Key Insights
The Oncology Based In Vivo CRO market is poised for significant expansion, projected to reach a substantial USD 2.28 billion in 2025, with an impressive Compound Annual Growth Rate (CAGR) of 10.9% during the forecast period of 2025-2033. This robust growth is propelled by a confluence of factors, primarily the escalating global cancer burden and the intensified R&D efforts by pharmaceutical and biopharmaceutical companies seeking novel therapeutic solutions. The increasing complexity of cancer biology, characterized by the emergence of resistant cell lines and diverse tumor types, necessitates sophisticated preclinical models for accurate drug efficacy and safety evaluation. Consequently, there is a growing demand for specialized Contract Research Organizations (CROs) that offer advanced in vivo oncology services. These services are crucial for understanding drug pharmacokinetics, pharmacodynamics, and toxicology, thereby de-risking the drug development pipeline and accelerating the journey from lab to clinic.
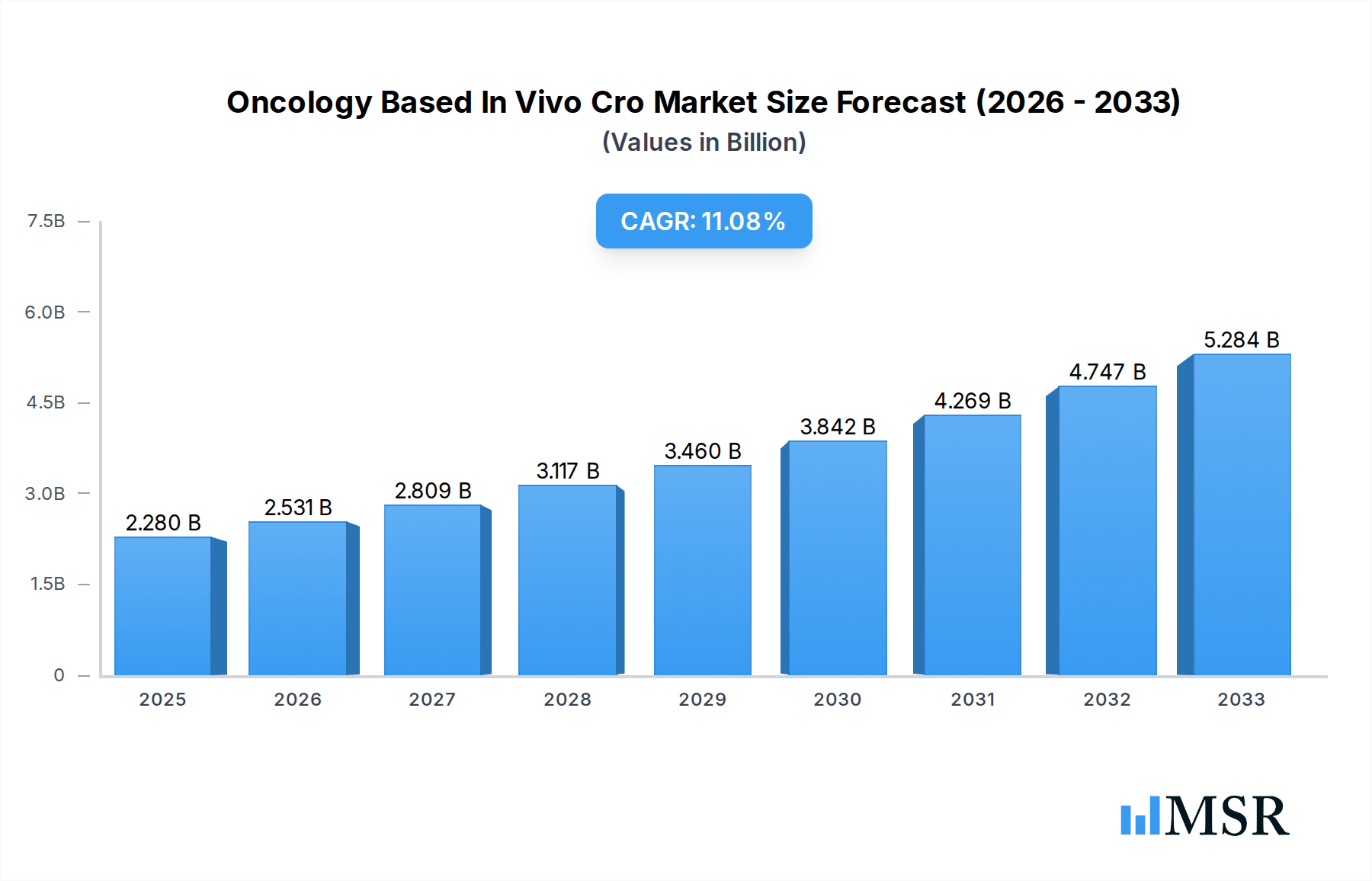
Oncology Based In Vivo Cro Market Size (In Billion)

The market's dynamism is further fueled by technological advancements in in vivo modeling, including the development of genetically engineered mouse models (GEMMs), patient-derived xenografts (PDXs), and advanced imaging techniques that provide deeper insights into tumor microenvironments and treatment responses. The increasing prevalence of blood cancers and solid tumors globally, coupled with a robust pipeline of oncology drugs in various stages of development, underpins the sustained demand for in vivo oncology research services. While the market enjoys strong growth, potential restraints such as the high cost of specialized in vivo studies and stringent regulatory hurdles could pose challenges. However, the increasing outsourcing trend by pharmaceutical and biopharmaceutical entities, driven by a need for specialized expertise and cost-efficiency, is expected to outweigh these limitations, ensuring a positive trajectory for the Oncology Based In Vivo CRO market.
Oncology Based In Vivo Cro Company Market Share

Here is your SEO-optimized, engaging report description for "Oncology Based In Vivo CRO," incorporating high-ranking keywords and adhering to all your specified requirements.
Oncology Based In Vivo CRO Market Research Report: Comprehensive Analysis 2019-2033
This in-depth market research report provides a definitive analysis of the Oncology Based In Vivo CRO market, offering critical insights for stakeholders in the pharmaceutical, biopharmaceutical, and medical device industries. Covering the period from 2019 to 2033, with a base year of 2025, this report delves into market dynamics, key trends, regional dominance, product innovations, challenges, and emerging opportunities within the oncology in vivo CRO services sector. Leveraging advanced analytics and proprietary data, this report offers actionable intelligence to guide strategic decision-making in this rapidly evolving landscape.
Oncology Based In Vivo Cro Market Concentration & Dynamics
The Oncology Based In Vivo CRO market exhibits a moderate concentration, with a few key players holding significant market share, alongside a growing number of specialized niche providers. Innovation ecosystems are robust, driven by continuous advancements in preclinical oncology models and assay development. Regulatory frameworks, primarily governed by agencies like the FDA and EMA, are stringent but are evolving to accommodate novel therapeutic modalities. Substitute products, such as advanced in vitro models and computational toxicology, are present but often serve complementary roles rather than direct replacements for in vivo efficacy and toxicity assessments. End-user trends highlight an increasing demand for complex patient-derived xenograft (PDX) models, immunotherapy models, and sophisticated biomarker analysis to accelerate drug development timelines. Mergers and acquisitions (M&A) are a significant driver of market consolidation and strategic expansion. For instance, the historical period saw approximately xx billion in M&A deal counts, reflecting a strategic push for enhanced capabilities and broader service offerings. The market share distribution sees Crown Bioscience and Charles River Laboratories leading, with significant contributions from ICON Plc. and Eurofins Scientific.
Key Market Dynamics:
- Market Share: Leading players like Crown Bioscience and Charles River Laboratories collectively hold an estimated xx billion market share.
- Innovation Ecosystems: Strong collaborations between CROs and academic institutions are fostering rapid development of next-generation in vivo models for oncology.
- Regulatory Frameworks: Evolving guidelines for novel therapies (e.g., cell and gene therapy) are shaping service requirements and demand for specialized expertise.
- Substitute Products: Advanced in vitro screening, organoids, and digital pathology are complementing but not fully replacing in vivo studies for efficacy validation.
- End-User Trends: Pharmaceutical and biopharmaceutical companies are increasingly outsourcing complex oncology studies to specialized CROs to leverage expertise and reduce internal costs.
- M&A Activities: Strategic acquisitions are focused on expanding service portfolios, geographical reach, and access to specialized technologies like AI-driven data analysis.
Oncology Based In Vivo Cro Industry Insights & Trends
The global Oncology Based In Vivo CRO market is experiencing robust growth, projected to reach a market size of over $xx billion by 2033, with a Compound Annual Growth Rate (CAGR) of approximately xx% from 2025 to 2033. This expansion is fueled by several critical factors. Firstly, the escalating global burden of cancer, coupled with a persistent need for novel and more effective cancer therapies, drives substantial investment in oncology drug discovery and development. Pharmaceutical and biopharmaceutical companies are increasingly reliant on Contract Research Organizations (CROs) to navigate the complexities of preclinical oncology research, including the development and validation of cutting-edge in vivo models. Technological disruptions are at the forefront of this growth. The advent of advanced preclinical models, such as genetically engineered mouse models (GEMMs), patient-derived xenografts (PDXs), and immunocompetent syngeneic models, allows for more accurate prediction of human responses to cancer treatments. Furthermore, the integration of artificial intelligence (AI) and machine learning (ML) into data analysis and experimental design is enhancing the efficiency and predictive power of in vivo studies, enabling faster identification of promising drug candidates. Evolving consumer behaviors, interpreted as the increasing demand from pharmaceutical companies for more personalized and precision oncology treatments, directly translate into a higher demand for specialized in vivo services that can assess drug efficacy in diverse genetic backgrounds and tumor microenvironments. The increasing complexity of cancer therapies, including immunotherapies, targeted therapies, and combination therapies, necessitates sophisticated in vivo testing platforms to evaluate synergistic effects and potential toxicities, further boosting market growth. The shift towards outsourcing non-core R&D activities by major pharmaceutical players to specialized CROs also plays a pivotal role, allowing them to focus on core competencies while benefiting from external expertise and infrastructure. The estimated market size for the Oncology Based In Vivo CRO market in 2025 is approximately $xx billion, with projected growth driven by these key industry trends. The increasing investment in oncology research, fueled by both public funding and private venture capital, is a significant market growth driver. Moreover, the growing pipeline of targeted therapies and immunotherapies necessitates rigorous preclinical validation, creating a sustained demand for in vivo CRO services. The report will further detail how these trends are shaping the competitive landscape and influencing strategic investments within the sector.
Key Markets & Segments Leading Oncology Based In Vivo Cro
The Oncology Based In Vivo CRO market is dominated by North America, driven by its established pharmaceutical and biopharmaceutical hubs, robust research infrastructure, and substantial R&D investments. The United States, in particular, represents a significant portion of the global market share, benefiting from a strong presence of leading pharmaceutical companies and innovative biotech startups.
Application Dominance:
- Pharmaceutical & Biopharmaceutical Industries: This segment is the primary driver of the Oncology Based In Vivo CRO market. Companies in this sector are continuously investing in the discovery and development of novel oncology therapeutics, ranging from small molecules and biologics to advanced cell and gene therapies. Their extensive R&D pipelines necessitate comprehensive preclinical in vivo testing for efficacy, pharmacokinetics, pharmacodynamics, and toxicology. The sheer volume of drug candidates and the complexity of oncology research within these industries create a perpetual demand for specialized CRO services. Economic growth in these sectors, coupled with favorable regulatory environments for drug approval, further solidifies their leading position.
- Medical Device Companies: While a smaller segment, medical device companies involved in oncology, such as those developing novel drug delivery systems or advanced diagnostic tools, also contribute to the market. Their need for in vivo studies is often focused on evaluating the performance and safety of their devices in a biological context.
- Others: This segment includes academic institutions and governmental research organizations that may engage CROs for specialized preclinical studies, particularly for early-stage research or when requiring access to advanced models or expertise not available in-house.
Type Dominance:
- Solid Tumors: The prevalence and complexity of solid tumors make this segment the largest contributor to the Oncology Based In Vivo CRO market. Research into lung cancer, breast cancer, colorectal cancer, prostate cancer, and other common solid malignancies represents a significant portion of the oncology drug development pipeline. The development of targeted therapies and immunotherapies for these tumor types requires extensive in vivo validation, often involving PDX models, GEMMs, and sophisticated biomarker analysis.
- Blood Cancer: While representing a smaller market share than solid tumors, the blood cancer segment (including leukemia, lymphoma, and multiple myeloma) is experiencing significant growth. The development of novel hematological oncology treatments, particularly in the realm of CAR-T cell therapies and targeted agents, drives demand for specialized in vivo models and assays that can accurately reflect the disease microenvironment.
- Other: This category includes less common or emerging cancer types where research is ongoing, as well as studies focusing on rare cancers or broad oncology research methodologies.
The dominance of North America is further reinforced by its well-established network of academic research institutions, a high concentration of venture capital funding for biotech innovation, and a proactive regulatory framework that facilitates clinical translation. Europe, particularly Germany, the UK, and Switzerland, also represents a significant market due to its strong pharmaceutical presence and a growing number of innovative biotech companies. Asia Pacific is emerging as a rapidly growing region, driven by increasing healthcare expenditure, a burgeoning biopharmaceutical sector, and a growing demand for outsourcing services from Western pharmaceutical giants. The report will provide a detailed breakdown of market share and growth projections for each region and key country, along with granular segment analysis.
Oncology Based In Vivo Cro Product Developments
Product developments in the Oncology Based In Vivo CRO market are characterized by continuous innovation in preclinical model development and assay technologies. Key advancements include the creation of more humanized and predictive in vivo models, such as patient-derived xenografts (PDXs) with detailed genomic and transcriptomic profiling, and genetically engineered mouse models (GEMMs) that recapitulate specific human cancer mutations. The integration of advanced imaging techniques like bioluminescence imaging (BLI), fluorescence imaging, and PET imaging allows for non-invasive monitoring of tumor growth and drug response in real-time. Furthermore, there's a growing focus on developing and validating complex immunotherapy models, including syngeneic models with intact immune systems and humanized mouse models engrafted with human immune cells, crucial for evaluating checkpoint inhibitors and other immunotherapeutic agents. Sophisticated biomarker discovery and validation platforms, coupled with advanced bioinformatics and AI-driven data analysis, are also key product developments, enabling deeper insights into drug mechanisms of action and patient stratification.
Challenges in the Oncology Based In Vivo Cro Market
The Oncology Based In Vivo CRO market faces several significant challenges that can impede growth and impact operational efficiency. High operational costs associated with maintaining specialized facilities, animal care, and highly skilled personnel represent a substantial financial barrier. Stringent and evolving regulatory requirements from bodies like the FDA and EMA necessitate continuous adaptation and investment in compliance, adding complexity and potential delays to study timelines. Ethical considerations and public perception surrounding animal research can lead to increased scrutiny and demand for robust ethical protocols and the exploration of alternative methods. Technical challenges such as the variability of animal models, the complexity of the tumor microenvironment, and the difficulty in accurately predicting human response in preclinical settings can lead to translation failures, impacting R&D success rates. Finally, intense competition among CROs, including both large, established players and smaller niche providers, exerts pricing pressure and demands continuous innovation to maintain a competitive edge.
- High Operational Costs: Significant investment in infrastructure, skilled personnel, and specialized animal models.
- Regulatory Hurdles: Compliance with evolving animal welfare regulations and drug development guidelines.
- Ethical Concerns: Public scrutiny and the imperative to develop and implement rigorous ethical protocols.
- Model Variability & Translation Challenges: Ensuring model reproducibility and accurate prediction of human efficacy and toxicity.
- Intense Competition: Pressure on pricing and the constant need for service differentiation.
Forces Driving Oncology Based In Vivo Cro Growth
Several powerful forces are propelling the Oncology Based In Vivo CRO market forward. A primary driver is the unmet medical need in oncology, characterized by the persistent burden of cancer globally and the continuous demand for more effective, targeted, and less toxic therapies. This fuels substantial investment in drug discovery and development by pharmaceutical and biopharmaceutical companies. Secondly, the increasing complexity of oncology drug pipelines, including novel immunotherapies, combination therapies, and targeted agents, necessitates highly specialized and sophisticated preclinical testing capabilities that CROs are uniquely positioned to provide. Furthermore, the global rise in outsourcing R&D activities by pharmaceutical companies to leverage external expertise, reduce operational costs, and accelerate development timelines is a significant growth accelerator. Technological advancements in preclinical model development, such as the creation of more predictive PDX and GEMM models, alongside advancements in imaging and bioinformatics, are enabling more robust and informative in vivo studies. The growth of emerging markets and the expansion of biopharmaceutical R&D activities in these regions also contribute to overall market expansion.
Challenges in the Oncology Based In Vivo Cro Market
Long-term growth catalysts in the Oncology Based In Vivo CRO market are intricately linked to ongoing scientific innovation and strategic market adaptations. The development and validation of highly predictive, humanized preclinical models that better recapitulate the complexity of human cancer, including the tumor microenvironment and the immune system, represent a significant long-term growth catalyst. Furthermore, the increasing sophistication of therapeutic modalities, such as gene therapies and advanced cell therapies, will demand specialized in vivo testing platforms and expertise, creating new avenues for CRO expansion. The integration of advanced technologies like AI and machine learning for predictive analytics, experimental design optimization, and data interpretation is poised to revolutionize preclinical research, enhancing efficiency and success rates, and thus driving long-term market value. Strategic partnerships and collaborations between CROs, pharmaceutical companies, and academic institutions will continue to foster innovation and accelerate the translation of research discoveries into clinical applications.
Emerging Opportunities in Oncology Based In Vivo Cro
Emerging opportunities within the Oncology Based In Vivo CRO market are vast and represent key areas for future growth and innovation. The expanding field of personalized medicine and precision oncology is creating a significant demand for in vivo services that can assess drug efficacy in specific patient genomic profiles and tumor subtypes, driving the development of highly tailored preclinical models. The rapid advancement of immunotherapies continues to fuel opportunities for CROs offering specialized models for evaluating immune checkpoint inhibitors, CAR-T cell therapies, and other complex immunomodulatory agents. The growing interest in combination therapy studies presents an opportunity for CROs to develop integrated platforms that can assess synergistic and antagonistic effects of multiple drug agents in vivo. Furthermore, the increasing focus on early-stage drug discovery and hit-to-lead optimization offers potential for CROs to provide high-throughput screening and rapid in vivo efficacy assessments. The burgeoning biopharmaceutical sector in emerging markets, particularly in Asia Pacific, presents substantial untapped potential for CROs looking to expand their global footprint and service offerings.
Leading Players in the Oncology Based In Vivo Cro Sector
- Crown Bioscience
- Charles River Laboratories
- ICON Plc.
- Eurofins Scientific
- Taconic Biosciences
- Covance
- EVOTEC
- The Jackson Laboratory
- Wuxi AppTec.
- MI Bioresearch, Inc.
- Champion Oncology, Inc.
- Xentech
Key Milestones in Oncology Based In Vivo Cro Industry
- 2019: Significant investment in advanced PDX model development by leading CROs, enabling more patient-specific research.
- 2020: Increased adoption of AI and machine learning for preclinical data analysis and predictive modeling in oncology.
- 2021: Emergence of specialized CROs focusing on complex immunotherapy models and combination therapy studies.
- 2022: Expansion of service offerings to include advanced biomarker analysis and translational research support.
- 2023: Strategic acquisitions by major CROs to enhance capabilities in gene editing technologies and humanized mouse models.
- 2024: Growing emphasis on integrated in vivo and in vitro platforms for comprehensive drug characterization.
- 2025 (Estimated): Further refinement of predictive preclinical models and increased demand for outsourcing of complex oncology studies.
- 2026-2033 (Forecast): Continued innovation in targeted therapy and immunotherapy models, driven by precision medicine trends and emerging therapeutic modalities.
Strategic Outlook for Oncology Based In Vivo Cro Market
The strategic outlook for the Oncology Based In Vivo CRO market is exceptionally positive, driven by sustained innovation and increasing demand from the pharmaceutical and biopharmaceutical sectors. Future growth will be significantly accelerated by the development and adoption of more predictive and translational preclinical models, including advanced PDX and GEMM platforms, and the integration of human immune components. The burgeoning field of precision oncology and the ongoing development of novel therapeutics like immunotherapies and combination therapies will continue to necessitate specialized in vivo services. CROs that invest in cutting-edge technologies, cultivate deep scientific expertise, and forge strategic partnerships are poised to capture substantial market share. Furthermore, the global expansion of R&D activities, particularly in emerging markets, presents significant opportunities for market penetration and growth. The ability to provide comprehensive, integrated services, from early-stage discovery to preclinical proof-of-concept, will be a key differentiator in this competitive landscape.
Oncology Based In Vivo Cro Segmentation
-
1. Application
- 1.1. Pharmaceutical & Biopharmaceutical Industries
- 1.2. Medical Device Companies
- 1.3. Others
-
2. Type
- 2.1. Blood Cancer
- 2.2. Solid Tumors
- 2.3. Other
Oncology Based In Vivo Cro Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
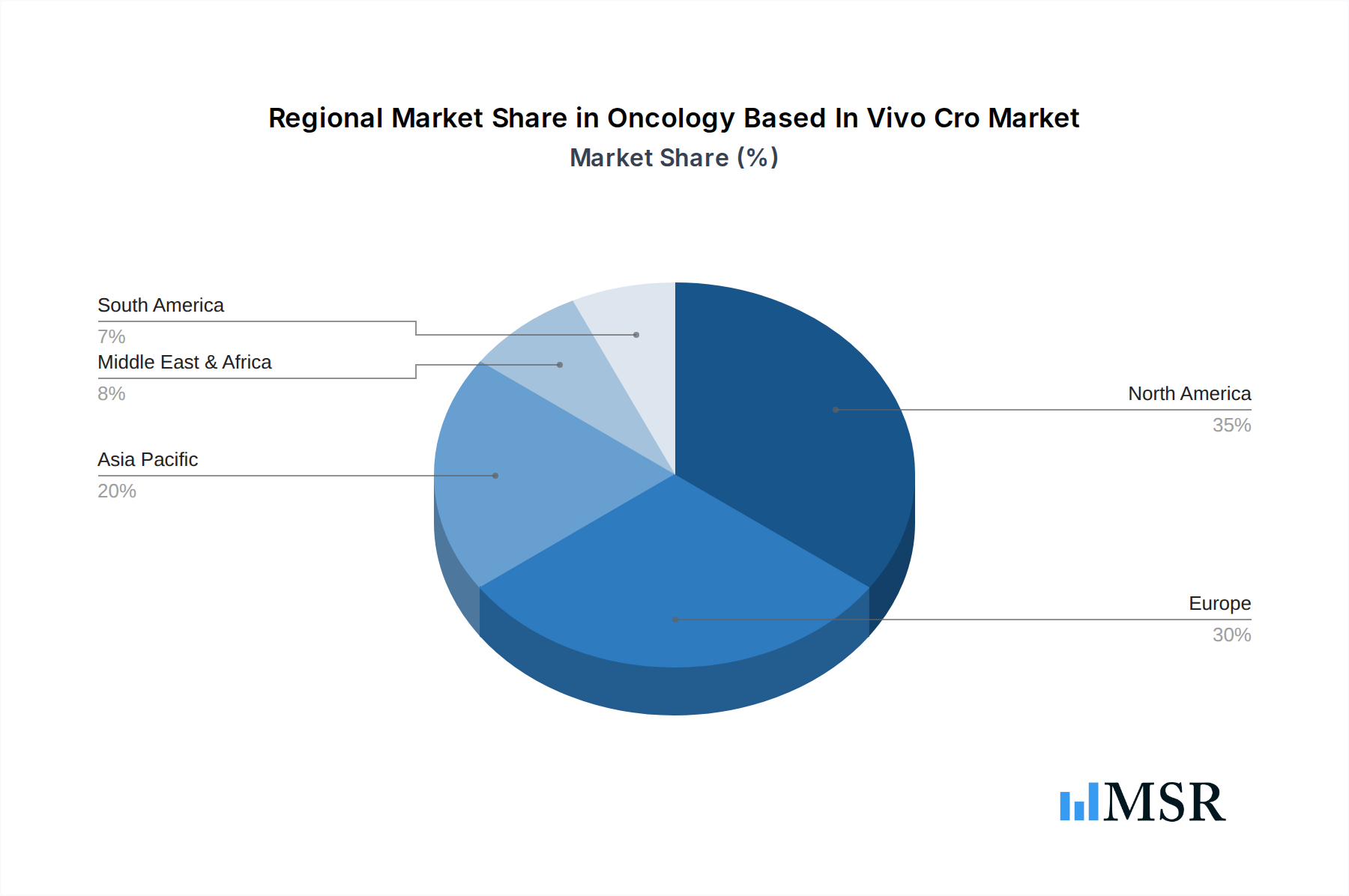
Oncology Based In Vivo Cro Regional Market Share

Geographic Coverage of Oncology Based In Vivo Cro
Oncology Based In Vivo Cro REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 10.9% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Oncology Based In Vivo Cro Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Pharmaceutical & Biopharmaceutical Industries
- 5.1.2. Medical Device Companies
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Blood Cancer
- 5.2.2. Solid Tumors
- 5.2.3. Other
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Oncology Based In Vivo Cro Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Pharmaceutical & Biopharmaceutical Industries
- 6.1.2. Medical Device Companies
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Blood Cancer
- 6.2.2. Solid Tumors
- 6.2.3. Other
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Oncology Based In Vivo Cro Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Pharmaceutical & Biopharmaceutical Industries
- 7.1.2. Medical Device Companies
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Blood Cancer
- 7.2.2. Solid Tumors
- 7.2.3. Other
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Oncology Based In Vivo Cro Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Pharmaceutical & Biopharmaceutical Industries
- 8.1.2. Medical Device Companies
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Blood Cancer
- 8.2.2. Solid Tumors
- 8.2.3. Other
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Oncology Based In Vivo Cro Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Pharmaceutical & Biopharmaceutical Industries
- 9.1.2. Medical Device Companies
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Blood Cancer
- 9.2.2. Solid Tumors
- 9.2.3. Other
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Oncology Based In Vivo Cro Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Pharmaceutical & Biopharmaceutical Industries
- 10.1.2. Medical Device Companies
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Blood Cancer
- 10.2.2. Solid Tumors
- 10.2.3. Other
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Crown Bioscience
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Charles River Laboratory
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 ICON Plc.
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Eurofins Scientific
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Taconic Biosciences
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Covance
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 EVOTEC
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 The Jackson Laboratory
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Wuxi AppTec.
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 MI Bioresearch Inc.
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Champion Oncology Inc.
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Xentech
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.1 Crown Bioscience
List of Figures
- Figure 1: Global Oncology Based In Vivo Cro Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Oncology Based In Vivo Cro Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Oncology Based In Vivo Cro Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Oncology Based In Vivo Cro Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America Oncology Based In Vivo Cro Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Oncology Based In Vivo Cro Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Oncology Based In Vivo Cro Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Oncology Based In Vivo Cro Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Oncology Based In Vivo Cro Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Oncology Based In Vivo Cro Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America Oncology Based In Vivo Cro Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Oncology Based In Vivo Cro Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Oncology Based In Vivo Cro Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Oncology Based In Vivo Cro Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Oncology Based In Vivo Cro Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Oncology Based In Vivo Cro Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe Oncology Based In Vivo Cro Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Oncology Based In Vivo Cro Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Oncology Based In Vivo Cro Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Oncology Based In Vivo Cro Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Oncology Based In Vivo Cro Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Oncology Based In Vivo Cro Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa Oncology Based In Vivo Cro Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Oncology Based In Vivo Cro Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Oncology Based In Vivo Cro Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Oncology Based In Vivo Cro Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Oncology Based In Vivo Cro Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Oncology Based In Vivo Cro Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific Oncology Based In Vivo Cro Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Oncology Based In Vivo Cro Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Oncology Based In Vivo Cro Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global Oncology Based In Vivo Cro Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Oncology Based In Vivo Cro Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Oncology Based In Vivo Cro?
The projected CAGR is approximately 10.9%.
2. Which companies are prominent players in the Oncology Based In Vivo Cro?
Key companies in the market include Crown Bioscience, Charles River Laboratory, ICON Plc., Eurofins Scientific, Taconic Biosciences, Covance, EVOTEC, The Jackson Laboratory, Wuxi AppTec., MI Bioresearch, Inc., Champion Oncology, Inc., Xentech.
3. What are the main segments of the Oncology Based In Vivo Cro?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Oncology Based In Vivo Cro," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Oncology Based In Vivo Cro report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Oncology Based In Vivo Cro?
To stay informed about further developments, trends, and reports in the Oncology Based In Vivo Cro, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
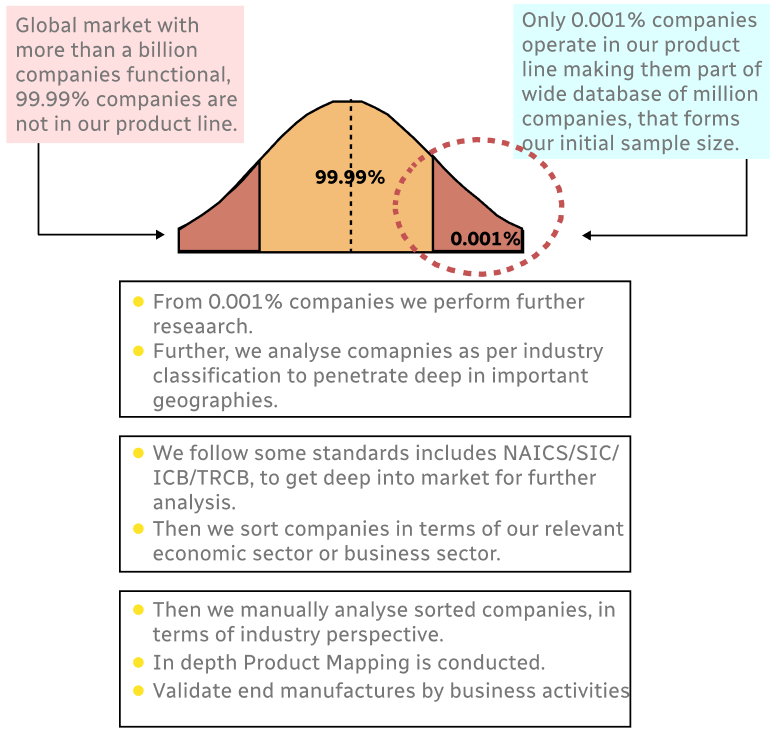
Step 1 - Identification of Relevant Samples Size from Population Database
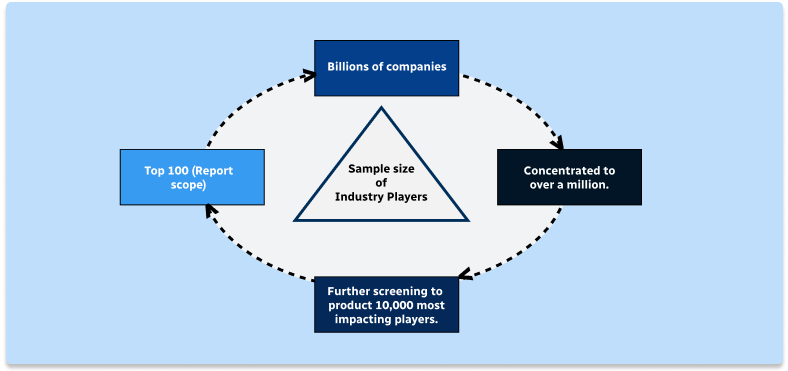
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
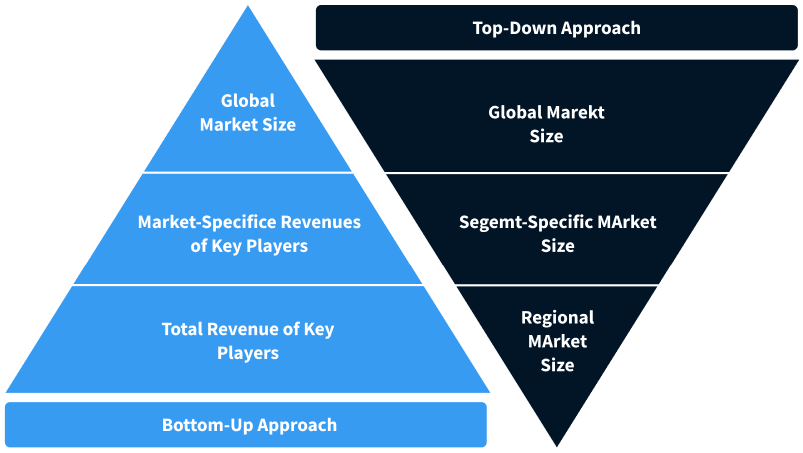
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
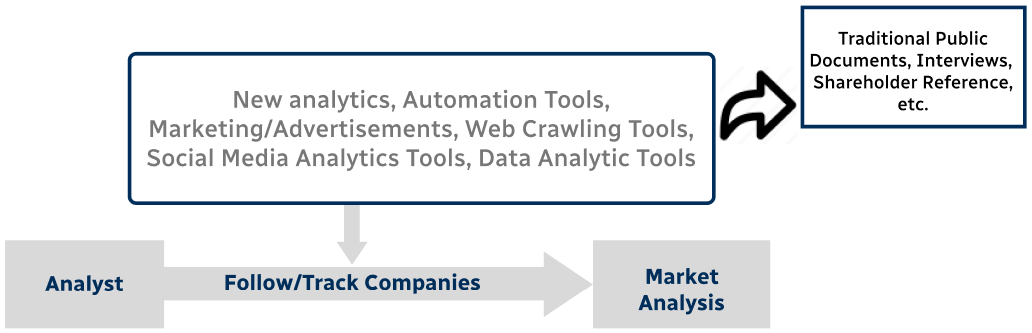
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


