Key Insights
The Rivaroxaban Tablets market is poised for significant growth, projected to reach $12.72 billion in 2025, with a robust CAGR of 8.17% expected to propel it further. This expansion is primarily fueled by the increasing prevalence of cardiovascular diseases, deep vein thrombosis (DVT), and pulmonary embolism (PE) globally. As an anticoagulant, Rivaroxaban plays a crucial role in preventing and treating these life-threatening conditions, leading to a sustained demand across various healthcare settings. The market's trajectory is further bolstered by advancements in pharmaceutical research and development, leading to improved formulations and wider accessibility. The presence of numerous key players, including Bayer and Janssen Pharmaceuticals, alongside emerging regional manufacturers, fosters a competitive landscape that drives innovation and market penetration. The growing adoption of Rivaroxaban for stroke prevention in patients with non-valvular atrial fibrillation also contributes substantially to its market value.
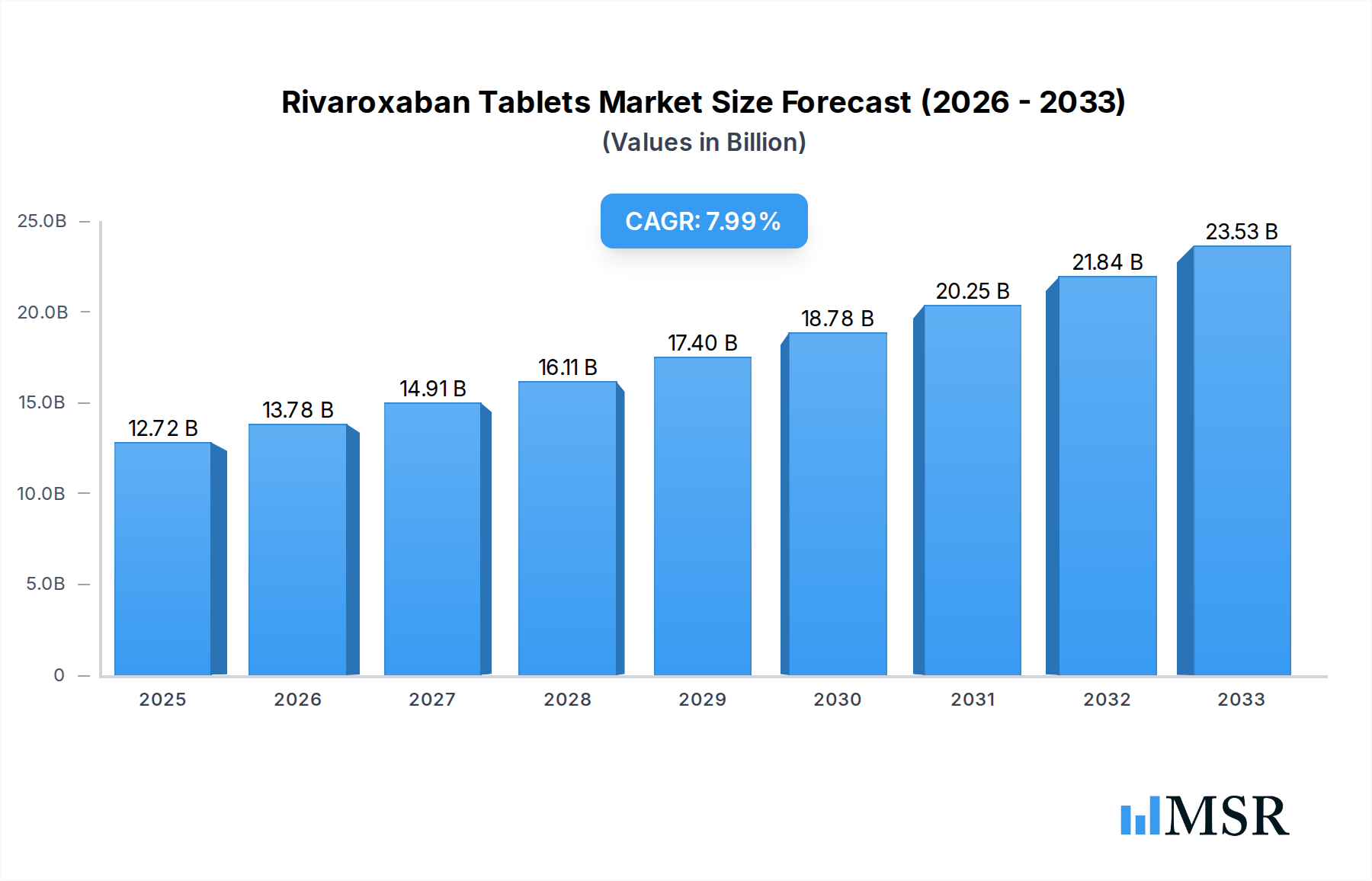
Rivaroxaban Tablets Market Size (In Billion)

The market's segmentation by application into hospitals, clinics, and drugstores highlights the diverse distribution channels for Rivaroxaban Tablets. Hospitals remain a primary consumption hub due to the critical nature of conditions treated, while clinics and drugstores cater to outpatient management and long-term treatment needs. The varying tablet strengths, such as 2.5mg, 10mg, 15mg, and 20mg, offer therapeutic flexibility, allowing for personalized treatment regimens. Geographically, North America and Europe currently lead the market, driven by advanced healthcare infrastructure, higher disease incidence, and strong reimbursement policies. However, the Asia Pacific region is anticipated to exhibit the fastest growth, attributed to a burgeoning patient population, increasing healthcare expenditure, and expanding access to modern therapeutics. While the market presents a promising outlook, factors such as the availability of generic alternatives and stringent regulatory approvals can act as potential restraints, necessitating strategic market approaches from key stakeholders.
Rivaroxaban Tablets Company Market Share

Here's an SEO-optimized and engaging report description for Rivaroxaban Tablets, designed to maximize visibility and attract industry stakeholders.
Rivaroxaban Tablets Market Concentration & Dynamics
The Rivaroxaban Tablets market exhibits a moderate to high concentration, with leading pharmaceutical giants like Bayer and Janssen Pharmaceuticals holding significant market share. The innovation ecosystem is characterized by ongoing research into novel formulations and expanded therapeutic applications, particularly in anticoagulant therapy. Regulatory frameworks, governed by bodies such as the FDA and EMA, play a crucial role in shaping market access and product approvals, influencing generic competition and the introduction of biosimilars. Substitute products, including other Direct Oral Anticoagulants (DOACs) and traditional anticoagulants like Warfarin, pose a continuous competitive threat, driving innovation and pricing strategies. End-user trends reveal a growing preference for convenient oral administration and reduced monitoring requirements, bolstering demand for Rivaroxaban. Mergers and Acquisitions (M&A) activity, while not consistently high, is strategic, aimed at expanding product portfolios and market reach. Estimated M&A deal counts for the historical period (2019-2024) are projected at 5, with a predicted rise to 7 within the forecast period (2025-2033), indicating potential consolidation and strategic partnerships. Market share for key players is dynamic, with estimates suggesting Bayer and Janssen collectively commanding over 70% of the global market in 2025.
Rivaroxaban Tablets Industry Insights & Trends
The Rivaroxaban Tablets industry is poised for robust expansion, driven by several interconnected factors. The aging global population and the increasing prevalence of cardiovascular diseases, including Atrial Fibrillation (AFib) and Venous Thromboembolism (VTE), are primary growth catalysts, creating a sustained demand for effective anticoagulant treatments. Technological disruptions are primarily centered around advancements in drug delivery systems and the development of more precise diagnostic tools to identify patient populations benefiting most from Rivaroxaban. Evolving consumer behaviors, influenced by increased health awareness and the desire for convenient self-administered medications, further propel the adoption of oral anticoagulants like Rivaroxaban over injectable alternatives. The global Rivaroxaban Tablets market size was valued at approximately 12 billion in 2023, with projections indicating a Compound Annual Growth Rate (CAGR) of 6.5% during the forecast period of 2025–2033. This growth trajectory is underpinned by increasing healthcare expenditure in emerging economies and expanding reimbursement policies for DOACs. The market is also witnessing a surge in research focused on off-label uses and the development of fixed-dose combinations to improve patient compliance and therapeutic outcomes, contributing significantly to market penetration and revenue generation. The ongoing competition from generic manufacturers after patent expiries in key regions will also influence pricing and accessibility, potentially driving volume growth.
Key Markets & Segments Leading Rivaroxaban Tablets
North America currently dominates the Rivaroxaban Tablets market, driven by a high prevalence of cardiovascular diseases, advanced healthcare infrastructure, and significant per capita healthcare spending. The United States represents the largest individual market within this region, fueled by a robust pharmaceutical market and widespread adoption of novel anticoagulants.
Dominant Segments:
- Application: Hospital: This segment is the primary driver due to the high incidence of VTE and AFib diagnoses, along with post-operative prophylaxis protocols that mandate anticoagulant therapy. The availability of sophisticated diagnostic tools and specialist cardiologists in hospital settings facilitates the initiation and management of Rivaroxaban treatment.
- Types: 20mg/Tablet: This strength is particularly crucial for treating conditions like AFib and preventing stroke, making it the most prescribed dosage. The effectiveness and established clinical outcomes associated with the 20mg strength solidify its market leadership.
Drivers for Dominance in North America & Hospital Segment:
- Economic Growth: Sustained economic prosperity in North America allows for higher healthcare expenditure and greater accessibility to advanced pharmaceutical treatments.
- Healthcare Infrastructure: A well-developed and accessible healthcare system, comprising leading research institutions and a vast network of hospitals and clinics, ensures broad patient reach.
- Technological Advancements: Early adoption of cutting-edge medical technologies, including advanced diagnostic imaging and personalized medicine approaches, supports the precise application of Rivaroxaban.
- Regulatory Support: Favorable regulatory pathways for drug approvals and post-market surveillance contribute to market stability and trust.
- High Disease Burden: The significant prevalence of target conditions like AFib and deep vein thrombosis in the North American population creates a substantial and ongoing demand.
The Clinic segment is also a significant contributor, reflecting the increasing trend towards outpatient management of chronic cardiovascular conditions. Drugstores play a vital role in the distribution of Rivaroxaban for ongoing patient treatment, driven by prescription refills and patient convenience. Other dosage strengths, such as 2.5mg/Tablet and 15mg/Tablet, cater to specific prophylactic and therapeutic needs, contributing to overall market breadth.
Rivaroxaban Tablets Product Developments
Rivaroxaban Tablets have seen continuous product development focused on enhancing patient compliance and therapeutic efficacy. Innovations include the development of fixed-dose combination therapies, such as Rivaroxaban with aspirin, for dual-pathway inhibition in specific cardiovascular patient groups, offering a simplified treatment regimen. Research is also exploring novel delivery systems and extended-release formulations to optimize pharmacokinetic profiles and minimize dosing frequency. The market relevance is underscored by ongoing clinical trials investigating Rivaroxaban for a wider range of thrombotic and embolic indications, further expanding its therapeutic applications and competitive edge in the anticoagulant landscape. The total market value for these product developments is estimated to be around 1.5 billion in research and development spending within the forecast period.
Challenges in the Rivaroxaban Tablets Market
The Rivaroxaban Tablets market faces several critical challenges. Intense competition from other DOACs and generic versions of Rivaroxaban exerts significant pricing pressure, potentially impacting profit margins. Stringent regulatory requirements for drug manufacturing and marketing, alongside complex reimbursement policies in various global markets, create barriers to entry and expansion. Supply chain disruptions, exacerbated by geopolitical factors and raw material availability, can affect product accessibility. Furthermore, patient adherence and physician prescribing patterns require continuous education and outreach to ensure optimal utilization and prevent suboptimal outcomes. The estimated impact of these challenges on market growth is a potential 2% reduction in the projected CAGR.
Forces Driving Rivaroxaban Tablets Growth
Several powerful forces are propelling the growth of the Rivaroxaban Tablets market. The increasing global burden of cardiovascular diseases, including Atrial Fibrillation and Venous Thromboembolism, directly translates into a greater need for effective anticoagulant therapies like Rivaroxaban. Technological advancements in diagnostics allow for earlier and more accurate identification of at-risk patients, expanding the potential patient pool. Furthermore, favorable regulatory environments in key markets, coupled with increasing healthcare expenditure and enhanced reimbursement policies for DOACs, are making these treatments more accessible and affordable. The convenience of oral administration and the reduced need for frequent monitoring, compared to traditional anticoagulants, are also significant drivers of physician and patient preference.
Challenges in the Rivaroxaban Tablets Market
Long-term growth catalysts for the Rivaroxaban Tablets market are deeply rooted in ongoing innovation and strategic market expansions. Continued investment in research and development for new indications and improved formulations will sustain its competitive advantage. Strategic partnerships and collaborations between pharmaceutical companies, healthcare providers, and technology firms can accelerate the adoption of Rivaroxaban through enhanced patient monitoring solutions and integrated care pathways. Furthermore, expansion into emerging markets with growing healthcare infrastructure and increasing awareness of cardiovascular disease management presents significant untapped potential. The predicted growth from these catalysts is estimated at 5 billion over the forecast period.
Emerging Opportunities in Rivaroxaban Tablets
Emerging trends and opportunities in the Rivaroxaban Tablets market are diverse and promising. The development of personalized medicine approaches, leveraging genetic profiling to identify individuals who would most benefit from Rivaroxaban, offers a significant avenue for growth. Expansion of its use in pediatric populations for specific thrombotic events presents a largely untapped market segment. Furthermore, the growing emphasis on integrated digital health platforms for remote patient monitoring and adherence management provides opportunities for enhanced patient care and data collection, which can inform future treatment strategies. The exploration of novel prophylactic applications in areas beyond traditional VTE and AFib treatment also holds considerable potential.
Leading Players in the Rivaroxaban Tablets Sector
- Bayer
- Janssen Pharmaceuticals
- Beijing SL Pharmaceutical
- Nanjing Zhengda Tianqing Pharmaceutical
- Fujian Cosunter Pharmaceutical
- Lunan Pharmaceutical
- Shanghai Huilun Pharmaceutical
- Jilin Boda Pharmaceutical
- Cspc Holdings Company
- Zhejiang Conba Pharmaceutical
- Suzhou No.3 Pharmaceutical
- Chengdu Baiyu Pharmaceuticals
- Jiangsu Zhongbang Pharmaceutical
Key Milestones in Rivaroxaban Tablets Industry
- 2019: Launch of expanded indications for Rivaroxaban in certain acute pulmonary embolism scenarios.
- 2020: Increased generic competition emerges in several European markets post-patent expiry.
- 2021: Introduction of real-world evidence studies highlighting the efficacy and safety of Rivaroxaban in diverse patient populations.
- 2022: Advancements in digital health tools for monitoring patient adherence and potential bleeding risks.
- 2023: Intensified focus on research for novel combinations and expanded prophylactic applications.
- 2024: Strategic acquisitions by smaller pharmaceutical entities to bolster their anticoagulant portfolios.
Strategic Outlook for Rivaroxaban Tablets Market
The strategic outlook for the Rivaroxaban Tablets market is highly positive, driven by a confluence of sustained demand and ongoing innovation. Growth accelerators will be primarily focused on expanding therapeutic applications through rigorous clinical research, particularly in areas like cancer-associated thrombosis and post-operative VTE prevention across a wider range of surgical procedures. Strategic opportunities also lie in leveraging digital health technologies to enhance patient outcomes and streamline treatment pathways, thereby improving adherence and reducing adverse events. Furthermore, the anticipated growth in emerging markets, coupled with increasing healthcare expenditure and a rising awareness of cardiovascular health, presents significant potential for market penetration and revenue expansion, projecting a market value of 18 billion by 2033.
Rivaroxaban Tablets Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Clinic
- 1.3. Drugstore
-
2. Types
- 2.1. 2.5mg/Tablet
- 2.2. 10mg/Tablet
- 2.3. 15mg/Tablet
- 2.4. 20mg/Tablet
Rivaroxaban Tablets Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
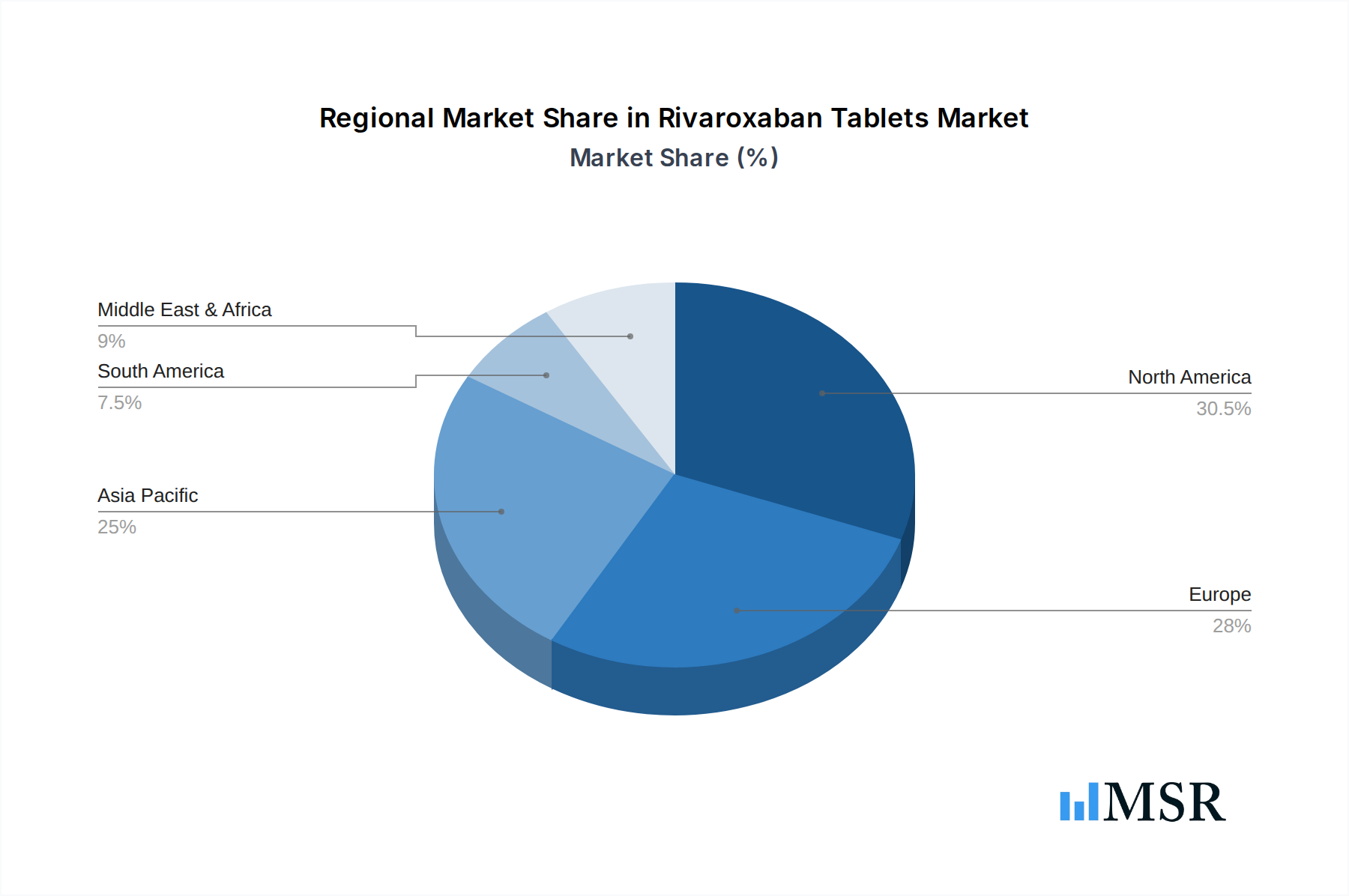
Rivaroxaban Tablets Regional Market Share

Geographic Coverage of Rivaroxaban Tablets
Rivaroxaban Tablets REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.17% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Rivaroxaban Tablets Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Clinic
- 5.1.3. Drugstore
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. 2.5mg/Tablet
- 5.2.2. 10mg/Tablet
- 5.2.3. 15mg/Tablet
- 5.2.4. 20mg/Tablet
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Rivaroxaban Tablets Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Clinic
- 6.1.3. Drugstore
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. 2.5mg/Tablet
- 6.2.2. 10mg/Tablet
- 6.2.3. 15mg/Tablet
- 6.2.4. 20mg/Tablet
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Rivaroxaban Tablets Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Clinic
- 7.1.3. Drugstore
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. 2.5mg/Tablet
- 7.2.2. 10mg/Tablet
- 7.2.3. 15mg/Tablet
- 7.2.4. 20mg/Tablet
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Rivaroxaban Tablets Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Clinic
- 8.1.3. Drugstore
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. 2.5mg/Tablet
- 8.2.2. 10mg/Tablet
- 8.2.3. 15mg/Tablet
- 8.2.4. 20mg/Tablet
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Rivaroxaban Tablets Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Clinic
- 9.1.3. Drugstore
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. 2.5mg/Tablet
- 9.2.2. 10mg/Tablet
- 9.2.3. 15mg/Tablet
- 9.2.4. 20mg/Tablet
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Rivaroxaban Tablets Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Clinic
- 10.1.3. Drugstore
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. 2.5mg/Tablet
- 10.2.2. 10mg/Tablet
- 10.2.3. 15mg/Tablet
- 10.2.4. 20mg/Tablet
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Bayer
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Janssen Pharmaceuticals
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Beijing SL Pharmaceutical
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Nanjing Zhengda Tianqing Pharmaceutical
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Fujian Cosunter Pharmaceutical
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Lunan Pharmaceutical
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Shanghai Huilun Pharmaceutical
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Jilin Boda Pharmaceutical
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Cspc Holdings Company
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Zhejiang Conba Pharmaceutical
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Suzhou No.3 Pharmaceutical
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Chengdu Baiyu Pharmaceuticals
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Jiangsu Zhongbang Pharmaceutical
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.1 Bayer
List of Figures
- Figure 1: Global Rivaroxaban Tablets Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Rivaroxaban Tablets Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Rivaroxaban Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Rivaroxaban Tablets Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Rivaroxaban Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Rivaroxaban Tablets Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Rivaroxaban Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Rivaroxaban Tablets Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Rivaroxaban Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Rivaroxaban Tablets Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Rivaroxaban Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Rivaroxaban Tablets Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Rivaroxaban Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Rivaroxaban Tablets Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Rivaroxaban Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Rivaroxaban Tablets Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Rivaroxaban Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Rivaroxaban Tablets Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Rivaroxaban Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Rivaroxaban Tablets Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Rivaroxaban Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Rivaroxaban Tablets Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Rivaroxaban Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Rivaroxaban Tablets Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Rivaroxaban Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Rivaroxaban Tablets Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Rivaroxaban Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Rivaroxaban Tablets Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Rivaroxaban Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Rivaroxaban Tablets Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Rivaroxaban Tablets Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Rivaroxaban Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Rivaroxaban Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Rivaroxaban Tablets Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Rivaroxaban Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Rivaroxaban Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Rivaroxaban Tablets Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Rivaroxaban Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Rivaroxaban Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Rivaroxaban Tablets Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Rivaroxaban Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Rivaroxaban Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Rivaroxaban Tablets Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Rivaroxaban Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Rivaroxaban Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Rivaroxaban Tablets Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Rivaroxaban Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Rivaroxaban Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Rivaroxaban Tablets Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Rivaroxaban Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Rivaroxaban Tablets?
The projected CAGR is approximately 8.17%.
2. Which companies are prominent players in the Rivaroxaban Tablets?
Key companies in the market include Bayer, Janssen Pharmaceuticals, Beijing SL Pharmaceutical, Nanjing Zhengda Tianqing Pharmaceutical, Fujian Cosunter Pharmaceutical, Lunan Pharmaceutical, Shanghai Huilun Pharmaceutical, Jilin Boda Pharmaceutical, Cspc Holdings Company, Zhejiang Conba Pharmaceutical, Suzhou No.3 Pharmaceutical, Chengdu Baiyu Pharmaceuticals, Jiangsu Zhongbang Pharmaceutical.
3. What are the main segments of the Rivaroxaban Tablets?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Rivaroxaban Tablets," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Rivaroxaban Tablets report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Rivaroxaban Tablets?
To stay informed about further developments, trends, and reports in the Rivaroxaban Tablets, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
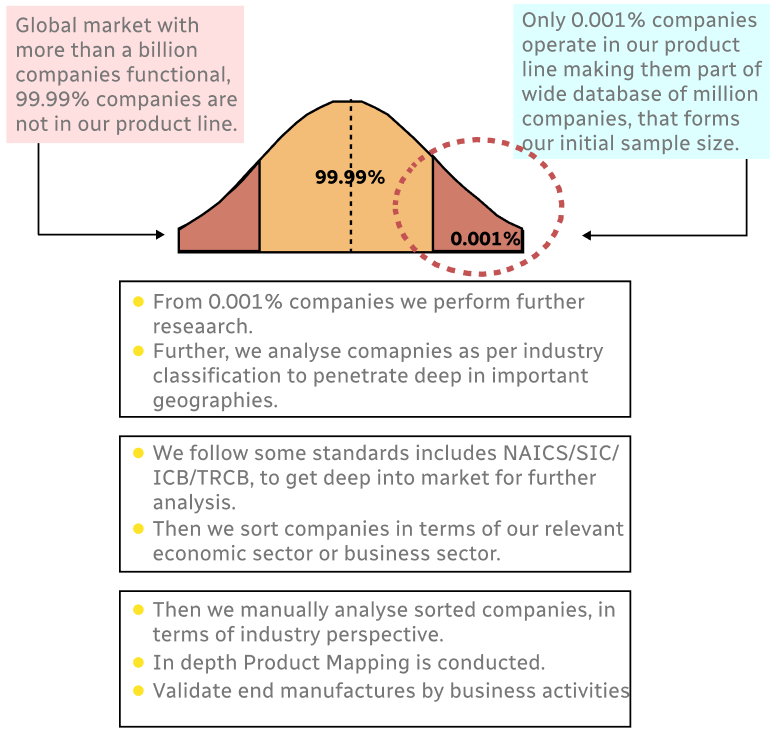
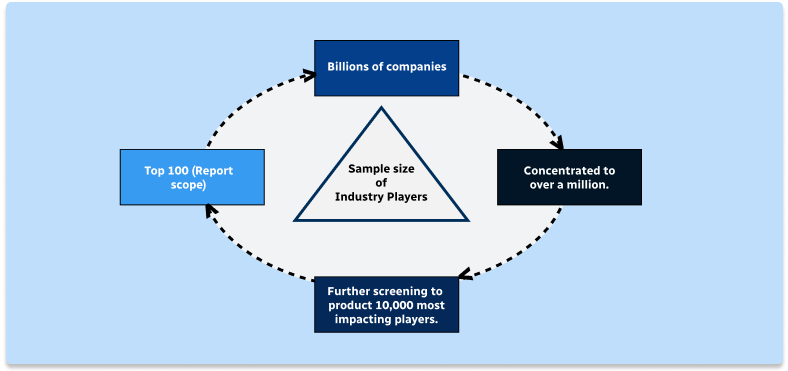
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
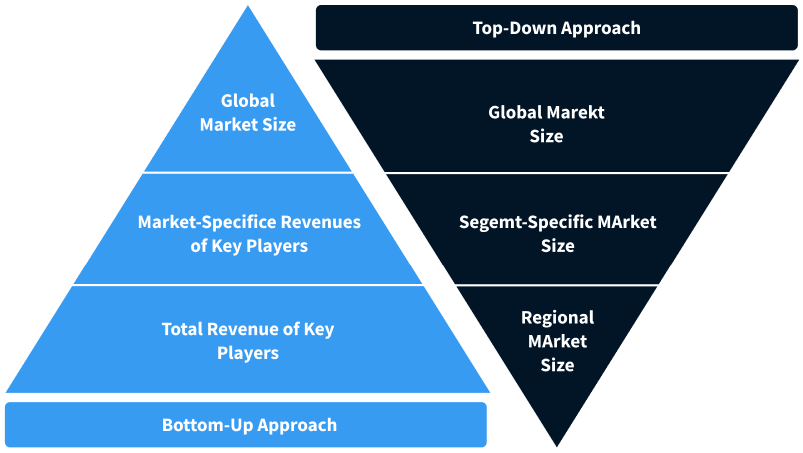
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
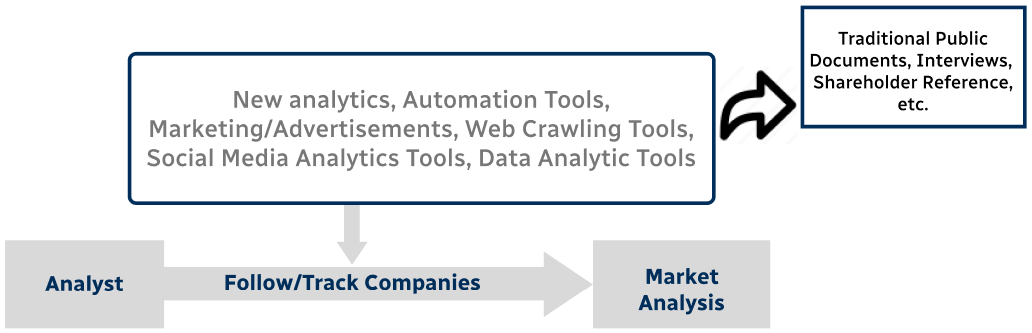
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


