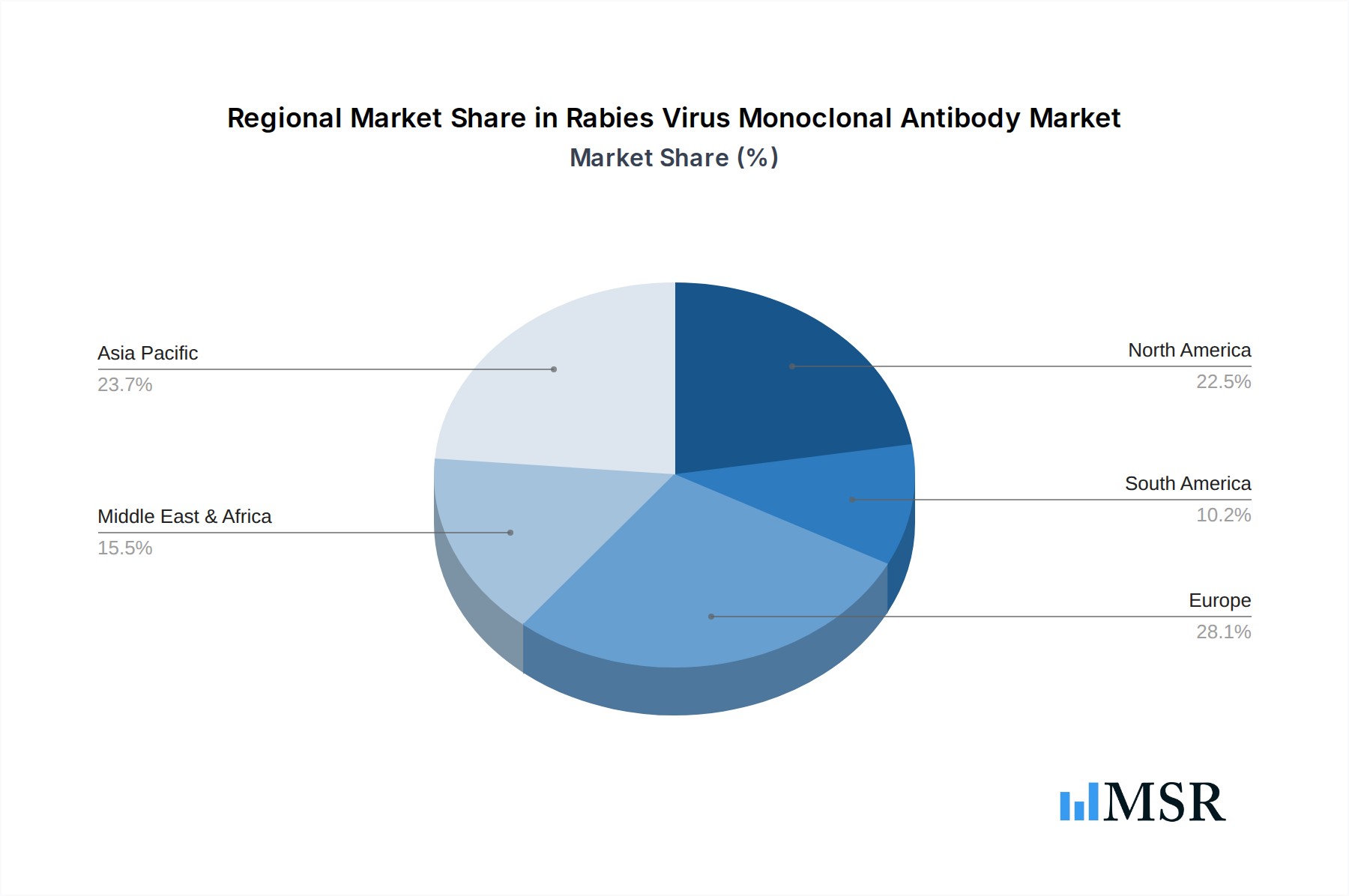
Rabies Virus Monoclonal Antibody Regional Market Share

Geographic Coverage of Rabies Virus Monoclonal Antibody
Rabies Virus Monoclonal Antibody REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 13.33% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Rabies Virus Monoclonal Antibody Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Category II Exposure
- 5.1.2. Category III Exposure
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. 100IU per Vial
- 5.2.2. 200IU per Vial
- 5.2.3. 250IU per Vial
- 5.2.4. 600IU per Vial
- 5.2.5. 1500IU per Vial
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Rabies Virus Monoclonal Antibody Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Category II Exposure
- 6.1.2. Category III Exposure
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. 100IU per Vial
- 6.2.2. 200IU per Vial
- 6.2.3. 250IU per Vial
- 6.2.4. 600IU per Vial
- 6.2.5. 1500IU per Vial
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Rabies Virus Monoclonal Antibody Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Category II Exposure
- 7.1.2. Category III Exposure
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. 100IU per Vial
- 7.2.2. 200IU per Vial
- 7.2.3. 250IU per Vial
- 7.2.4. 600IU per Vial
- 7.2.5. 1500IU per Vial
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Rabies Virus Monoclonal Antibody Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Category II Exposure
- 8.1.2. Category III Exposure
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. 100IU per Vial
- 8.2.2. 200IU per Vial
- 8.2.3. 250IU per Vial
- 8.2.4. 600IU per Vial
- 8.2.5. 1500IU per Vial
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Rabies Virus Monoclonal Antibody Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Category II Exposure
- 9.1.2. Category III Exposure
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. 100IU per Vial
- 9.2.2. 200IU per Vial
- 9.2.3. 250IU per Vial
- 9.2.4. 600IU per Vial
- 9.2.5. 1500IU per Vial
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Rabies Virus Monoclonal Antibody Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Category II Exposure
- 10.1.2. Category III Exposure
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. 100IU per Vial
- 10.2.2. 200IU per Vial
- 10.2.3. 250IU per Vial
- 10.2.4. 600IU per Vial
- 10.2.5. 1500IU per Vial
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Serum Institute of India
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Zydus Lifesciences
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 North China Pharmaceutical
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Xingmeng Biopharmaceuticals (Suzhou)
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.1 Serum Institute of India
List of Figures
- Figure 1: Global Rabies Virus Monoclonal Antibody Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Rabies Virus Monoclonal Antibody Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Rabies Virus Monoclonal Antibody Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Rabies Virus Monoclonal Antibody Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Rabies Virus Monoclonal Antibody Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Rabies Virus Monoclonal Antibody Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Rabies Virus Monoclonal Antibody Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Rabies Virus Monoclonal Antibody Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Rabies Virus Monoclonal Antibody Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Rabies Virus Monoclonal Antibody Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Rabies Virus Monoclonal Antibody Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Rabies Virus Monoclonal Antibody Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Rabies Virus Monoclonal Antibody Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Rabies Virus Monoclonal Antibody Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Rabies Virus Monoclonal Antibody Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Rabies Virus Monoclonal Antibody Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Rabies Virus Monoclonal Antibody Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Rabies Virus Monoclonal Antibody Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Rabies Virus Monoclonal Antibody Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Rabies Virus Monoclonal Antibody Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Rabies Virus Monoclonal Antibody Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Rabies Virus Monoclonal Antibody Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Rabies Virus Monoclonal Antibody Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Rabies Virus Monoclonal Antibody Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Rabies Virus Monoclonal Antibody Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Rabies Virus Monoclonal Antibody Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Rabies Virus Monoclonal Antibody Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Rabies Virus Monoclonal Antibody Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Rabies Virus Monoclonal Antibody Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Rabies Virus Monoclonal Antibody Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Rabies Virus Monoclonal Antibody Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Rabies Virus Monoclonal Antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Rabies Virus Monoclonal Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Rabies Virus Monoclonal Antibody?
The projected CAGR is approximately 13.33%.
2. Which companies are prominent players in the Rabies Virus Monoclonal Antibody?
Key companies in the market include Serum Institute of India, Zydus Lifesciences, North China Pharmaceutical, Xingmeng Biopharmaceuticals (Suzhou).
3. What are the main segments of the Rabies Virus Monoclonal Antibody?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4900.00, USD 7350.00, and USD 9800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Rabies Virus Monoclonal Antibody," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Rabies Virus Monoclonal Antibody report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Rabies Virus Monoclonal Antibody?
To stay informed about further developments, trends, and reports in the Rabies Virus Monoclonal Antibody, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
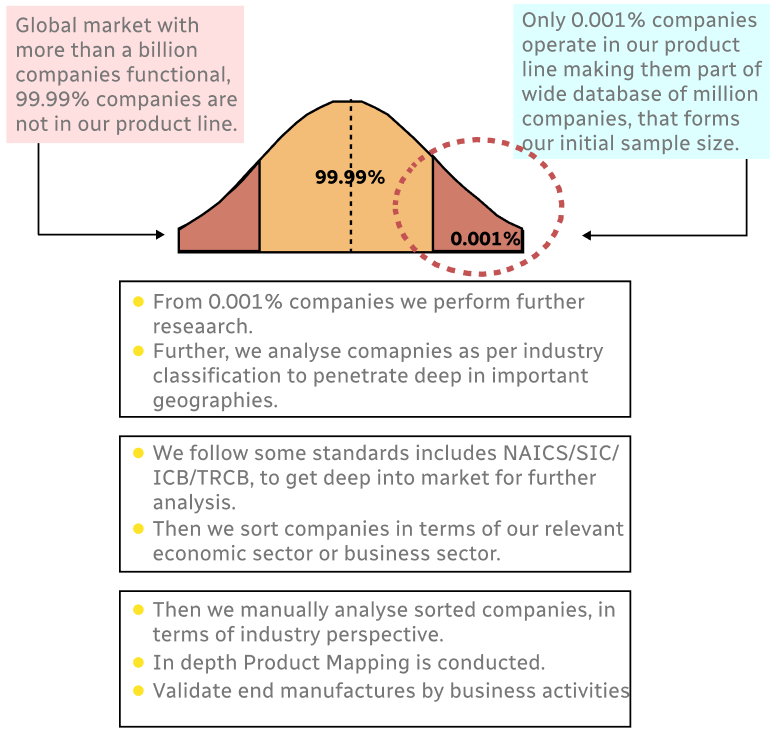
Step 1 - Identification of Relevant Samples Size from Population Database
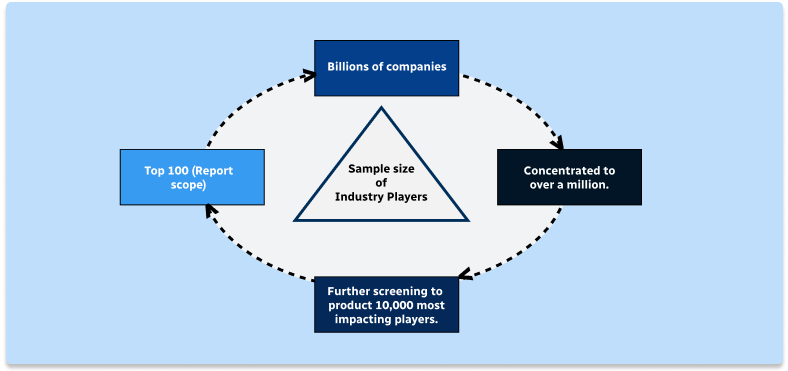
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
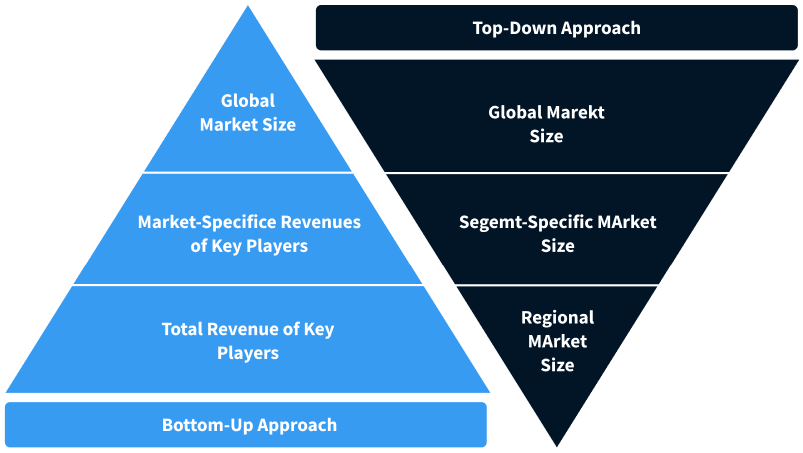
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
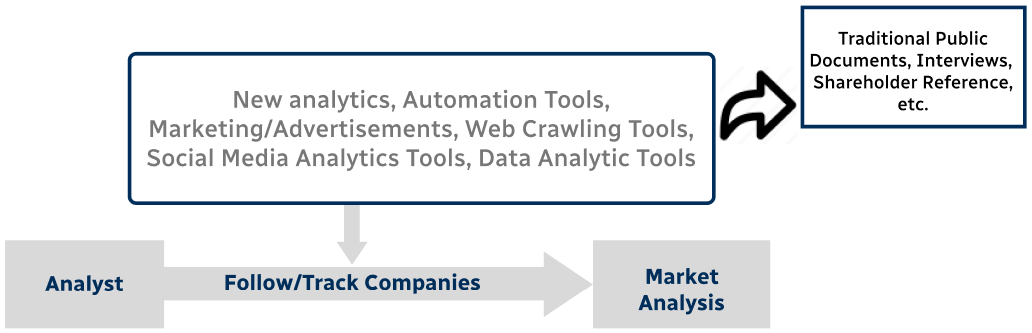
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


